Abstract
Rationale
The combination of CDP-choline, an α7 nicotinic acetylcholine receptor (α7 nAChR) agonist, with galantamine, a positive allosteric modulator of nAChRs, is believed to counter the fast desensitization rate of the α7 nAChRs and may be of interest for schizophrenia (SCZ) patients. Beyond the positive and negative clinical symptoms, deficits in early auditory prediction-error processes are also observed in SCZ. Regularity violations activate these mechanisms that are indexed by electroencephalography-derived mismatch negativity (MMN) event-related potentials (ERPs) in response to auditory deviance.
Objectives/methods
This pilot study in thirty-three healthy humans assessed the effects of an optimized α7 nAChR strategy combining CDP-choline (500 mg) with galantamine (16 mg) on speech-elicited MMN amplitude and latency measures. The randomized, double-blinded, placebo-controlled, and counterbalanced design with a baseline stratification method allowed for assessment of individual response differences.
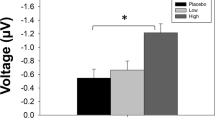
Results
Increases in MMN generation mediated by the acute CDP-choline/galantamine treatment in individuals with low baseline MMN amplitude for frequency, intensity, duration, and vowel deviants were revealed.
Conclusions
These results, observed primarily at temporal recording sites overlying the auditory cortex, implicate α7 nAChRs in the enhancement of speech deviance detection and warrant further examination with respect to dysfunctional auditory deviance processing in individuals with SCZ.










Similar content being viewed by others
References
Addington J, Addington D (1993) Premorbid functioning, cognitive functioning, symptoms and outcome in schizophrenia. J Psychiatry Neurosci 18:18–23
Adler LE, Olincy A, Cawthra EM et al (2004) Varied effects of atypical neuroleptics on P50 auditory gating in schizophrenia patients. Am J Psychiatry 161:1822–1828. https://doi.org/10.1176/appi.ajp.161.10.1822
Adler LE, Cawthra EM, Donovan KA et al (2005) Improved P50 auditory gating with ondansetron in medicated schizophrenia patients. Am J Psychiatry 162:386–388. https://doi.org/10.1176/appi.ajp.162.2.386
Aidelbaum R, Labelle A, Baddeley A, Knott V (2018) Assessing the acute effects of CDP-choline on sensory gating in schizophrenia: a pilot study. J Psychopharmacol 32:541–551. https://doi.org/10.1177/0269881117746903
Albuquerque EX, Pereira EFR, Braga MFM, Alkondon M (1998) Contribution of nicotinic receptors to the function of synapses in the central nervous system: the action of choline as a selective agonist of α7 receptors. J Physiol Paris 92:309–316. https://doi.org/10.1016/S0928-4257(98)80039-9
Askew CE, Metherate R (2016) Synaptic interactions and inhibitory regulation in auditory cortex. Biol Psychol 116:4–9. https://doi.org/10.1016/j.biopsycho.2015.11.001
Azzopardi E, Typlt M, Jenkins B, Schmid S (2013) Sensorimotor gating and spatial learning in α7-nicotinic receptor knockout mice. Genes Brain Behav 12:414–423. https://doi.org/10.1111/gbb.12038
Baldeweg T, Klugman A, Gruzelier JH, Hirsch SR (2002) Impairment in frontal but not temporal components of mismatch negativity in schizophrenia. Int J Psychophysiol 43:111–122. https://doi.org/10.1016/S0167-8760(01)00183-0
Baldeweg T, Klugman A, Gruzelier J, Hirsch SR (2004) Mismatch negativity potentials and cognitive impairment in schizophrenia. Schizophr Res 69:203–217. https://doi.org/10.1016/j.schres.2003.09.009
Baldeweg T, Wong D, Stephan KE (2006) Nicotinic modulation of human auditory sensory memory: evidence from mismatch negativity potentials. Int J Psychophysiol 59:49–58. https://doi.org/10.1016/j.ijpsycho.2005.07.014
Bali ZK, Nagy LV, Hernádi I (2017) Alpha7 nicotinic acetylcholine receptors play a predominant role in the cholinergic potentiation of N-methyl-D-aspartate evoked firing responses of hippocampal CA1 pyramidal cells. Front Cell Neurosci 11:1–13. https://doi.org/10.3389/fncel.2017.00271
Bali ZK, Nagy LV, Budai D, Hernádi I (2019) Facilitation and inhibition of firing activity and N-methyl-D-aspartate-evoked responses of CA1 hippocampal pyramidal cells by alpha7 nicotinic acetylcholine receptor selective compounds in vivo. Sci Rep 9:1–13. https://doi.org/10.1038/s41598-019-45796-7
Bertrand D, Gopalakrishnan M (2007) Allosteric modulation of nicotinic acetylcholine receptors. Biochem Pharmacol 74:1155–1163. https://doi.org/10.1016/j.bcp.2007.07.011
