Abstract
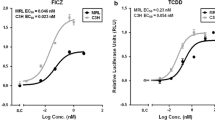
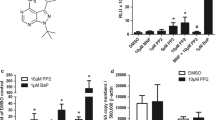
The dioxin receptor, also known as arylhydrocarbon receptor (AhR), is a ligand-activated transcription factor that mediates the toxicity of dioxins and related environmental contaminants. In addition, there is a growing list of natural compounds, mainly plant polyphenols that can modulate AhR function and downstream signaling with quite unknown consequences for cellular function. We investigate the potential of four different β-carboline alkaloids to stimulate AhR signaling in human hepatoma cells and keratinocytes. Three test substances, namely rutaecarpine, annomontine and xestomanzamine A, increase AhR-driven reporter gene activity as well as expression of two AhR target genes in a dose-dependent and time-dependent manner. Additionally, the three test alkaloids stimulate cytochrome P450 (CYP) 1 enzyme activity without showing any antagonistic effects regarding benzo(a)pyrene-stimulated CYP1 activation. The AhR-activating property of the β-carbolines is completely abrogated in AhR-deficient cells providing evidence that rutaecarpine, annomontine and xestomanzamine A are natural stimulators of the human AhR. The toxicological relevance of beta-carboline-mediated AhR activation is discussed.





Similar content being viewed by others
Abbreviations
- AhR:
-
Arylhydrocarbon receptor
- AhRR:
-
Arylhydrocarbon receptor repressor
- ARNT:
-
Arylhydrocarbon receptor nuclear translocator
- BaP:
-
Benzo(a)pyrene
- bHLH-PAS:
-
Basic Helix-Loop-Helix Per/ARNT/Sim
- CYP:
-
Cytochrome P450
- EGCG:
-
Epigallocatechine-3-gallate
- EROD:
-
Ethoxyresorufin-O-deethylase
- FICZ:
-
6-Formylindolo[3,2-b]carbazole
- HSP90:
-
Heat-shock protein 90
- MNF:
-
3′-Methoxy-4′-nitroflavone
- 3MC:
-
3-Methylcholanthrene
- PKC:
-
Protein kinase C
- TCDD:
-
2,3,7,8-Tetrachlorodibenzo-p-dioxin
- UV:
-
Ultraviolet
- XRE:
-
Xenobiotic responsive element
References
Agostinis P, Garmyn M, Van Laethem A (2007) The Aryl hydrocarbon receptor: an illuminating effector of the UVB response. Sci STKE 2007:e49
Amakura Y, Tsutsumi T, Sasaki K, Nakamura M, Yoshida T, Maitani T (2008) Influence of food polyphenols on aryl hydrocarbon receptor-signaling pathway estimated by in vitro bioassay. Phytochemistry 69:3117–3130
Asres K, Seyoum A, Veeresham C, Bucar F, Gibbons S (2005) Naturally derived anti-HIV agents. Phytother Res 19:557–581
Bock KW, Köhle C (2006) Ah receptor: dioxin-mediated toxic responses as hints to deregulated physiologic functions. Biochem Pharmacol 72:393–404
Breyer-Pfaff U, Wiatr G, Stevens I, Gaertner HJ, Mundle G, Mann K (1996) Elevated norharman plasma levels in alcoholic patients and controls resulting from tobacco smoking. Life Sci 58:1425–1432
Cao R, Peng W, Wang Z, Xu A (2007) Beta-carboline alkaloids: biochemical and pharmacological functions. Curr Med Chem 14:479–500
Chao HR, Wang YF, Chen HT, Ko YC, Chang EE, Huang YJ, Tsai FY, Tsai CH, Wu CH, Tsou TC (2007) Differential effect of arecoline on the endogenous dioxin-responsive cytochrome P450 1A1 and on a stably transfected dioxin-responsive element-driven reporter in human hepatoma cells. J Hazard Mater 149:234–237
