Abstract
Summary
This study evaluated the peri-implant bone repair in orchiectomized rats receiving intermittently PTH 1-34. The treatment returned the bone quality and quantity of the animals to normal in the computerized microtomography, laser confocal microscopy, and histological analysis. The PTH 1-34 promoted marked bone formation with increased volume, improved quality, and greater bone turnover.
Introduction
Osteoporosis can be a problem in implant osseointegration. So this study aimed to evaluate the quantity and quality of peri-implant bone repair in orchiectomized Wistar rats receiving intermittently administered PTH 1-34.
Methods
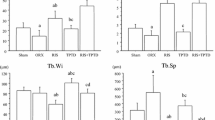
Animals (n = 24) were divided into 3 groups: healthy control (SHAM), orchiectomized (ORQ), and orchiectomized and treated with 0.5 μg/kg/day PTH 1-34 (TERI), and each received an implant in the right and left tibial metaphysis, which was allowed to repair for 60 days. The resultant bone formation was evaluated through computerized microtomography (micro-CT) to compare the percent bone volume (BV/TV), trabecular thickness (Tb.Th), trabecular number and separation (Tb.N, Tb.Sp), and bone implant contact (BIC) through the intersection surface (i.S) between groups. Laser confocal microscopy was used to evaluate fluorochrome areas for mineral apposition rate (MAR) and neoformed bone area (NBA). In addition, histological evaluation of calcified tissues with Stevenel blue and alizarin red staining was performed.
Results
Treatment with PTH 1-34 returned the bone quality and quantity of the osteoporotic animal to normal, as the TERI group presented statistically significant higher values for BV/TV, Tb.Th, and BIC parameters compared with ORQ (p < 0.05), but when compared with SHAM (p > 0.05), no statistical difference was noted. In addition, in the bone turnover analysis (MAR, NBA) for TERI, the highest results are presented, followed by SHAM, and then ORQ (TERI × ORQ: p < 0.05).
Conclusions
Intermittent treatment with PTH 1-34 on orchiectomized animals promoted marked bone formation with increased volume, improved quality, and greater bone turnover in the peri-implant space, returning the bone quality and quantity to the present standard in healthy animals.





Similar content being viewed by others
References
Foundation IO (2009) Facts and statistics about osteoporosis and its implications. www.iofbonehealth.org/facts-and-statistics.html
Drage NA, Palmer RM, Blake G, Wilson R, Crane F, Fogelman I (2007) A comparison of bone mineral density in the spine, hip and jaws of edentulous subjects. Clin Oral Implants Res 18(4):496–500. https://doi.org/10.1111/j.1600-0501.2007.01379.x
Lirani-Galvao AP, Lazaretti-Castro M (2010) Physical approach for prevention and treatment of osteoporosis. Arq Bras Endocrinol Metabol 54(2):171–178. https://doi.org/10.1590/s0004-27302010000200013
Shapurian T, Damoulis PD, Reiser GM, Griffin TJ, Rand WM (2006) Quantitative evaluation of bone density using the Hounsfield index. Int J Oral Maxillofac Implants 21(2):290–297
Hagino H (2016) Diagnosis of male osteoporosis. Clin Calcium 26(7):995–1001
Kaufman JM, Lapauw B, Goemaere S (2014) Current and future treatments of osteoporosis in men. Best Pract Res Clin Endocrinol Metab 28(6):871–884. https://doi.org/10.1016/j.beem.2014.09.002
Niimi R, Kono T, Nishihara A, Hasegawa M, Matsumine A, Kono T, Sudo A (2015) Analysis of daily teriparatide treatment for osteoporosis in men. Osteoporos Int 26(4):1303–1309. https://doi.org/10.1007/s00198-014-3001-1