Bertrand D, Terry AV (2018) The wonderland of neuronal nicotinic acetylcholine receptors. Biochem Pharmacol 151:214–225. https://doi.org/10.1016/j.bcp.2017.12.008
Bodatsch M, Ruhrmann S, Wagner M et al (2011) Prediction of psychosis by mismatch negativity. Biol Psychiatry 69:959–966. https://doi.org/10.1016/j.biopsych.2010.09.057
Boggs D, Carlson J, Cortes-Briones J et al (2014) Going up in smoke? A review of nAChRs-based treatment strategies for improving cognition in schizophrenia. Curr Pharm Des 20:5077–5092. https://doi.org/10.2174/1381612819666131216121019
Boggs DL, Surti TS, Esterlis I et al (2018) Minimal effects of prolonged smoking abstinence or resumption on cognitive performance challenge the “self-medication” hypothesis in schizophrenia. Schizophr Res 194:62–69. https://doi.org/10.1016/j.schres.2017.03.047
Braff DL (2011) Gating in schizophrenia: from genes to cognition (to real world function?). Biol Psychiatry 69:395–396. https://doi.org/10.1016/j.biopsych.2011.01.002
Broide RS, Leslie FM (1999) The α7 nicotinic acetylcholine receptor in neuronal plasticity. Mol Neurobiol 20:1–16. https://doi.org/10.1007/BF02741361
Brown M, Kuperberg GR (2015a) A hierarchical generative framework of language processing: linking language perception, interpretation, and production abnormalities in schizophrenia. Front Hum Neurosci 9:643. https://doi.org/10.3389/fnhum.2015.00643
Brown M, Kuperberg GR (2015b) A hierarchical generative framework of language processing: linking language perception, interpretation, and production abnormalities in schizophrenia. Front Hum Neurosci 9:1–23. https://doi.org/10.3389/fnhum.2015.00643
Buchanan RW, Conley RR, Dickinson D et al (2008) Galantamine for the treatment of cognitive impairments in people with schizophrenia. Am J Psychiatry 165:82–89. https://doi.org/10.1176/appi.ajp.2007.07050724
Butler PD, Chen Y, Ford JM et al (2012) Perceptual measurement in schizophrenia: promising electrophysiology and neuroimaging paradigms from CNTRICS. Schizophr Bull:81–91
Castner SA, Smagin GN, Piser TM et al (2011) Immediate and sustained improvements in working memory after selective stimulation of α7 nicotinic acetylcholine receptors. Biol Psychiatry 69:12–18. https://doi.org/10.1016/j.biopsych.2010.08.006
Cather C, Pachas GN, Cieslak KM, Evins AE (2017) Achieving smoking cessation in individuals with schizophrenia: special considerations. CNS Drugs 31:471–481. https://doi.org/10.1007/s40263-017-0438-8
Changeux JP, Bertrand D, Corringer PJ et al (1998) Brain nicotinic receptors: structure and regulation, role in learning and reinforcement. Brain Res Brain Res Rev 26:198–216
Chou HH, Twamley E, Swerdlow NR (2012) Towards medication-enhancement of cognitive interventions in schizophrenia. Handb Exp Pharmacol 213:81–111. https://doi.org/10.1007/978-3-642-25758-2_4
Choueiry J, Blais CM, Shah D et al (2019a) Combining CDP-choline and galantamine: effects of a selective α7 nicotinic acetylcholine receptor agonist strategy on P50 sensory gating of speech sounds in healthy volunteers. J Psychopharmacol 33:688–699. https://doi.org/10.1177/0269881119836217
Choueiry J, Blais CM, Shah D et al (2019b) Combining CDP-choline and galantamine, an optimized α7 nicotinic strategy, to ameliorate sensory gating to speech stimuli in schizophrenia. Int J Psychophysiol 145:70–82. https://doi.org/10.1016/j.ijpsycho.2019.02.005
Court J, Spurden D, Lloyd S et al (1999) Neuronal nicotinic receptors in dementia with Lewy bodies and schizophrenia: alpha-bungarotoxin and nicotine binding in the thalamus. J Neurochem 73:1590–1597. https://doi.org/10.1046/j.1471-4159.1999.0731590.x
Coyle J, Balu D, Benneyworth M et al (2012) Beyond the dopamine receptor: novel therapeutic targets for treating schizophrenia. Dialogues Clin Neurosci 12:359–392
D’Souza MS, Markou A (2012) Schizophrenia and tobacco smoking comorbidity: NAChR agonists in the treatment of schizophrenia-associated cognitive deficits. Neuropharmacology 62:1564–1573
De La Salle S, Shah D, Choueiry J et al (2019) NMDA receptor antagonist effects on speech-related mismatch negativity and its underlying oscillatory and source activity in healthy humans. Front Pharmacol 10:1–21. https://doi.org/10.3389/fphar.2019.00455
Deutsch SI, Rosse RB, Schwartz BL et al (2008a) Effects of CDP-choline and the combination of CDP-choline and galantamine differ in an animal model of schizophrenia: development of a selective alpha7 nicotinic acetylcholine receptor agonist strategy. Eur Neuropsychopharmacol 18:147–151. https://doi.org/10.1016/j.euroneuro.2007.05.008
