Abstract
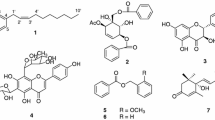
The tubers of Cremastra appendiculata (D. Don) Makino (Orchidaceae) are used in traditional Chinese medicine for the treatment of cancer and bacterial infections. Its 95% ethanol extract was found to display potent inhibitory activities on butyrylcholinesterase (BChE) (IC50 = 23.66 µg/mL) and β-amyloid peptide aggregation (74.09% at 100 μg/mL). Active ethyl acetate extract afforded one bibenzyl (1), four phenanthrenes (2–5), and one rotenoid (6) by a bioassay-guided isolation. Compounds 2, 3, and 5 exhibited potent BChE inhibitory effects with IC50 values of 19.66, 32.80, and 37.79 µM, respectively. Kinetic studies indicated that both 2 and 3 were mixed-type BChE inhibitors. Their strong binding affinities to BChE were further confirmed by fluorescence quenching analysis. Meanwhile, 2 and 3 showed the effective DPPH and ABTS radical scavenging activities (EC50 < 11 μM) and inhibition effect on β-amyloid peptide aggregation (64.49 and 29.50% at 20 μM, respectively), suggesting they could serve as multifunctional potential agents for AD drugs development. This paper revealed the potential medicinal and economic values of C. appendiculata and its chemical constituents in AD prevention/treatment.




Similar content being viewed by others
References
Broering TJ, Morrow GW (1999) Oxygenated dihydrophenanthrenes via quinol acetals: a brief synthesis of orchinol. Synth Commun 29:1135–1142
Brus B, Kosak U, Turk S, Pišlar A, Coquelle N, Kos J, Stojan J, Colletier JP, Gobec S (2014) Discovery, biological evaluation, and crystal structure of a novel nanomolar selective butyrylcholinesterase inhibitor. J Med Chem 57:8167–8179
Coban G, Carlino L, Tarikogullari AH, Parlar S, Sarıkaya G, Alptüzün V, Alpan AS, Güneş HS, Erciyas E (2016) 1H-benzimidazole derivatives as butyrylcholinesterase inhibitors: synthesis and molecular modeling studies. Med Chem Res 25:2005–2014
Cummings JL (2004) Alzheimer’s disease. N Engl J Med 351:56–67
Fang L, Gou S, Fang X, Cheng L, Fleck C (2013) Current progresses of novel natural products and their derivatives/analogs as anti-Alzheimer candidates: an update. Mini Rev Med Chem 13:870–887
Furukawa-Hibi Y, Alkam T, Nitta A, Matsuyama A, Mizoguchi H, Suzuki K, Moussaoui S, Yu QS, Greig NH, Nagai T, Yamada K (2011) Butyrylcholinesterase inhibitors ameliorate cognitive dysfunction induced by amyloid-β peptide in mice. Behav Brain Res 225:222–229
Godyń J, Jończyk J, Panek D, Malawska B (2016) Therapeutic strategies for Alzheimer’s disease in clinical trials. Pharmacol Rep 68:127–138
Greig NH, Utsuki T, Ingram DK, Wang Y, Pepeu G, Scali C, Yu Q, Mamczarz J, Holloway HW, Giordano T, Chen D, Furukawa K, Sambamurti K, Brossi A, Lahiri DK (2005) Selective butyrylcholinesterase inhibition elevates brain acetylcholine, augments learning and lowers Alzheimer β-amyloid peptide in rodent. Proc Natl Acad Sci USA 102:17213–17218
Han SH, Mook-Jung I (2014) Diverse molecular targets for therapeutic strategies in Alzheimer’s disease. J Korean Med Sci 29:893–902
Huang L, Su T, Li X (2013) Natural products as sources of new lead compounds for the treatment of Alzheimer’s disease. Curr Top Med Chem 13:1864–1878
Ikeda Y, Nonaka H, Furumai T, Igarashi Y (2005) Cremastrine, a pyrrolizidine alkaloid from Cremastra appendiculata. J Nat Prod 68:572–573
Krautwurst KD, Tochtermann W (1981) Polycyclische Verbindungen, XX1) Eine einfache Orchinol-Synthese. Chem Ber 114:214–219
Kumar A, Singh A (2015) A review on Alzheimer’s disease pathophysiology and its management: an update. Pharmacol Rep 67:195–203
Leong YW, Kang CC, Harrison LJ, Powell AD (1997) Phenanthrenes, dihydrophenanthrenes and bibenzyls from the orchid Bulbophyllum vaginatum. Phytochemistry 44:157–165
