Abstract
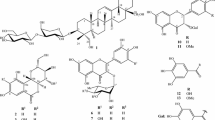
Phytochemical investigation of the CHCl3 extract of the seed embryos of Nelumbo nucifera Gaertn resulted in the isolation of a new naturally occurring bisbenzylisoquinoline alkaloid, O-methylneferine (1), together with five known alkaloids, neferine (2), armepavine (3), (–)-(1R)-N-methylcoclaurine (4), nuciferine (5), and pronuciferine (6). The structures of these compounds were characterized by spectroscopic methods and comparison of physical properties with those reported in the literature. Among them, compounds 1 and 2 exhibited significant activity against human colon adenocarcinoma cell line (HT-29), with IC50 values of 0.70 and 1.61 µM, respectively, which were 8- and 3.5-fold higher than that of the reference anticancer drug, doxorubicin (IC50 5.63 µM). Moreover, compounds 1 and 2 displayed less cytotoxic activity against the non-cancerous HEK 239 cells with the IC50 values of 42.48 and 12.19 μM, respectively, whereas the cytotoxicity of doxorubicin against this cell line was 0.22 μM. The very potent cytotoxicity against HT-29 cell line and very high selectivity index (60.6-fold) of the alkaloid 1 is of particular significant; it could be considered as a promising structure lead for anti-colon cancer drug development.

Similar content being viewed by others
References
Duan XH, Pei L, Jiang JQ (2013) Cytotoxic alkaloids from stems of Nelumbo nucifera. Zhongguo Zhong Yao Za Zhi (China J Chin Mat Med) 38:4104–4108
Fajardo V, Araya M, Cuada P, Oyarzum A, Gallardo A, Cueto M, Diaz-Marrero AR, Darias J, Villarroel L, Alvarez C, Mora-Perez Y, Joseph-Nathan P (2009) Pronuciferine N-Oxide, a proaporphine N-Oxide alkaloid from Berberis coletioides. J Nat Prod 72:1355–1356
Itoh A, Saitoh T, Tani K, Uchigaki M, Sugimoto Y, Yamada J, Nakajima H, Ohshiro H, Sun S, Tanahashi T (2001) Bisbenzylisoquinoline alkaloids from Nelumbo nucifera. Chem Pharm Bull 59:947–951
Jung HA, Jin SE, Choi RJ, Kim DH, Kim YS, Ryu JH, Kim DW, Son TK, Park JJ, Choi JS (2010) Anti-amnesis activity of neferine with antioxidant and anti-inflammatory capacities, as well as inhibition of ChEs and BACE1. Life Sci 87:420–430
Kashiwada Y, Aoshima A, Ikeshiro Y, Chen YP, Furukawa H, Itoigawa M, Fujioka T, Mihashi K, Cosentino LM, Morris-Natschke SL, Lee KH (2005) Anti-HIV benzylisoquinoline alkaloids and flavonoids from the leaves of Nelumbo nucifera, and structure-activity correlations with related alkaloids. Bioorg Med Chem 13:443–448
Leet JE, Fajardo V, Freyer AJ, Shamma M (1983) Some dimeric benzylisoquinoline alkaloids with an unusual oxygenation pattern. J Nat Prod 46:908–912
Mehta NR, Patani EPPV, Shah B (2013) Nelumbo nucifera (lotus): A review on ethnobotany, phytochemistry and pharmacology. Indian J PharmBiol Res 1:152–167
Nishimura K, Horii S, Tanahashi T, Sugimoto Y, Yamada J (2013) Synthesis and pharmacological activity of alkaloids from embryo of lotus, Nelumbo nucifera. Chem Pharm Bull 61:59–68
Oian JQ (2002) Cardiovascular pharmacological effects of bisbenzylisoquinoline alkaloid derivatives. Acta Pharm Sin 23:1086–1092
Rai S, Wahile A, Mukherjee K, Saha BP, Mukherjee PK (2006) Antioxidant activity of Nelumbo nucifera (sacred lotus) seeds. J Ethnopharmcol 104:322–327
Saidi N, Morita H, Litaudon M, Mukhtar M, Awang K, Hadi AHA (2011) Benzylisoquinoline alkaloids from bark of Cryptocarya rugulosa. Indo J Chem 11:59–66
Sugimoto Y, Furutani S, Itoh A, Tanahashi T, Nakajima H, Oshiro H, Sun S, Yamada J (2008) Effects of extracts and neferine from the embryo of Nelumbo nucifera seeds on the central nervous system. Phytomedicine 15:1117–1124
Van Bergen PF, Hatcher PG, Boon JJ, Xollinson ME, De Leeuw JW (1997) Macromolecular composition of the propagule wall of Nelumbo nucifera. Phytochemistry 45:601–617
Yang Z, Song Z, Xue W, Sheng J, Shu Z, Shi Y, Liang J, Yao X (2014) Synthesis and structure-activity relationship of nuciferine derivative as potential acetylcholinesterase inhibitors. Med Chem Res 23:3178–3186
Zhang XX, Ma SC, Mao P, Wang Y (2016) A new bisbenzylisoquinoline alkaloid from Plumula nelumbinis. Chin Chem Lett 27:1755–1758
Acknowledgements
This work was supported by The Thailand Research Fund (grant no. DBG5980003) and the Center of Excellence for Innovation in Chemistry (PERCH-CIC), Office of the Higher Education Commission.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no competing interests.
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Chaichompoo, W., Chokchaisiri, R., Apiratikul, N. et al. Cytotoxic alkaloids against human colon adenocarcinoma cell line (HT-29) from the seed embryos of Nelumbo nucifera . Med Chem Res 27, 939–943 (2018). https://doi.org/10.1007/s00044-017-2115-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00044-017-2115-3