Abstract
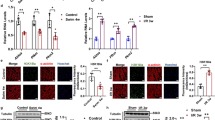
The role of angiotensin II type 1 (AT1) receptors in muscle development and hypertrophy remains unclear. This study was designed to reveal the effects that a loss of AT1 receptors has on skeletal muscle development and hypertrophy in mice. Eight-week-old male AT1a receptor knockout (AT1a−/−) mice were used for this experiment. The plantaris muscle to body weight ratio, muscle fiber cross-sectional area, and number of muscle fibers of AT1a−/− mice was significantly greater than wild type (WT) mice in the non-intervention condition. Next, the functional overload (OL) model was used to induce plantaris muscle hypertrophy by surgically removing the two triceps muscles consisting of the calf, soleus, and gastrocnemius muscles in mice. After 14 days of OL intervention, the plantaris muscle weight, the amount of fiber, and the fiber area increased. However, the magnitude of the increment of plantaris weight was not different between the two strains. Agtr1a mRNA expression did not change after OL in WT muscle. Actually, the Agt mRNA expression level of WT-OL was lower than WT-Control (C) muscle. An atrophy-related gene, atrogin-1 mRNA expression levels of AT1a−/−-C, WT-OL, and AT1a−/−-OL muscle were lower than that of WT-C muscle. Our findings suggest that AT1 receptor contributes to plantaris muscle development via atrogin-1 in mice.



Similar content being viewed by others
References
Akiho M, Nakashima H, Sakata M, Yamasa Y, Yamaguchi A, Sakuma K (2010) Expression profile of Notch-1 in mechanically overloaded plantaris muscle of mice. Life Sci 86(1–2):59–65. doi:10.1016/j.lfs.2009.11.011
Akimoto T, Okuhira K, Aizawa K, Wada S, Honda H, Fukubayashi T, Ushida T (2013) Skeletal muscle adaptation in response to mechanical stress in p130cas−/− mice. Am J Physiol Cell Physiol 304(6):C541–C547. doi:10.1152/ajpcell.00243.2012
Brewster UC, Setaro JF, Perazella MA (2003) The renin–angiotensin–aldosterone system: cardiorenal effects and implications for renal and cardiovascular disease states. Am J Med Sci 326(1):15–24
Brink M, Price SR, Chrast J, Bailey JL, Anwar A, Mitch WE, Delafontaine P (2001) Angiotensin II induces skeletal muscle wasting through enhanced protein degradation and down-regulates autocrine insulin-like growth factor I. Endocrinology 142(4):1489–1496
Di Bari M, van de Poll-Franse LV, Onder G, Kritchevsky SB, Newman A, Harris TB, Williamson JD, Marchionni N, Pahor M; Health, Aging and Body Composition Study (2004) Antihypertensive medications and differences in muscle mass in older persons: the Health, Aging and Body Composition Study. J Am Geriatr Soc 52(6):961–966. doi:10.1111/j.1532-5415.2004.52265.x
Dodson S, Baracos VE, Jatoi A, Evans WJ, Cella D, Dalton JT, Steiner MS (2011) Muscle wasting in cancer cachexia: clinical implications, diagnosis, and emerging treatment strategies. Annu Rev Med 62:265–279. doi:10.1146/annurev-med-061509-131248
Domański M, Ciechanowski K (2012) Sarcopenia: a major challenge in elderly patients with end-stage renal disease. J Aging Res 2012:754739. doi:10.1155/2012/754739
Fearon KC (2008) Cancer cachexia: developing multimodal therapy for a multidimensional problem. Eur J Cancer 44(8):1124–1132. doi:10.1016/j.ejca.2008.02.033