Deutsch SI, Schwartz BL, Schooler NR et al (2008b) First administration of cytidine diphosphocholine and galantamine in schizophrenia: a sustained alpha7 nicotinic agonist strategy. Clin Neuropharmacol 31:34–39. https://doi.org/10.1097/wnf.0b013e31806462ba
Deutsch SI, Schwartz BL, Schooler NR et al (2013) Targeting alpha-7 nicotinic neurotransmission in schizophrenia: a novel agonist strategy. Schizophr Res 148:138–144. https://doi.org/10.1016/j.schres.2013.05.023
Dulude L, Labelle A, Knott VJ (2010) Acute nicotine alteration of sensory memory impairment in smokers with schizophrenia. J Clin Psychopharmacol 30:541–548. https://doi.org/10.1097/JCP.0b013e3181f0c9c6
Dunbar G, Boeijinga PH, Demazières A et al (2007) Effects of TC-1734 (AZD3480), a selective neuronal nicotinic receptor agonist, on cognitive performance and the EEG of young healthy male volunteers. Psychopharmacology 191:919–929. https://doi.org/10.1007/s00213-006-0675-x
El Karoui I, King JR, Sitt J et al (2015) Event-related potential, time-frequency, and functional connectivity facets of local and global auditory novelty processing: an intracranial study in humans. Cereb Cortex 25:4203–4212. https://doi.org/10.1093/cercor/bhu143
Ettinger U, Williams SCR, Patel D et al (2009) Effects of acute nicotine on brain function in healthy smokers and non-smokers: estimation of inter-individual response heterogeneity. Neuroimage 45:549–561. https://doi.org/10.1016/j.neuroimage.2008.12.029
Featherstone RE, Siegel SJ (2015) The role of nicotine in schizophrenia, 1st edn. Elsevier
Fisher DJ, Salisbury DF (2019) The neurophysiology of schizophrenia: current update and future directions. Int J Psychophysiol 145:1–4. https://doi.org/10.1016/j.ijpsycho.2019.08.005
Fisher D, Labelle A, Knott V (2008) Auditory hallucinations and the mismatch negativity: processing speech and non-speech sounds in schizophrenia. Int J Psychophysiol 70:3–15
Fisher DJ, Grant B, Smith DM et al (2012) Nicotine and the hallucinating brain: effects on mismatch negativity (MMN) in schizophrenia. Psychiatry Res 196:181–187. https://doi.org/10.1016/j.psychres.2012.01.026
Fisher DJ, Rudolph ED, Ells EML et al (2019) Mismatch negativity-indexed auditory change detection of speech sounds in early and chronic schizophrenia. Psychiatry Res Neuroimaging 287:1–9. https://doi.org/10.1016/j.pscychresns.2019.03.010
Freedman R, Hall M, Adler LE, Leonard S (1995) Evidence in postmortem brain tissue for decreased numbers of hippocampal nicotinic receptors in schizophrenia. Biol Psychiatry 38:22–33. https://doi.org/10.1016/0006-3223(94)00252-X
Gareri P, Castagna A, Cotroneo AM et al (2015) The role of citicoline in cognitive impairment: pharmacological characteristics, possible advantages, and doubts for an old drug with new perspectives. Clin Interv Aging 10:1421–1429. https://doi.org/10.2147/CIA.S87886
Gareri P, Castagna A, Cotroneo AM et al (2017) The citicholinage study: citicoline plus cholinesterase inhibitors in aged patients affected with Alzheimer’s disease study. J Alzheimers Dis 56:557–565. https://doi.org/10.3233/JAD-160808
Gee KW, Olincy A, Kanner R et al (2017) First in human trial of a type I positive allosteric modulator of alpha7-nicotinic acetylcholine receptors: pharmacokinetics, safety, and evidence for neurocognitive effect of AVL-3288. J Psychopharmacol 31:434–441. https://doi.org/10.1177/0269881117691590
Gilbert DG, Gilbert BO (1995) Personality, psychopathology, and nicotine response as mediators of the genetics of smoking. Behav Genet 25:133–147. https://doi.org/10.1007/BF02196923
Gratton G, Coles MGH, Donchin E (1983) A new method for off-line removal of ocular artifact. Electroencephalogr Clin Neurophysiol 55:468–484. https://doi.org/10.1016/0013-4694(83)90135-9
Green MF, Kern RS, Braff DL, Mintz J (2000) Neurocognitive deficits and functional outcome in schizophrenia: are we measuring the “right stuff”? Schizophr Bull 26:119–136. https://doi.org/10.1093/oxfordjournals.schbul.a033430
Green MF, Kern RS, Heaton RK (2004) Longitudinal studies of cognition and functional outcome in schizophrenia: implications for MATRICS. Schizophr Res:41–51
Green MF, Butler PD, Chen Y et al (2009) Perception measurement in clinical trials of schizophrenia: promising paradigms from CNTRICS. Schizophr Bull 35:163–181
