Abstract
Summary
The Effectiveness of Discontinuing Bisphosphonates (EDGE) study is a planned pragmatic clinical trial to guide “drug holiday” clinical decision making. This pilot study assessed work flow and feasibility of such a study. While participant recruitment and treatment adherence were suboptimal, administrative procedures were generally feasible and minimally disrupted clinic flow.
Introduction
The comparative effectiveness of continuing or discontinuing long-term alendronate (ALN) on fractures is unknown. A large pragmatic ALN discontinuation study has potential to answer this question.
Methods
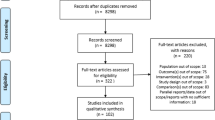
We conducted a 6-month pilot study of the planned the EDGE study among current long-term ALN users (women aged ≥65 with ≥3 years of ALN use) to determine study work flow and feasibility including evaluating the administrative aspects of trial conduct (e.g., time to contract, institutional review board (IRB) approval), assessing rates of site and participant recruitment, and evaluating post-randomization outcomes, including adherence, bisphosphonate-associated adverse events, and participant and site satisfaction. We assessed outcomes 1 and 6 months after randomization.
Results
Nine sites participated, including seven community-based medical practices and two academic medical centers. On average (SD), contract execution took 3.4 (2.3) months and IRB approval took 13.9 (4.1) days. Sites recruited 27 participants (13 to continue ALN and 14 to discontinue ALN). Over follow-up, 22% of participants did not adhere to their randomization assignment: 30.8% in the continuation arm and 14.3% in the discontinuation arm. No fractures or adverse events were reported. Sites reported no issues regarding work flow, and participants were highly satisfied with the study.
Conclusions
Administrative procedures of the EDGE study were generally feasible, with minimal disruption to clinic flow. In this convenience sample, participant recruitment was suboptimal across most practice sites. Accounting for low treatment arm adherence, a comprehensive recruitment approach will be needed to effectively achieve the scientific goals of the EDGE study.



Similar content being viewed by others
Change history
07 June 2017
An erratum to this article has been published.
References
Ensrud KE et al (2007) Frailty and risk of falls, fracture, and mortality in older women: the study of osteoporotic fractures. J Gerontol A Biol Sci Med Sci 62(7):744–751
Rubenstein LZ (2006) Falls in older people: epidemiology, risk factors and strategies for prevention. Age Ageing 35(Suppl 2):ii37–ii41
Compston JE, Bilezikian JP (2012) Bisphosphonate therapy for osteoporosis: the long and short of it. J Bone Miner Res 27(2):240–242
Diab DL, Watts NB (2012) Bisphosphonates in the treatment of osteoporosis. Endocrinol Metab Clin N Am 41(3):487–506
McClung M et al (2013) Bisphosphonate therapy for osteoporosis: benefits, risks, and drug holiday. Am J Med 126(1):13–20
Shane E et al (2014) Atypical subtrochanteric and diaphyseal femoral fractures: second report of a task force of the American Society for Bone and Mineral Research. J Bone Miner Res 29(1):1–23
Wysowski DK, Greene P (2013) Trends in osteoporosis treatment with oral and intravenous bisphosphonates in the United States, 2002-2012. Bone 57(2):423–428
Burch J et al (2014) Systematic review of the use of bone turnover markers for monitoring the response to osteoporosis treatment: the secondary prevention of fractures, and primary prevention of fractures in high-risk groups. Health Technol Assess 18(11):1–180
Vasikaran S et al (2011) Markers of bone turnover for the prediction of fracture risk and monitoring of osteoporosis treatment: a need for international reference standards. Osteoporos Int 22(2):391–420