Costa EV, Pinheiro ML, Xavier CM, Silva JR, Amaral AC, Souza AD, Barison A, Campos FR, Ferreira AG, Machado GM, Leon LL (2006) A pyrimidine-beta-carboline and other alkaloids from Annona foetida with antileishmanial activity. J Nat Prod 69:292–294
de Meester C (1995) Genotoxic potential of beta-carbolines: a review. Mutat Res 339:139–153
Delescluse C, Ledirac N, de Sousa G, Pralavorio M, Botta-Fridlund D, Letreut Y, Rahmani R (1997) Comparative study of CYPlAl induction by 3-methylcholanthrene in various human hepatic and epidermal cell types. Toxicol In Vitro 11:443–450
Dorow R, Horowski R, Paschelke G, Amin M (1983) Severe anxiety induced by FG 7142, a beta-carboline ligand for benzodiazepine receptors. Lancet 2:98–99
Fekkes D, Tuiten A, Bom I, Pepplinkhuizen L (2001) Pharmacokinetics of the beta-carboline norharman in man. Life Sci 69:2113–2121
Fernandez-Salguero PM, Hilbert DM, Rudikoff S, Ward JM, Gonzalez FJ (1996) Aryl-hydrocarbon receptor-deficient mice are resistant to 2, 3, 7, 8-tetrachlorodibenzo-p-dioxin-induced toxicity. Toxicol Appl Pharmacol 140:173–179
Fritsche E, Cline JE, Nguyen NH, Scanlan TS, Abel J (2005) Polychlorinated biphenyls disturb differentiation of normal human neural progenitor cells: clue for involvement of thyroid hormone receptors. Environ Health Perspect 113:871–876
Fritsche E, Schafer C, Calles C, Bernsmann T, Bernshausen T, Wurm M, Hubenthal U, Cline JE, Hajimiragha H, Schroeder P, Klotz LO, Rannug A, Furst P, Hanenberg H, Abel J, Krutmann J (2007) Lightening up the UV response by identification of the arylhydrocarbon receptor as a cytoplasmatic target for ultraviolet B radiation. Proc Natl Acad Sci USA 104:8851–8856
Gradin K, Wilhelmsson A, Poellinger L, Berghard A (1993) Nonresponsiveness of normal human fibroblasts to dioxin correlates with the presence of a constitutive xenobiotic response element-binding factor. J Biol Chem 268:4061–4068
Haarmann-Stemmann T, Abel J (2006) The arylhydrocarbon receptor repressor (AhRR): structure, expression, and function. Biol Chem 387:1195–1199
Haarmann-Stemmann T, Bothe H, Kohli A, Sydlik U, Abel J, Fritsche E (2007) Analysis of the transcriptional regulation and molecular function of the aryl hydrocarbon receptor repressor in human cell lines. Drug Metab Dispos 35:2262–2269
Haarmann-Stemmann T, Bothe H, Abel J (2009) Growth factors, cytokines and their receptors as downstream targets of arylhydrocarbon receptor (AhR) signaling pathways. Biochem Pharmacol 77:508–520
Hahn ME (2002) Aryl hydrocarbon receptors: diversity and evolution. Chem Biol Interact 141:131–160
Herraiz T (2000) Analysis of the bioactive alkaloids tetrahydro-beta-carboline and beta-carboline in food. J Chromatogr A 881:483–499
Herraiz T, Guillen H, Aran VJ (2008) Oxidative metabolism of the bioactive and naturally occurring beta-carboline alkaloids, norharman and harman, by human cytochrome P450 enzymes. Chem Res Toxicol 21:2172–2180
Ioannides C, Lewis DF (2004) Cytochromes P450 in the bioactivation of chemicals. Curr Top Med Chem 4:1767–1788
Iwanari M, Nakajima M, Kizu R, Hayakawa K, Yokoi T (2002) Induction of CYP1A1, CYP1A2, and CYP1B1 mRNAs by nitropolycyclic aromatic hydrocarbons in various human tissue-derived cells: chemical-, cytochrome P450 isoform-, and cell-specific differences. Arch Toxicol 76:287–298