Foley RN, Wang C, Ishani A, Collins AJ, Murray AM (2007) Kidney function and sarcopenia in the United States general population: NHANES III. Am J Nephrol 27(3):279–286. doi:10.1159/000101827
Gensch N, Borchardt T, Schneider A, Riethmacher D, Braun T (2008) Different autonomous myogenic cell populations revealed by ablation of Myf5-expressing cells during mouse embryogenesis. Development 135(9):1597–1604. doi:10.1242/dev.019331
Gordon SE, Davis BS, Carlson CJ, Booth FW (2001) ANG II is required for optimal overload-induced skeletal muscle hypertrophy. Am J Physiol Endocrinol Metab 280(1):E150–E159
Gumucio JP, Mendias CL (2013) Atrogin-1, MuRF-1, and sarcopenia. Endocrine 43(1):12–21. doi:10.1007/s12020-012-9751-7
Hughes SM, Taylor JM, Tapscott SJ, Gurley CM, Carter WJ, Peterson CA (1993) Selective accumulation of MyoD and myogenin mRNAs in fast and slow adult skeletal muscle is controlled by innervation and hormones. Development 118(4):1137–1147
Kim SJ, Roy RR, Kim JA, Zhong H, Haddad F, Baldwin KM, Edgerton VR (2008) Gene expression during inactivity-induced muscle atrophy: effects of brief bouts of a forceful contraction countermeasure. J Appl Physiol 105(4):1246–1254. doi:10.1152/japplphysiol.90668.2008
Kim TN, Park MS, Lim KI, Yang SJ, Yoo HJ, Kang HJ, Song W, Seo JA, Kim SG, Kim NH, Baik SH, Choi DS, Choi KM (2011) Skeletal muscle mass to visceral fat area ratio is associated with metabolic syndrome and arterial stiffness: the Korean Sarcopenic Obesity Study (KSOS). Diabetes Res Clin Pract 93(2):285–291. doi:10.1016/j.diabres.2011.06.013
Macpherson PC, Wang X, Goldman D (2011) Myogenin regulates denervation-dependent muscle atrophy in mouse soleus muscle. J Cell Biochem 112(8):2149–2159. doi:10.1002/jcb.23136
McBride TA (2006) AT1 receptors are necessary for eccentric training-induced hypertrophy and strength gains in rat skeletal muscle. Exp Physiol 91(2):413–421. doi:10.1113/expphysiol.2005.032490
Miyazaki M, McCarthy JJ, Fedele MJ, Esser KA (2011) Early activation of mTORC1 signalling in response to mechanical overload is independent of phosphoinositide 3-kinase/Akt signalling. J Physiol 589(Pt 7):1831–1846. doi:10.1113/jphysiol.2011.205658
Novak ML, Billich W, Smith SM, Sukhija KB, McLoughlin TJ, Hornberger TA, Koh TJ (2009) COX-2 inhibitor reduces skeletal muscle hypertrophy in mice. Am J Physiol Regul Integr Comp Physiol 296(4):R1132–R1139. doi:10.1152/ajpregu.90874.2008
Oishi Y, Ogata T, Yamamoto KI, Terada M, Ohira T, Ohira Y, Taniguchi K, Roy RR (2008) Cellular adaptations in soleus muscle during recovery after hindlimb unloading. Acta Physiol 192(3):381–395. doi:10.1111/j.1748-1716.2007.01747.x
Onder G, Penninx BW, Balkrishnan R, Fried LP, Chaves PH, Williamson J, Carter C, Di Bari M, Guralnik JM, Pahor M (2002) Relation between use of angiotensin-converting enzyme inhibitors and muscle strength and physical function in older women: an observational study. Lancet 359(9310):926–930
Park SW, Goodpaster BH, Lee JS, Kuller LH, Boudreau R, de Rekeneire N, Harris TB, Kritchevsky S, Tylavsky FA, Nevitt M, Cho YW, Newman AB; Health, Aging, and Body Composition Study (2009) Excessive loss of skeletal muscle mass in older adults with type 2 diabetes. Diabetes Care 32(11):1993–1997. doi:10.2337/dc09-0264