Greenwood LM, Leung S, Michie PT et al (2018) The effects of glycine on auditory mismatch negativity in schizophrenia. Schizophr Res 191:61–69. https://doi.org/10.1016/j.schres.2017.05.031
Guan ZZ, Zhang X, Ravid R, Nordberg A (2000) Decreased protein levels of nicotinic receptor subunits in the hippocampus and temporal cortex of patients with Alzheimer’s disease. J Neurochem 74:237–243. https://doi.org/10.1046/j.1471-4159.2000.0740237.x
Hahn B, Shoaib M, Stolerman IP (2002) Nicotine-induced enhancement of attention in the five-choice serial reaction time task: the influence of task demands. Psychopharmacology. https://doi.org/10.1007/s00213-002-1005-6
Hamilton HK, D’Souza DC, Ford JM et al (2018) Interactive effects of an N-methyl-D-aspartate receptor antagonist and a nicotinic acetylcholine receptor agonist on mismatch negativity: implications for schizophrenia. Schizophr Res 191:87–94. https://doi.org/10.1016/j.schres.2017.06.040
Harkrider AW, Hedrick MS (2005) Acute effect of nicotine on auditory gating in smokers and non-smokers. Hear Res 202:114–128. https://doi.org/10.1016/j.heares.2004.11.009
Hashimoto K (2015) Targeting of α7 nicotinic acetylcholine receptors in the treatment of schizophrenia and the use of auditory sensory gating as a translational biomarker. Curr Pharm Des 21:3797–3806. https://doi.org/10.1016/j.bcp.2015.08.030
Hofer A, Baumgartner S, Bodner T et al (2005) Patient outcomes in schizophrenia II: the impact of cognition. Eur Psychiatry 20:395–402. https://doi.org/10.1016/j.eurpsy.2005.02.006
Hyde M, Choueiry J, Smith D et al (2016) Cholinergic modulation of auditory P3 event-related potentials as indexed by CHRNA4 and CHRNA7 genotype variation in healthy volunteers. Neurosci Lett 623:36–41. https://doi.org/10.1016/j.neulet.2016.04.040
Inami R, Kirino E (2019a) Nicotine effect on mismatch negativity in smoking and nonsmoking patients with schizophrenia. Schizophr Res:1–2. https://doi.org/10.1016/j.schres.2019.01.002
Inami R, Kirino E (2019b) Nicotine effect on mismatch negativity in smoking and nonsmoking patients with schizophrenia. Schizophr Res:1–2. https://doi.org/10.1016/j.schres.2019.01.002
Inami R, Kirino E, Inoue R, Arai H (2005) Transdermal nicotine administration enhances automatic auditory processing reflected by mismatch negativity. Pharmacol Biochem Behav 80:453–461. https://doi.org/10.1016/j.pbb.2005.01.001
Inami R, Kirino E, Inoue R et al (2008) Nicotine effects on mismatch negativity in nonsmoking schizophrenic patients. Neuropsychobiology 56:64–72. https://doi.org/10.1159/000111536
Javitt D (1996) Role of cortical N-methyl-D-aspartate receptors in auditory sensory memory and mismatch negativity generation: implications for schizophrenia. Proc Natl Acad Sci U S A 93:11962–11967
Javitt DC (2000) Intracortical mechanisms of mismatch negativity dysfunction in schizophrenia. Audiol Neuro-Otol 5:207–215. https://doi.org/10.1159/000013882
Javitt DC, Freedman R (2015) Sensory processing dysfunction in the personal experience and neuronal machinery of schizophrenia. Am J Psychiatry 172:17–31. https://doi.org/10.1176/appi.ajp.2014.13121691
Javitt D, Sweet R (2015) Auditory dysfunction in schizophrenics: integrating clinical and basic features. Nat Rev Neurosci 16:535–550
Javitt D, Zukin S, Heresco-Levy U, Umbricht D (2012) Has an angel shown the way? Etiological and therapeutic amplifications of the PCP/NMDA model of schizophrenia. Schizophr Bull 38:958–966
Javitt DC, Lee M, Kantrowitz JT, Martinez A (2018) Mismatch negativity as a biomarker of theta band oscillatory dysfunction in schizophrenia. Schizophr Res 191:51–60. https://doi.org/10.1016/j.schres.2017.06.023
Jones LA, Hills PJ, Dick KM et al (2016) Cognitive mechanisms associated with auditory sensory gating. Brain Cogn 102:33–45. https://doi.org/10.1016/j.bandc.2015.12.005
Jurcak V, Tsuzuki D, Dan I (2007) 10/20, 10/10, and 10/5 systems revisited: their validity as relative head-surface-based positioning systems. Neuroimage 34:1600–1611. https://doi.org/10.1016/j.neuroimage.2006.09.024
Kantrowitz JT (2018) N-methyl-D-aspartate-type glutamate receptor modulators and related medications for the enhancement of auditory system plasticity in schizophrenia. Schizophr Res. https://doi.org/10.1016/j.schres.2018.02.003
Kantrowitz J, Javitt D (2010) N-methyl-D-aspartate (NMDA) receptor dysfunction or dysregulation: the final common pathway on the road to schizophrenia? Brain Res Bull 30:108–121