Lee SK, Kim NH, Lee J, Kim DH, Lee ES, Choi HG, Chang HW, Jahng Y, Jeong TC (2004) Induction of cytochrome P450 s by rutaecarpine and metabolism of rutaecarpine by cytochrome P450 s. Planta Med 70:753–757
Lee SK, Bista SR, Jeong H, Kim DH, Kang MJ, Jahng Y, Jeong TC (2007) The effects of rutaecarpine on the pharmacokinetics of acetaminophen in rats. Arch Pharm Res 30:1629–1634
Lu YF, Santostefano M, Cunningham BD, Threadgill MD, Safe S (1995) Identification of 3′-methoxy-4′-nitroflavone as a pure aryl hydrocarbon (Ah) receptor antagonist and evidence for more than one form of the nuclear Ah receptor in MCF-7 human breast cancer cells. Arch Biochem Biophys 316:470–477
Machemer DE, Tukey RH (2005) The role of protein kinase C in regulation of TCDD-mediated CYP1A1 gene expression. Toxicol Sci 87:27–37
Medjakovic S, Jungbauer A (2008) Red clover isoflavones biochanin A and formononetin are potent ligands of the human aryl hydrocarbon receptor. J Steroid Biochem Mol Biol 108:171–177
Meggio F, Donella Deana A, Ruzzene M, Brunati A, Cesaro L, Guerra B, Meyer T, Mett H, Fabbro D, Furet P, Dobrowolska G, Pinna L (1995) Different susceptibility of protein kinases to staurosporine inhibition. Kinetic studies and molecular bases for the resistance of protein kinase CK2. Eur J Biochem 234:317–322
Mimura J, Ema M, Sogawa K, Fujii-Kuriyama Y (1999) Identification of a novel mechanism of regulation of Ah (dioxin) receptor function. Genes Dev 13:20–25
Moon YJ, Wang X, Morris ME (2006) Dietary flavonoids: effects on xenobiotic and carcinogen metabolism. Toxicol In Vitro 20:187–210
Murk AJ, Legler J, Denison MS, Giesy JP, van de Guchte C, Brouwer A (1996) Chemical-activated luciferase gene expression (CALUX): a novel in vitro bioassay for Ah receptor active compounds in sediments and pore water. Fundam Appl Toxicol 33:149–160
Nebert DW, Dalton TP, Okey AB, Gonzalez FJ (2004) Role of aryl hydrocarbon receptor-mediated induction of the CYP1 enzymes in environmental toxicity and cancer. J Biol Chem 279:23847–23850
Nishihashi H, Kanno Y, Tomuro K, Nakahama T, Inouye Y (2006) Primary structure and organ-specific expression of the rat aryl hydrocarbon receptor repressor gene. Biol Pharm Bull 29:640–647
Palermo CM, Westlake CA, Gasiewicz TA (2005) Epigallocatechin gallate inhibits aryl hydrocarbon receptor gene transcription through an indirect mechanism involving binding to a 90 kDa heat shock protein. Biochemistry 44:5041–5052
Park S, Henry EC, Gasiewicz TA (2000) Regulation of DNA binding activity of the ligand-activated aryl hydrocarbon receptor by tyrosine phosphorylation. Arch Biochem Biophys 381:302–312
Pfau W, Skog K (2004) Exposure to beta-carbolines norharman and harman. J Chromatogr B Analyt Technol Biomed Life Sci 802:115–126
Ramadoss P, Marcus C, Perdew GH (2005) Role of the aryl hydrocarbon receptor in drug metabolism. Expert Opin Drug Metab Toxicol 1:9–21
Rovescalli AC, Brunello N, Franzetti C, Racagni G (1986) Interaction of putative endogenous tryptolines with the hypothalamic serotonergic system and prolactin secretion in adult male rats. Neuroendocrinology 43:603–610
Schmidt JV, Su GH, Reddy JK, Simon MC, Bradfield CA (1996) Characterization of a murine Ahr null allele: involvement of the Ah receptor in hepatic growth and development. Proc Natl Acad Sci U S A 93:6731–6736