Farahmand P, Spiegel R, Ringe JD (2016) Male osteoporosis. Z Rheumatol 75(5):459–465. https://doi.org/10.1007/s00393-016-0078-2
Kendler DL, Marin F, Zerbini CAF, Russo LA, Greenspan SL, Zikan V, Bagur A, Malouf-Sierra J, Lakatos P, Fahrleitner-Pammer A, Lespessailles E, Minisola S, Body JJ, Geusens P, Moricke R, Lopez-Romero P (2018) Effects of teriparatide and risedronate on new fractures in post-menopausal women with severe osteoporosis (VERO): a multicentre, double-blind, double-dummy, randomised controlled trial. Lancet (London, England) 391(10117):230–240. https://doi.org/10.1016/s0140-6736(17)32137-2
de Oliveira D, de Oliveira PI, Silva Gomes-Ferreira PH, Palin LP, Matsumoto MA, Okamoto R (2019) Effect of intermittent teriparatide (PTH 1-34) on the alveolar healing process in orchiectomized rats. Clin Oral Investig 23(5):2313–2322. https://doi.org/10.1007/s00784-018-2672-y
de Oliveira PI, Gomes-Ferreira P, de Oliveira D, Hassumi JS, Goncalves PZ, Okamoto R (2019) Teriparatide improves alveolar bone modelling after tooth extraction in orchiectomized rats. Arch Oral Biol 102:147–154. https://doi.org/10.1016/j.archoralbio.2019.04.007
Hadaya D, Gkouveris I, Soundia A, Bezouglaia O, Boyce RW, Stolina M, Dwyer D, Dry SM, Pirih FQ, Aghaloo TL, Tetradis S (2019) Clinically relevant doses of sclerostin antibody do not induce osteonecrosis of the jaw (ONJ) in rats with experimental periodontitis. J Bone Miner Res 34(1):171–181. https://doi.org/10.1002/jbmr.3581
Hayashida S, Soutome S, Yanamoto S, Fujita S, Hasegawa T, Komori T, Kojima Y, Miyamoto H, Shibuya Y, Ueda N, Kirita T, Nakahara H, Shinohara M, Umeda M (2017) Evaluation of the treatment strategies for medication-related osteonecrosis of the jaws (MRONJ) and the factors affecting treatment outcome: a multicenter retrospective study with propensity score matching analysis. J Bone Miner Res 32(10):2022–2029. https://doi.org/10.1002/jbmr.3191
Faverani LP, Polo TOB, Ramalho-Ferreira G, Momesso GAC, Hassumi JS, Rossi AC, Freire AR, Prado FB, Luvizuto ER, Gruber R, Okamoto R (2018) Raloxifene but not alendronate can compensate the impaired osseointegration in osteoporotic rats. Clin Oral Investig 22(1):255–265. https://doi.org/10.1007/s00784-017-2106-2
Kilkenny C, Browne W, Cuthill IC, Emerson M, Altman DG (2011) Animal research: reporting in vivo experiments--the ARRIVE guidelines. J Cereb Blood Flow Metab 31(4):991–993. https://doi.org/10.1038/jcbfm.2010.220
Seifi M, Ezzati B, Saedi S, Hedayati M (2015) The effect of ovariectomy and orchiectomy on orthodontic tooth movement and root resorption in Wistar rats. J Dent (Shiraz, Iran) 16(4):302–309
Bouxsein ML, Boyd SK, Christiansen BA, Guldberg RE, Jepsen KJ, Muller R (2010) Guidelines for assessment of bone microstructure in rodents using micro-computed tomography. J Bone Miner Res 25(7):1468–1486. https://doi.org/10.1002/jbmr.141
Dempster DW, Compston JE, Drezner MK, Glorieux FH, Kanis JA, Malluche H, Meunier PJ, Ott SM, Recker RR, Parfitt AM (2013) Standardized nomenclature, symbols, and units for bone histomorphometry: a 2012 update of the report of the ASBMR Histomorphometry Nomenclature Committee. J Bone Miner Res 28(1):2–17. https://doi.org/10.1002/jbmr.1805
Cosman F (2014) Anabolic and antiresorptive therapy for osteoporosis: combination and sequential approaches. Curr Osteoporos Rep 12(4):385–395. https://doi.org/10.1007/s11914-014-0237-9
Drake MT, Khosla S (2012) Male osteoporosis. Endocrinol Metab Clin N Am 41(3):629–641. https://doi.org/10.1016/j.ecl.2012.05.001