Kantrowitz J, Hoptman M, Leitman D et al (2015) Neural substrates of auditory emotion recognition deficits in schizophrenia. J Neurosci 35:14909–14921
Kantrowitz JT, Epstein ML, Beggel O et al (2016) Neurophysiological mechanisms of cortical plasticity impairments in schizophrenia and modulation by the NMDA receptor agonist D-serine. Brain 139:3281–3295. https://doi.org/10.1093/brain/aww262
Kantrowitz JT, Epstein ML, Lee M et al (2018) Improvement in mismatch negativity generation during D-serine treatment in schizophrenia: correlation with symptoms. Schizophr Res 191:70–79. https://doi.org/10.1016/j.schres.2017.02.027
Kantrowitz JT, Javitt DC, Freedman R et al (2020) Double blind, two dose, randomized, placebo-controlled, cross-over clinical trial of the positive allosteric modulator at the alpha7 nicotinic cholinergic receptor AVL-3288 in schizophrenia patients. Neuropsychopharmacology. https://doi.org/10.1038/s41386-020-0628-9
Kasai K, Nakagome K, Itoh K et al (2002) Impaired cortical network for preattentive detection of change in speech sounds in schizophrenia: a high-resolution event-related potential study. Am J Psychiatry 159(4)
Kawakubo Y, Kasai K, Kudo N et al (2006) Phonetic mismatch negativity predicts verbal memory deficits in schizophrenia. Neuroreport 17:1043–1046
Kawakubo Y, Kamco S, Nose T et al (2007) Phonetic mismatch negativity predicts social skills acquisition in schizophrenia. Psychiatry Res 152:261–265
Kem WR, Olincy A, Johnson L et al (2018) Pharmacokinetic limitations on effects of an alpha7-nicotinic receptor agonist in schizophrenia: randomized trial with an extended-release formulation. Neuropsychopharmacology 43:583–589. https://doi.org/10.1038/npp.2017.182
Knott V, Fisher D, Millar A (2010) Differential effects of nicotine on P50 amplitude, its gating, and their neural sources in low and high suppressors. Neuroscience 170:816–826. https://doi.org/10.1016/j.neuroscience.2010.07.012
Knott, Millar, McIntosh et al (2011) Separate and combined effects of low dose ketamine and nicotine on behavioural and neural correlates of sustained attention. Biol Psychol 88:83–93. https://doi.org/10.1016/j.biopsycho.2011.06.012
Knott VJ, De La Salle S, Smith D et al (2013) Baseline dependency of nicotine’s sensory gating actions: similarities and differences in low, medium and high P50 suppressors. J Psychopharmacol 27:790–800. https://doi.org/10.1177/0269881113490449
Knott V, Impey D, Philippe T et al (2014a) Modulation of auditory deviance detection by acute nicotine is baseline and deviant dependent in healthy nonsmokers: a mismatch negativity study. Hum Psychopharmacol 29:446–458. https://doi.org/10.1002/hup.2418
Knott V, Smith D, De La Salle S et al (2014b) CDP-choline: effects of the procholine supplement on sensory gating and executive function in healthy volunteers stratified for low, medium and high P50 suppression. J Psychopharmacol 28:1095–1108. https://doi.org/10.1177/0269881114553254
Knott V, De La Salle S, Choueiry J et al (2015a) Neurocognitive effects of acute choline supplementation in low, medium and high performer healthy volunteers. Pharmacol Biochem Behav 131:119–129. https://doi.org/10.1016/j.pbb.2015.02.004
Knott V, Impey D, Choueiry J et al (2015b) An acute dose, randomized trial of the effects of CDP-choline on mismatch negativity (MMN) in healthy volunteers stratified by deviance detection level. Neuropsychiatr Electrophys:1. https://doi.org/10.1186/s40810-014-0002-4
Kohlhaas KL, Robb HM, Roderwald VA, Rueter LE (2015) Nicotinic modulation of auditory evoked potential electroencephalography in a rodent neurodevelopmental model of schizophrenia. Biochem Pharmacol 97:482–487. https://doi.org/10.1016/j.bcp.2015.05.011
Koola MM (2018) Attenuated mismatch negativity in attenuated psychosis syndrome predicts psychosis: can galantamine-memantine combination prevent psychosis? Mol Neuropsychiatry 4:71–74. https://doi.org/10.4088/PCC.17nr02235
Koychev I, Mcmullen K, Lees J et al (2012) A validation of cognitive biomarkers for the early identification of cognitive enhancing agents in schizotypy: a three-center double-blind placebo-controlled study. Eur Neuropsychopharmacol 22:469–481. https://doi.org/10.1016/j.euroneuro.2011.10.005
Kumari V, Postma P (2005) Nicotine use in schizophrenia: the self medication hypotheses. Neurosci Biobehav Rev 25:1021–1034. https://doi.org/10.1016/j.neubiorev.2005.02.006