Miller PD et al (2008) Effect of denosumab on bone density and turnover in postmenopausal women with low bone mass after long-term continued, discontinued, and restarting of therapy: a randomized blinded phase 2 clinical trial. Bone 43(2):222–229
Reid DM et al (2009) Zoledronic acid and risedronate in the prevention and treatment of glucocorticoid-induced osteoporosis (HORIZON): a multicentre, double-blind, double-dummy, randomised controlled trial. Lancet 373(9671):1253–1263
Rosen CJ et al (2005) Treatment with once-weekly alendronate 70 mg compared with once-weekly risedronate 35 mg in women with postmenopausal osteoporosis: a randomized double-blind study. J Bone Miner Res 20(1):141–151
Saag KG et al (2007) Teriparatide or alendronate in glucocorticoid-induced osteoporosis. N Engl J Med 357(20):2028–2039
Boonen S et al (2012) Postmenopausal osteoporosis treatment with antiresorptives: effects of discontinuation or long-term continuation on bone turnover and fracture risk—a perspective. J Bone Miner Res 27(5):963–974
Abrahamsen B, Eiken P, Eastell R (2009) Subtrochanteric and diaphyseal femur fractures in patients treated with alendronate: a register-based national cohort study. J Bone Miner Res 24(6):1095–1102
Brown, J.P., et al., Comparison of the effect of denosumab and alendronate on bone mineral density and biochemical markers of bone turnover in postmenopausal women with low bone mass: a randomized, blinded, phase 3 trial. J Bone Miner Res, 2009. Epub ahead of print: p. 1–34.
Girgis CM, Sher D, Seibel MJ (2010) Atypical femoral fractures and bisphosphonate use. N Engl J Med 362(19):1848–1849
Green J et al (2010) Oral bisphosphonates and risk of cancer of oesophagus, stomach, and colorectum: case-control analysis within a UK primary care cohort. BMJ 341:c4444
Kendler DL et al (2009) Effects of denosumab on bone mineral density and bone turnover in postmenopausal women transitioning from alendronate therapy. J Bone Miner Res 25(1):72–81
Lenart BA, Lorich DG, Lane JM (2008) Atypical fractures of the femoral diaphysis in postmenopausal women taking alendronate. N Engl J Med 358(12):1304–1306
Lenart BA et al (2009) Association of low-energy femoral fractures with prolonged bisphosphonate use: a case control study. Osteoporos Int 20(8):1353–1362
Mammo Z et al (2016) Oral bisphosphonates and risk of wet age-related macular degeneration. Am J Ophthalmol 168:62–67
Mavrokokki T et al (2007) Nature and frequency of bisphosphonate-associated osteonecrosis of the jaws in Australia. J Oral Maxillofac Surg 65(3):415–423
Odvina CV et al (2005) Severely suppressed bone turnover: a potential complication of alendronate therapy. J Clin Endocrinol Metab 90(3):1294–1301
Ott SM (2001) Fractures after long-term alendronate therapy. J Clin Endocrinol Metab 86(4):1835–1836
Rizzoli R et al (2008) Osteonecrosis of the jaw and bisphosphonate treatment for osteoporosis. Bone 42(5):841–847
Ruggiero SL et al (2004) Osteonecrosis of the jaws associated with the use of bisphosphonates: a review of 63 cases. J Oral Maxillofac Surg 62(5):527–534
Shane, E., et al., Atypical subtrochanteric and diaphyseal femoral fractures: report of a task force of the American Society for Bone and Mineral Research. J Bone Miner Res, 2010.
Wysowski DK (2009) Reports of esophageal cancer with oral bisphosphonate use. N Engl J Med 360(1):89–90
Black DM et al (2006) Effects of continuing or stopping alendronate after 5 years of treatment: the Fracture Intervention Trial Long-term Extension (FLEX): a randomized trial. JAMA 296(24):2927–2938
Khosla S, Shane E (2016) A crisis in the treatment of osteoporosis. J Bone Miner Res 31(8):1485–1487
Berry, D.A., Statistical innovations in cancer research., in Cancer Medicine, 6th Edition., E.F. J. Holland, D. W. Kufe, R. E. Pollock, R. R. Weichselbaum, R. C. Bast and T. S. Gansler, Editor. 2003, BC Decker: Hamilton. p. 465–478.