Lindsay R, Krege JH, Marin F, Jin L, Stepan JJ (2016) Teriparatide for osteoporosis: importance of the full course. Osteoporos Int 27(8):2395–2410. https://doi.org/10.1007/s00198-016-3534-6
Ramalho-Ferreira G, Faverani LP, Grossi-Oliveira GA, Okamoto T, Okamoto R (2015) Alveolar bone dynamics in osteoporotic rats treated with raloxifene or alendronate: confocal microscopy analysis. J Biomed Opt 20(3):038003. https://doi.org/10.1117/1.jbo.20.3.038003
Oliveira D, Hassumi JS, Gomes-Ferreira PH, Polo TO, Ferreira GR, Faverani LP, Okamoto R (2017) Short term sodium alendronate administration improves the peri-implant bone quality in osteoporotic animals. J Appl Oral Sci 25(1):42–52. https://doi.org/10.1590/1678-77572016-0165
Ozawa S, Ogawa T, Iida K, Sukotjo C, Hasegawa H, Nishimura RD, Nishimura I (2002) Ovariectomy hinders the early stage of bone-implant integration: histomorphometric, biomechanical, and molecular analyses. Bone 30(1):137–143. https://doi.org/10.1016/s8756-3282(01)00646-9
Yamazaki M, Shirota T, Tokugawa Y, Motohashi M, Ohno K, Michi K, Yamaguchi A (1999) Bone reactions to titanium screw implants in ovariectomized animals. Oral Surg Oral Med Oral Pathol Oral Radiol Endod 87(4):411–418. https://doi.org/10.1016/s1079-2104(99)70239-8
Duckworth AD, McQueen MM, Tuck CE, Tobias JH, Wilkinson JM, Biant LC, Pulford EC, Aldridge S, Edwards C, Roberts CP, Ramachandran M, McAndrew AR, Cheng KC, Johnston P, Shah NH, Mathew P, Harvie J, Hanusch BC, Harkess R, Rodriguez A, Murray GD, Ralston SH (2019) Effect of alendronic acid on fracture healing: a multicenter randomized placebo-controlled trial. J Bone Miner Res 34(6):1025–1032. https://doi.org/10.1002/jbmr.3679
Abrahamsen B, Eiken P, Prieto-Alhambra D, Eastell R (2016) Risk of hip, subtrochanteric, and femoral shaft fractures among mid and long term users of alendronate: nationwide cohort and nested case-control study. BMJ (Clin Res ed) 353:i3365. https://doi.org/10.1136/bmj.i3365
Aydogan NH, Ozel I, Iltar S, Kara T, Ozmeric A, Alemdaroglu KB (2016) The effect of vitamin D and bisphosphonate on fracture healing: an experimental study. J Clin Orthop Trauma 7(2):90–94. https://doi.org/10.1016/j.jcot.2016.01.003
Chacon GE, Stine EA, Larsen PE, Beck FM, McGlumphy EA (2006) Effect of alendronate on endosseous implant integration: an in vivo study in rabbits. J Oral Maxillofac Surg 64(7):1005–1009. https://doi.org/10.1016/j.joms.2006.01.007
Ducy P, Schinke T, Karsenty G (2000) The osteoblast: a sophisticated fibroblast under central surveillance. Science (New York, NY) 289(5484):1501–1504. https://doi.org/10.1126/science.289.5484.1501
Fan YP, Chen XY, Chen Y, Yang GL, Wang HM, He FM (2017) Positive effect of strontium-oxide layer on the osseointegration of moderately rough titanium surface in non-osteoporotic rabbits. Clin Oral Implants Res 28(8):911–919. https://doi.org/10.1111/clr.12897
Lekhokm UZG (1985) Patient selection and preparation. In: Tissue integrated prostheses: osseointegration in clinical dentistry. Quintessene Publ Co, Chicago, pp 199–209
Miller PD, Derman RJ (2010) What is the best balance of benefits and risks among anti-resorptive therapies for postmenopausal osteoporosis? Osteoporos Int 21(11):1793–1802. https://doi.org/10.1007/s00198-010-1208-3
Oh KC, Moon HS, Lee JH, Park YB, Kim JH (2015) Effects of alendronate on the peri-implant bone in rats. Oral Dis 21(2):248–256. https://doi.org/10.1111/odi.12258