Kupferschmidt DA, Funk D, Erb S, Lê AD (2010) Age-related effects of acute nicotine on behavioural and neuronal measures of anxiety. Behav Brain Res 213:288–292. https://doi.org/10.1016/j.bbr.2010.05.022
Leonard S, Freedman R (2006) Genetics of chromosome 15q13-q14 in schizophrenia. Biol Psychiatry 60:115–122. https://doi.org/10.1016/j.biopsych.2006.03.054
Levin ED (2012) alpha7-Nicotinic receptors and cognition. Curr Drug Targets 13:602–606
Lewis AS, van Schalkwyk GI, Bloch MH (2017) Alpha-7 nicotinic agonists for cognitive deficits in neuropsychiatric disorders: a translational meta-analysis of rodent and human studies. Prog Neuro-Psychopharmacol Biol Psychiatry 75:45–53. https://doi.org/10.1016/j.pnpbp.2017.01.001
Li M, Mead A, Bevins RA (2009) Individual differences in responses to nicotine: tracking changes from adolescence to adulthood. Acta Pharmacol Sin 30:868–878. https://doi.org/10.1038/aps.2009.55
Light GA, Swerdlow NR, Thomas ML et al (2015) Validation of mismatch negativity and P3a for use in multi-site studies of schizophrenia: characterization of demographic, clinical, cognitive, and functional correlates in COGS-2. Schizophr Res 163:63–72. https://doi.org/10.1016/j.schres.2014.09.042
Mackowick KM, Barr MS, Wing VC et al (2014) Neurocognitive endophenotypes in schizophrenia: modulation by nicotinic receptor systems. Prog Neuro-Psychopharmacol Biol Psychiatry 52:79–85. https://doi.org/10.1016/j.pnpbp.2013.07.010
Marcus MM, Björkholm C, Malmerfelt A et al (2016) Alpha7 nicotinic acetylcholine receptor agonists and PAMs as adjunctive treatment in schizophrenia. An experimental study. Eur Neuropsychopharmacol 26:1401–1411. https://doi.org/10.1016/j.euroneuro.2016.07.004
Martin LF, Davalos DB, Kisley MA (2009) Nicotine enhances automatic temporal processing as measured by the mismatch negativity waveform. Nicotine Tob Res 11:698–706. https://doi.org/10.1093/ntr/ntp052
Martin-Ruiz CM, Haroutunian VH, Long P et al (2003) Dementia rating and nicotinic receptor expression in the prefrontal cortex in schizophrenia. Biol Psychiatry 54:1222–1233. https://doi.org/10.1016/S0006-3223(03)00348-2
Mathalon DH, Ahn KH, Perry EB et al (2014) Effects of nicotine on the neurophysiological and behavioral effects of ketamine in humans. Front Psychiatry 5. https://doi.org/10.3389/fpsyt.2014.00003
Maxwell E (1992) Family interview for genetic studies (FIGS)
Mike A, Castro NG, Albuquerque EX (2000) Choline and acetylcholine have similar kinetic properties of activation and desensitization on the α7 nicotinic receptors in rat hippocampal neurons. Brain Res. https://doi.org/10.1016/S0006-8993(00)02863-8
Moghaddam R, Javitt D (2012) From revolution to evolution: the glutamate hypothesis of schizophrenia and its implications for treatment. Neuropsychopharmacology 37:4–15
Näätänen R, Kähkönen S (2009) Central auditory dysfunction in schizophrenia as revealed by the mismatch negativity (MMN) and its magnetic equivalent MMNm: a review. Int J Neuropsychopharmacol 12:125–135. https://doi.org/10.1017/S1461145708009322
Näätänen R, Pakarinen S, Rinne T, Takegata R (2004) The mismatch negativity (MMN): towards the optimal paradigm. Clin Neurophysiol 115:140–144. https://doi.org/10.1093/joc/54.3.402
Näätänen R, Kujala T, Escera C et al (2012) The mismatch negativity (MMN)--a unique window to disturbed central auditory processing in ageing and different clinical conditions. Clin Neurophysiol 123:424–458. https://doi.org/10.1016/j.clinph.2011.09.020
Näätänen R, Todd J, Schall U (2016) Mismatch negativity (MMN) as biomarker predicting psychosis in clinically at-risk individuals. Biol Psychol 116:36–40. https://doi.org/10.1016/j.biopsycho.2015.10.010
Nagai T, Tada M, Kirihara K et al (2013) Mismatch negativity as a “translatable” brain marker toward early intervention for psychosis: a review. Front Psychiatry 4:115. https://doi.org/10.3389/fpsyt.2013.00115
Newhouse PA, Potter A, Singh A (2004) Effects of nicotinic stimulation on cognitive performance. Curr Opin Pharmacol 4:36–46. https://doi.org/10.1016/j.coph.2003.11.001
Newhouse PA, Potter AS, Dumas JA, Thiel CM (2011) Functional brain imaging of nicotinic effects on higher cognitive processes. Biochem Pharmacol:943–951
Paavilainen P, Alho K, Reinikainen K, Sams M, Näätänen R (1991) Right hemisphere dominance of different mismatch negativities. Electroencephalogr Clin Neurophysiol 78:466–479
Pakarinen S, Lovio R, Huotilainen M et al (2009) Fast multi-feature paradigm for recording several mismatch negativities (MMNs) to phonetic and acoustic changes in speech sounds. Biol Psychol 82:219–226. https://doi.org/10.1016/j.biopsycho.2009.07.008