Godwin M et al (2003) Pragmatic controlled clinical trials in primary care: the struggle between external and internal validity. BMC Med Res Methodol 3:28
Warriner AH et al (2016) A pragmatic randomized trial comparing tablet computer informed consent to traditional paper-based methods for an osteoporosis study. Contemp Clin Trials Commun 3:32–38
Khosla S et al (2007) Bisphosphonate-associated osteonecrosis of the jaw: report of a task force of the American Society for Bone and Mineral Research. J Bone Miner Res 22(10):1479–1491
Kroenke K et al (2014) Operating characteristics of PROMIS four-item depression and anxiety scales in primary care patients with chronic pain. Pain Med 15(11):1892–1901
Pilkonis PA et al (2011) Item banks for measuring emotional distress from the Patient-Reported Outcomes Measurement Information System (PROMIS(R)): depression, anxiety, and anger. Assessment 18(3):263–283
Yun H et al (2013) Generic alendronate use among Medicare beneficiaries: are Part D data complete? Pharmacoepidemiol Drug Saf 22(1):55–63
Wright NC, Cadarette SM, Smith WK, Burden AM, Warriner AH, Foster PJ, Yun H, Melton ME, Curtis JR, Saag KG (2014) Long-term oral bisphosphonate use for osteoporosis among older women—US and Canadian perspective. Arthritis & Rheumatism 66(10):S20
Kolata, G., Osteoporosis drugs shunned for fear of rare side effects, in New York Times 2016, New York Times Company: New York. p. A1.
Black DM et al (1996) Randomised trial of effect of alendronate on risk of fracture in women with existing vertebral fractures. Fracture Intervention Trial Research Group. Lancet 348(9041):1535–1541
Wright NC, Foster PJ, Fullman S, Randall S, Melton ME, Pace W, Calmbach W, Saag KG (2015) Patient perspectives on participating in the Effectiveness of Discontinuing Bisphosphonates (EDGE) study. J Bone Miner Res 30(Suppl 1)
Jha S et al (2015) Trends in media reports, oral bisphosphonate prescriptions, and hip fractures 1996-2012: an ecological analysis. J Bone Miner Res 30(12):2179–2187
Lewiecki, E.M., et al., Hip fractures and declining DXA testing: at a breaking point? J Bone Miner Res 2016. 31(Suppl 1).
Acknowledgments
The authors would like to extend their most sincere appreciation to the physicians and staffs of the participating community practices for their diligent efforts and support of this project. The research reported in this publication was supported by the National Institute of Arthritis and Musculoskeletal and Skin Diseases under award number 1R21AR062300. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institute of Arthritis and Musculoskeletal and Skin Diseases.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
NCW—Research contracts: Amgen; Consultant: Pfizer.
PJF—None.
ASM—None.
JAM—None.
EML—Research contracts: Amgen, Merck, and Lilly; Scientific Advisory Boards: Amgen, Merck, Lilly, Radius Health, Shire, Alexion, and AbbVie; Speakers’ Bureau: Shire and Alexion.
WJS—None.
JRC—Research contracts: Amgen; Consultant: Amgen.
GRC—None.
MID—None.
MLK—Research contracts: Amgen.
CEL—None.
SLM—None.
DTR—None.
AHW—None.
KGS—Research contracts: Amgen.
Additional information
An erratum to this article is available at https://doi.org/10.1007/s00198-017-4105-1.
Rights and permissions
About this article
Cite this article
Wright, N.C., Foster, P.J., Mudano, A.S. et al. Assessing the feasibility of the Effectiveness of Discontinuing Bisphosphonates trial: a pilot study. Osteoporos Int 28, 2495–2503 (2017). https://doi.org/10.1007/s00198-017-4073-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00198-017-4073-5