Verzola MH, Frizzera F, de Oliveira GJ, Pereira RM, Rodrigues-Filho UP, Nonaka KO, Orrico SR (2015) Effects of the long-term administration of alendronate on the mechanical properties of the basal bone and on osseointegration. Clin Oral Implants Res 26(12):1466–1475. https://doi.org/10.1111/clr.12492
Bliuc D, Tran T, van Geel T, Adachi JD, Berger C, van den Bergh J, Eisman JA, Geusens P, Goltzman D, Hanley DA, Josse R, Kaiser S, Kovacs CS, Langsetmo L, Prior JC, Nguyen TV, Center JR (2019) Reduced bone loss is associated with reduced mortality risk in subjects exposed to nitrogen bisphosphonates: a mediation analysis. J Bone Miner Res 34(11):2001–2011. https://doi.org/10.1002/jbmr.3816
Ruggiero SL, Dodson TB, Fantasia J, Goodday R, Aghaloo T, Mehrotra B, O'Ryan F (2014) American Association of Oral and Maxillofacial Surgeons position paper on medication-related osteonecrosis of the jaw--2014 update. J Oral Maxillofac Surg 72(10):1938–1956. https://doi.org/10.1016/j.joms.2014.04.031
von Wowern N, Kollerup G (1992) Symptomatic osteoporosis: a risk factor for residual ridge reduction of the jaws. J Prosthet Dent 67(5):656–660. https://doi.org/10.1016/0022-3913(92)90165-7
Li H, Zhou Q, Bai BL, Weng SJ, Wu ZY, Xie ZJ, Feng ZH, Cheng L, Boodhun V, Yang L (2018) Effects of combined human parathyroid hormone (1-34) and menaquinone-4 treatment on the interface of hydroxyapatite-coated titanium implants in the femur of osteoporotic rats. J Bone Miner Metab 36(6):691–699. https://doi.org/10.1007/s00774-017-0893-9
Delos D, Yang X, Ricciardi BF, Myers ER, Bostrom MP, Camacho NP (2008) The effects of RANKL inhibition on fracture healing and bone strength in a mouse model of osteogenesis imperfecta. J Orthop Res 26(2):153–164. https://doi.org/10.1002/jor.20469
Gardner MJ, Ricciardi BF, Wright TM, Bostrom MP, van der Meulen MC (2008) Pause insertions during cyclic in vivo loading affect bone healing. Clin Orthop Relat Res 466(5):1232–1238. https://doi.org/10.1007/s11999-008-0155-1
Gardner MJ, van der Meulen MC, Carson J, Zelken J, Ricciardi BF, Wright TM, Lane JM, Bostrom MP (2007) Role of parathyroid hormone in the mechanosensitivity of fracture healing. J Orthop Res 25(11):1474–1480. https://doi.org/10.1002/jor.20427
Ramalho-Ferreira G, Faverani LP, Prado FB, Garcia IR Jr, Okamoto R (2015) Raloxifene enhances peri-implant bone healing in osteoporotic rats. Int J Oral Maxillofac Surg 44(6):798–805. https://doi.org/10.1016/j.ijom.2015.02.018
Acknowledgments
The company Neodent is acknowledged for supplying the implants to make this work possible.
Funding
The authors would like to thank the São Paulo Research Foundation (FAPESP) for funding this work (process number: 2015/04649-7 and 2015/14688-0), CAPES (UNESP/CAPES/Print Project), the Department of Diagnosis and Surgery, as well as Prof. Dr. Elcio Marcantonio Júnior and laboratory technician Ana Cláudia Gregolin Costa Miranda, for facilitating the processing of the bone in the EXAKT system.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
None.
Ethical approval
This work was approved by the Institutional Animal Ethics Committee of the Araçatuba Dental School (CEUA) with the registration number (CEUA: 2015-00238) and followed the ARRIVE guidelines for its preparation.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Gomes-Ferreira, P.H.S., de Oliveira, D., Frigério, P.B. et al. Teriparatide improves microarchitectural characteristics of peri-implant bone in orchiectomized rats. Osteoporos Int 31, 1807–1815 (2020). https://doi.org/10.1007/s00198-020-05431-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00198-020-05431-y