Pakarinen S, Sokka L, Leinikka M et al (2014) Fast determination of MMN and P3a responses to linguistically and emotionally relevant changes in pseudoword stimuli. Neurosci Lett 577:28–33. https://doi.org/10.1016/j.neulet.2014.06.004
Perez VB, Woods SW, Roach BJ et al (2014) Automatic auditory processing deficits in schizophrenia and clinical high-risk patients: forecasting psychosis risk with mismatch negativity. Biol Psychiatry 75:459–469. https://doi.org/10.1016/j.biopsych.2013.07.038
Perkins KA (1995) Individual variability in responses to nicotine. Behav Genet 25:119–132. https://doi.org/10.1007/BF02196922
Perkins KA (1999) Baseline-dependency of nicotine affects: a review. Behav Pharmacol 10:597–615
Perkins KA (2009) Sex differences in nicotine reinforcement and reward: influences on the persistence of tobacco smoking. In: Bevins RA, Caggiuls AR (eds) Motivational impact of nicotine and its role in tobacco use. Springer, New York, pp 143–169
Petrovsky N, Ettinger U, Quednow BB et al (2012) Nicotine differentially modulates antisaccade performance in healthy male non-smoking volunteers stratified for low and high accuracy. Psychopharmacology 221:27–38. https://doi.org/10.1007/s00213-011-2540-9
Picciotto MR (2003) Nicotine as a modulator of behavior: beyond the inverted U. Trends Pharmacol Sci 24:493–499
Poltavski DV, Petros T (2005) Effects of transdermal nicotine on prose memory and attention in smokers and nonsmokers. Physiol Behav 83:833–843. https://doi.org/10.1016/j.physbeh.2004.10.005
Poorthuis RB, Mansvelder HD (2013) Nicotinic acetylcholine receptors controlling attention: behavior, circuits and sensitivity to disruption by nicotine. Biochem Pharmacol 86:1089–1098. https://doi.org/10.1016/j.bcp.2013.07.003
Potasiewicz A, Nikiforuk A, Hołuj M, Popik P (2017) Stimulation of nicotinic acetylcholine alpha7 receptors rescue schizophrenia-like cognitive impairments in rats. J Psychopharmacol 31:260–271. https://doi.org/10.1177/0269881116675509
Preskorn SH, Gawryl M, Dgetluck N et al (2014) Normalizing effects of EVP-6124, an alpha-7 nicotinic partial agonist, on event-related potentials and cognition: a proof of concept, randomized trial in patients with schizophrenia. J Psychiatr Pract 20:12–24. https://doi.org/10.1097/01.pra.0000442935.15833.c5
Prickaerts J, Van Goethem NP, Chesworth R et al (2012) EVP-6124, a novel and selective α7 nicotinic acetylcholine receptor partial agonist, improves memory performance by potentiating the acetylcholine response of α7 nicotinic acetylcholine receptors. Neuropharmacology. https://doi.org/10.1016/j.neuropharm.2011.10.024
Reichenberg A, Caspi A, Harrington H et al (2010) Static and dynamic cognitive deficits in childhood preceding adult schizophrenia: a 30-year study. Am J Psychiatry 167:160–169. https://doi.org/10.1176/appi.ajp.2009.09040574
Rosburg T, Trautner P, Dietl T et al (2005) Subdural recordings of the mismatch negativity (MMN) in patients with focal epilepsy. Brain 128:819–828. https://doi.org/10.1093/brain/awh442
Ross RG, Hunter SK, McCarthy L et al (2013) Perinatal choline effects on neonatal pathophysiology related to later schizophrenia risk. Am J Psychiatry 170:290–298. https://doi.org/10.1176/appi.ajp.2012.12070940
Sarkar AK, Ghosh D, Haldar D et al (2012) A rapid LC-ESI-MS/MS method for the quantitation of choline, an active metabolite of citicoline: application to in vivo pharmacokinetic and bioequivalence study in Indian healthy male volunteers. J Pharm Biomed Anal 71:144–147. https://doi.org/10.1016/j.jpba.2012.07.003
Sato Y, Yabe H, Todd J et al (2003) Impairment in activation of a frontal attention-switch mechanism in schizophrenic patients. Biol Psychol 62:49–63. https://doi.org/10.1016/S0301-0511(02)00113-8
Secades JJ (2011) Citicoline: pharmacological and clinical review, 2010 update. Rev Neurol 52
Shtyrov Y, Kujala T, Ahveninen J et al (1998) Background acoustic noise and the hemispheric lateralization of speech processing in the human brain: magnetic mismatch negativity study. Neurosci Lett 251:141–144. https://doi.org/10.1016/S0304-3940(98)00529-1
Smith DM, Fisher D, Blier P et al (2015) The separate and combined effects of monoamine oxidase A inhibition and nicotine on the mismatch negativity event related potential. Pharmacol Biochem Behav 137:44–52. https://doi.org/10.1016/j.pbb.2015.07.008