Li Q, Tu Y, Zhu C, Luo W, Huang W, Liu W, Li Y (2017) Cholinesterase, β-amyloid aggregation inhibitory and antioxidant capacities of Chinese medicinal plants. Ind Crops Prod 108:512–519
Libro R, Giacoppo S, Rajan TS, Bramanti P, Mazzon E (2016) Natural phytochemicals in the treatment and prevention of dementia: an overview. Molecules 21:518
Liu L, Li J, Zeng KW, Jiang Y, Tu PF (2015) Five new benzylphenanthrenes from Cremastra appendiculata. Fitoterapia 103:27–32
Majumder PL, Banerjee S (1988) Structure of flavanthrin, the first dimeric 9, 10-dihydrophenanthrene derivative from the orchid Eria flava. Tetrahedron 44:7303–7308
Ng YP, Or TCT, Ip NY (2015) Plant alkaloids as drug leads for Alzheimer’s disease. Neurochem Int 89:260–270
Palmer AM (2011) Neuroprotective therapeutics for Alzheimer’s disease: progress and prospects. Trends Pharmacol Sci 32:141–147
Prince M, Comas-Herrera A, Knapp M, Guerchet M, Karagiannidou M (2016) World Alzheimer report 2016: improving healthcare for people living with dementia: coverage, quality and costs now and in the future. Alzheimer’s Disease International (ADI), London, UK
Shan WJ, Huang L, Zhou Q, Meng FC, Li XS (2011) Synthesis, biological evaluation of 9-N-substituted berberine derivatives as multi-functional agents of antioxidant, inhibitors of acetylcholinesterase, butyrylcholinesterase and amyloid-β aggregation. Eur J Med Chem 46:5885–5893
Shim JS, Kim JH, Lee J, Kim SN, Kwon HJ (2004) Anti-angiogenic activity of a homoisoflavanone from Cremastra appendiculata. Planta Med 70:171–173
Singhal AK, Sharma RP, Baruah JN, Govindan SV, Herz W (1982) Rotenoids from roots of Millettia pachycarpa. Phytochemistry 21:949–951
Tu Y, Zhong Y, Du H, Luo W, Wen Y, Li Q, Zhu C, Li Y (2015) Anticholinesterases and antioxidant alkamides from Piper nigrum fruits. Nat Prod Res 30:1945–1949
Unsal-Tan O, Ozadali-Sari K, Ayazgok B, Küçükkılınç TT, Balkan A (2017) Novel 2-arylbenzimidazole derivatives as multi-targeting agents to treat Alzheimer’s disease. Med Chem Res 26:1506–1515
Wang Y, Guan SH, Meng YH, Zhang YB, Cheng CR, Shi YY, Feng RH, Zeng F, Wu ZY, Zhang JX, Yang M, Liu X, Li Q, Chen XH, Bi KS, Guo DA (2013) Phenanthrenes, 9,10-dihydrophenanthrenes, bibenzyls, with their derivatives, and malate or tartrate benzyl ester glucosides from tubers of Cremastra appendiculata. Phytochemistry 94:268–276
Williams P, Sorribas A, Howes MJR (2011) Natural products as a source of Alzheimer’s drug leads. Nat Prod Rep 28:48–77
Wu D, Yan J, Wang J, Wang Q, Li H (2015) Characterisation of interaction between food colourant allura red AC and human serum albumin: multispectroscopic analyses and docking simulations. Food Chem 170:423–429
Xia X, Cao J, Zheng Y, Wang Q, Xiao J (2014) Flavonoid concentrations and bioactivity of flavonoid extracts from 19 species of ferns from China. Ind Crops Prod 58:91–98
Xue Z, Li S, Wang S, Wang Y, Yang Y, Shi J, He L (2006) Mono-, bi-, and triphenanthrenes from the tubers of Cremastra appendiculata. J Nat Prod 69:907–913
Yamaki M, Bai L, Inoue K, Takagi S (1989) Biphenanthrenes from Bletilla striata. Phytochemistry 28:3503–3505
Zuroff L, Daley D, Black KL, Koronyo-Hamaoui M (2017) Clearance of cerebral Aβ in Alzheimer’s disease: reassessing the role of microglia and monocytes. Cell Mol Life Sci 74:2167–2201
Acknowledgements
Financial support from the Science and Technology Department of Sichuan Province in China (No. 2016JY0062) is gratefully acknowledged.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no competing interests.
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Tu, Y., Huang, J. & Li, Y. Anticholinesterase, antioxidant, and beta-amyloid aggregation inhibitory constituents from Cremastra appendiculata . Med Chem Res 27, 857–863 (2018). https://doi.org/10.1007/s00044-017-2108-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00044-017-2108-2