Abstract
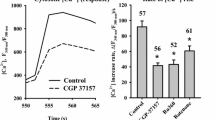
The effect of three different glutamate receptor ligands on mitochondrial membrane potential has been studied in rat pup dissociated cerebellar cells by measuring rhodamine 123 fluorescence. L-glutamate, NMDA (N-methyl-D-aspartate) and kainate (from 10−8 to 10−3 M) decreased in a concentration-dependent manner the mitochondrial membrane potential with EC50 values of 6.7±1.7, 3.8±0.5, and 37.4±14 μM, respectively.
Dizocilpine ((+)MK 801) was able to inhibit the NMDA- and L-glutamate-induced decrease in rhodamine 123 fluorescence, while kainate-induced fluorescence-decreases were unaffected. However, 6-cyano-7-nitroquinoxaline-2,3-dione (CNQX) totally prevented the effect of kainate on mitochondrial membrane potential, but failed to block the L-glutamate effect. It is concluded that, in our cell preparation, L-glutamate exerts its action mainly through NMDA-subtype receptors, and that Ca2+ and Na+ entry through ionotropic glutamate receptors could be responsible for an impairment of mitochondrial membrane potential.
Similar content being viewed by others
References
Ankarcrona M, Dypbukt JM, Bonfoco E, Zhivotovsky B, Orrenius S, Lipton SA, Nicotera P (1995) Glutamate-induced neuronal death: a succession of necrosis or apoptosis depending on mitochondrial function. Neuron 15:961–973
Auer RN (1995) Combination therapy with U74006F (tirilazad mesylate), MK-801, insulin and diazepam in transient forebrain ischaemia. Neurol Res 17:132–136
Bowling AC, Beal MF (1995) Bioenergetic and oxidative stress in neurodegenerative diseases. Life Sci 56:1151–1171
Chen LB (1988) Mitochondrial membrane potential in living cells. Ann Rev Cell Biol 4:155–181
Choi DW (1995) Calcium: still center-stage in hypoxic-ischemic neuronal death. Trends Neurosci 18:58–60
Choi DW (1992) Excitotoxic cell death. J Neurobiol 23:1261–1276
Coyle JT, Puttfarcken P (1993) Oxidative stress, glutamate, and neurodegenerative disorders. Science 262:689–695
Di Porzio U, Smith SV, Novotny EA, Morelli F, Barker JL (1993) Two functionally different glutamate receptors of the kainate subtype in embryonic rat mesencephalic cells. Exp Neurol 120:202–213
Domenici MR, Longo R, Scotti de Carolis A, Frank C, Sagratella S (1993) Protective actions of 21-aminosteroids and MK-801 on hypoxia-induced electrophysiological changes in rat hippocampal slices. Eur J Pharmacol 233:291–293
Dubinsky JM (1993) Examination of the role of calcium in neuronal death. Ann NY Acad Sci 679:34–42
Dykens JA (1994) Isolated cerebral and cerebellar mitochondria produce free radicals when exposed to elevated Ca2+ and Na2+: implications for neurodegeneration. J Neurochem 63:584–591
Frandsen A, Drejer J, Schousboe A (1989) Direct evidence that excitotoxicity in cultured neurons is mediated via N-methyl-D-aspartate (NMDA) as well as non-NMDA receptors. J Neurochem 53:297–299
Grierson JP, Petroski RE, O'Connell SM, Geller HM (1992) Calcium homeostasis in dissociated embryonic neurons: a flow cytometric analysis. J Neurophysiol 67:704–714
Hirose K, Chan PH (1993) Blockade of glutamate excitotoxicity and its clinical applications. Neurochem Res 18:479–483
Holopainen I, Enkvist MOK, Akerman KEO (1989) Glutamate receptor agonists increase intracellular Ca2+ independently of voltage-gated Ca2+ channels in rat cerebellar granule cells. Neurosci Lett 98:57–62
Jesberger JA, Richardson JS (1991) Oxygen free radicals and brain dysfunction. Int J Neurosci 57:1–17
Lopez Lozano JJ, Notter MFD, Gash DM, Leary JF (1989) Selective flow cytometric sorting of viable dopamine neurons. Brain Res 486:351–356
Mandler RN, Schaffner AE, Novotny EA, Lange GD, Barker JL (1988) Flow cytometric analysis of membrane potential in embryonic rat spinal cord cells. J Neurosci Methods 22:203–213
Marcaida G, Miana MD, Grisolfa S, Felipo V (1995) Lack of correlation between glutamate-induced deplection of ATP and neuronal death in primary cultures of cerebellum. Brain Res 695:146–150
Monyer H, Hartley DM, Choi DW (1990) 21-Aminosteroids attenuate excitotoxic neuronal injury in cortical cell cultures. Neuron 5:121–126
Nicotera P, Bellomo G, Orrenius S (1992) Calcium-mediated mechanisms in chemically induced cell death. Annu Rev Pharmacol Toxicol 32:449–470
Oyama Y, Hayashi A, Ueha T, Maekawa K (1995) Characterization of 2′,7′-dichlorofluorescin fluorescence in dissociated mammalian brain neurons: estimation on intracellular content of hydrogen peroxide. Brain Res 635:113–117
Rogawski MA (1995) The NMDA receptor, NMDA antagonists and epilepsy therapy. Drugs 44:279–292
Schieren I, MacDermott A (1988) Flow cytometric identification and purification of cells by ligand-induced changes in intracellular calcium. J Neurosci Methods 26:35–44
St. John PA, Kell WM, Mazzetta JS, Lange GD, Barker JL (1986) Analysis and isolation of embryonic mammalian neurons by fluorescence activated cell sorting. J Neurosci 6:1492–1512
Sureda FX, Camins A, Trullas R, Camarasa J, Escubedo E (1996) A flow cytometric study of N-methyl-D-aspartate effects on dissociated cerebellar cells. Brain Res (in press)
Taylor CP, Meldrum BS (1995) Na+ channels as targets for neuroprotective drugs. Trends Pharmacol Sci 16:309–316
Trump BF, Berezesky IK (1995) Calcium-mediated cell injury and cell death. FASEB J 9:219–228
Wong EHF, Kemp J (1991) Sites for antagonism on the N-methyl-D-aspartate receptor channel complex. Annu Rev Pharmacol Toxicol 31:401–425
Wolf ME, Kapatos G (1989) Flow cytometric analysis of rat striatal nerve terminals. J Neurosci 9:94–105
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Sureda, F.X., Escubedo, E., Gabriel, C. et al. Effect of glutamate receptor ligands on mitochondrial membrane potential in rat dissociated cerebellar cells. Naunyn-Schmiedeberg's Arch Pharmacol 354, 420–423 (1996). https://doi.org/10.1007/BF00168431
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00168431