Seidel SD, Li V, Winter GM, Rogers WJ, Martinez EI, Denison MS (2000) Ah receptor-based chemical screening bioassays: application and limitations for the detection of Ah receptor agonists. Toxicol Sci 55:107–115
Sheu J (1999) Pharmacological effects of rutaecarpine, an alkaloid isolated from Evodia rutaecarpa. Cardiovasc Drug Rev 17:237–245
Totsuka Y, Takamura-Enya T, Nishigaki R, Sugimura T, Wakabayashi K (2004) Mutagens formed from beta-carbolines with aromatic amines. J Chromatogr B Analyt Technol Biomed Life Sci 802:135–141
Tsuchiya Y, Nakajima M, Itoh S, Iwanari M, Yokoi T (2003) Expression of aryl hydrocarbon receptor repressor in normal human tissues and inducibility by polycyclic aromatic hydrocarbons in human tumor-derived cell lines. Toxicol Sci 72:253–259
Ueng YF, Wang JJ, Lin LC, Park SS, Chen CF (2001) Induction of cytochrome P450-dependent monooxygenase in mouse liver and kidney by rutaecarpine, an alkaloid of the herbal drug Evodia rutaecarpa. Life Sci 70:207–217
Ueng YF, Jan WC, Lin LC, Chen TL, Guengerich FP, Chen CF (2002) The alkaloid rutaecarpine is a selective inhibitor of cytochrome P450 1A in mouse and human liver microsomes. Drug Metab Dispos 30:349–353
Ueng YF, Tsai TH, Don MJ, Chen RM, Chen TL (2005) Alteration of the pharmacokinetics of theophylline by rutaecarpine, an alkaloid of the medicinal herb Evodia rutaecarpa, in rats. J Pharm Pharmacol 57:227–232
Vanden Heuvel JP, Clark GC, Thompson CL, McCoy Z, Miller CR, Lucier GW, Bell DA (1993) CYP1A1 mRNA levels as a human exposure biomarker: use of quantitative polymerase chain reaction to measure CYP1A1 expression in human peripheral blood lymphocytes. Carcinogenesis 14:2003–2006
Venault P, Chapouthier G (2007) From the behavioral pharmacology of beta-carbolines to seizures, anxiety, and memory. ScientificWorldJournal 7:204–223
Vogel CF, Matsumura F (2009) A new cross-talk between the aryl hydrocarbon receptor and RelB, a member of the NF-kappaB family. Biochem Pharmacol 77:734–745
Yu AM, Idle JR, Krausz KW, Kupfer A, Gonzalez FJ (2003) Contribution of individual cytochrome P450 isozymes to the O-demethylation of the psychotropic beta-carboline alkaloids harmaline and harmine. J Pharmacol Exp Ther 305:315–322
Zeng X, Zeng X (1999) Relationship between the clinical effects of berberine on severe congestive heart failure and its concentration in plasma studied by HPLC. Biomed Chromatogr 13:442–444
Zhang S, Qin C, Safe SH (2003) Flavonoids as aryl hydrocarbon receptor agonists/antagonists: effects of structure and cell context. Environ Health Perspect 111:1877–1882
Acknowledgments
We are grateful to Katarina Gradin and Lorenz Poellinger (Karolinska Institutet, Sweden) for generously providing the XRE-HepG2 reporter cells and to Yoshio Inouye (Toho University, Japan) for the kind donation of the pcDNA5-rAhRR plasmid. We thank Birgit Neumann and Ulrike Hübenthal for technical support. This work was supported by the graduate school 1427 of the Deutsche Forschungsgemeinschaft (DFG, Bonn, Germany).
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Haarmann-Stemmann, T., Sendker, J., Götz, C. et al. Regulation of dioxin receptor function by different beta-carboline alkaloids. Arch Toxicol 84, 619–629 (2010). https://doi.org/10.1007/s00204-010-0548-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00204-010-0548-2