Stevens KE, Choo KS, Stitzel JA et al (2014) Long-term improvements in sensory inhibition with gestational choline supplementation linked to α7 nicotinic receptors through studies in Chrna7 null mutation mice. Brain Res. https://doi.org/10.1016/j.brainres.2014.01.022
Stevens KE, Zheng L, Floyd KL, Stitzel JA (2015) Maximizing the effect of an α7 nicotinic receptor PAM in a mouse model of schizophrenia-like sensory inhibition deficits. Brain Res 1611:8–17. https://doi.org/10.1016/j.brainres.2015.02.044
Swerdlow NR, Bhakta S, Chou HH et al (2016) Memantine effects on sensorimotor gating and mismatch negativity in patients with chronic psychosis. Neuropsychopharmacology 41:419–430. https://doi.org/10.1038/npp.2015.162
Tada M, Kirihara K, Mizutani S et al (2019) Mismatch negativity (MMN) as a tool for translational investigations into early psychosis: a review. Int J Psychophysiol:1–0
Tandon R, Nasrallah HA, Keshavan MS (2009) Schizophrenia, “just the facts” 4. Clinical features and conceptualization. Schizophr Res 110:1–3):1–23
Tikhonravov D, Neuvonen T, Pertovaara A et al (2008) Effects of an NMDA-receptor antagonist MK-801 on an MMN-like response recorded in anaesthetized rats. Brain Res 1203:97–102
Todd J, Harms L, Schal l U, Michie PT (2013) Mismatch negativity: translating the potential. Front Psychiatry 4:171
Uteshev VV (2014) The therapeutic promise of positive allosteric modulation of nicotinic receptors. Eur J Pharmacol 727:181–185. https://doi.org/10.1016/j.ejphar.2014.01.072 The
Wallace TL, Bertrand D (2015) Neuronal alpha7 nicotinic receptors as a target for the treatment of schizophrenia, 1st edn. Elsevier
Werkheiser JL, Sydserff S, Hubbs SJ et al (2011) Ultra-low exposure to alpha-7 nicotinic acetylcholine receptor partial agonists elicits an improvement in cognition that corresponds with an increase in alpha-7 receptor expression in rodents: implications for low dose clinical efficacy. Neuroscience. https://doi.org/10.1016/j.neuroscience.2011.04.033
Williams JBW, Gibbon M, First MB et al (1992) The structured clinical interview for DSM-III-R (SCID). Arch Gen Psychiatry 49:630–636. https://doi.org/10.1001/archpsyc.1992.01820080038006
Wing VC, Wass CE, Soh DW, George TP (2012) A review of neurobiological vulnerability factors and treatment implications for comorbid tobacco dependence in schizophrenia. Ann N Y Acad Sci 1248:89–106. https://doi.org/10.1111/j.1749-6632.2011.06261.x
Winterer G, Gallinat J, Brinkmeyer J et al (2013) Allosteric alpha-7 nicotinic receptor modulation and P50 sensory gating in schizophrenia: a proof-of-mechanism study. Neuropharmacology 64:197–204. https://doi.org/10.1016/j.neuropharm.2012.06.040
Wurtman RJ, Regan M, Ulus I, Yu L (2000) Effect of oral CDP-choline on plasma choline and uridine levels in humans. Biochem Pharmacol 60:989–992. https://doi.org/10.1016/S0006-2952(00)00436-6
Yamasue H, Yamada H, Yumoto M et al (2004) Abnormal association between reduced magnetic mismatch field to speech sounds and smaller left planum temporale volume in schizophrenia. Neuroimage 22:720–727. https://doi.org/10.1016/j.neuroimage.2004.01.042
Yang T, Xiao T, Sun Q, Wang K (2017) The current agonists and positive allosteric modulators of α7 nAChR for CNS indications in clinical trials. Acta Pharm Sin B 7:611–622. https://doi.org/10.1016/j.apsb.2017.09.001
Zhang ZW, Vijayaraghavan S, Berg DK (1994) Neuronal acetylcholine receptors that bind alpha-bungarotoxin with high affinity function as ligand-gated ion channels. Neuron 12:167–177
Funding
This work was supported in part by a grant to VK from the University of Ottawa Medical Research Fund (UMRF) and a Frederick Banting and Charles Best Canada Graduate Scholarship from the Canadian Institute of Health Research (CIHR) to JC.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Recruitment of healthy volunteers and study procedures were approved and complied with the guidelines of the Research Ethics Boards of The Royal Ottawa Mental Health Care Group and the University of Ottawa.
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Choueiry, J., Blais, C.M., Shah, D. et al. CDP-choline and galantamine, a personalized α7 nicotinic acetylcholine receptor targeted treatment for the modulation of speech MMN indexed deviance detection in healthy volunteers: a pilot study. Psychopharmacology 237, 3665–3687 (2020). https://doi.org/10.1007/s00213-020-05646-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00213-020-05646-1