Pierzchalski P, Reiss K, Cheng W, Cirielli C, Kajstura J, Nitahara JA, Rizk M, Capogrossi MC, Anversa P (1997) p53 Induces myocyte apoptosis via the activation of the renin–angiotensin system. Exp Cell Res 234(1):57–65. doi:10.1006/excr.1997.3604
Putman CT, Düsterhöft S, Pette D (2000) Satellite cell proliferation in low frequency-stimulated fast muscle of hypothyroid rat. Am J Physiol Cell Physiol 279(3):C682–C690
Rezk BM, Yoshida T, Semprun-Prieto L, Higashi Y, Sukhanov S, Delafontaine P (2012) Angiotensin II infusion induces marked diaphragmatic skeletal muscle atrophy. PLoS One 7(1):e30276. doi:10.1371/journal.pone.0030276
Roubenoff R, Castaneda C (2001) Sarcopenia-understanding the dynamics of aging muscle. JAMA 286(10):1230–1231
Russell ST, Sanders PM, Tisdale MJ (2006) Angiotensin II directly inhibits protein synthesis in murine myotubes. Cancer Lett 231(2):290–294. doi:10.1016/j.canlet.2005.02.007
Sanada K, Iemitsu M, Murakami H, Gando Y, Kawano H, Kawakami R, Tabata I, Miyachi M (2012) Adverse effects of coexistence of sarcopenia and metabolic syndrome in Japanese women. Eur J Clin Nutr 66(10):1093–1098. doi:10.1038/ejcn.2012.43
Sanders PM, Russell ST, Tisdale MJ (2005) Angiotensin II directly induces muscle protein catabolism through the ubiquitin-proteasome proteolytic pathway and may play a role in cancer cachexia. Br J Cancer 93(4):425–434. doi:10.1038/sj.bjc.6602725
Semprun-Prieto LC, Sukhanov S, Yoshida T, Rezk BM, Gonzalez-Villalobos RA, Vaughn C, Michael Tabony A, Delafontaine P (2011) Angiotensin II induced catabolic effect and muscle atrophy are redox dependent. Biochem Biophys Res Commun 409(2):217–221. doi:10.1016/j.bbrc.2011.04.122
Song YH, Li Y, Du J, Mitch WE, Rosenthal N, Delafontaine P (2005) Muscle-specific expression of IGF-1 blocks angiotensin II-induced skeletal muscle wasting. J Clin Invest 115(2):451–458. doi:10.1172/JCI22324
Strawbridge WJ, Shema SJ, Balfour JL, Higby HR, Kaplan GA (1998) Antecedents of frailty over three decades in an older cohort. J Gerontol B Psychol Sci Soc Sci 53(1):S9–S16
Sugaya T, Nishimatsu S, Tanimoto K, Takimoto E, Yamagishi T, Imamura K, Goto S, Imaizumi K, Hisada Y, Otsuka A, Uchida H, Sugiura M, Fukuta K, Fukamizu A, Murakami K (1995) Angiotensin II type 1a receptor-deficient mice with hypotension and hyperreninemia. J Biol Chem 270(32):18719–18722
Sukhanov S, Semprun-Prieto L, Yoshida T, Michael Tabony A, Higashi Y, Galvez S, Delafontaine P (2011) Angiotensin II, oxidative stress and skeletal muscle wasting. Am J Med Sci 342(2):143–147. doi:10.1097/MAJ.0b013e318222e620
Sumukadas D, Witham MD, Struthers AD, McMurdo ME (2007) Effect of perindopril on physical function in elderly people with functional impairment: a randomized controlled trial. CMAJ 177(8):867–874. doi:10.1503/cmaj.061339
Yoshida T, Semprun-Prieto L, Sukhanov S, Delafontaine P (2010) IGF-1 prevents ANG II-induced skeletal muscle atrophy via Akt- and Foxo-dependent inhibition of the ubiquitin ligase atrogin-1 expression. Am J Physiol Heart Circ Physiol 298(5):H1565–H1570. doi:10.1152/ajpheart.00146.2010
Acknowledgments
We are grateful to Professor Akiyoshi Fukamizu for providing the ATla−/− mice. We would also like to thank Ms. Noriko Tamura and Ms. Yasuko Matsuda for their excellent technical assistance.
Grants
This work was supported by the Japan Society for the Promotion of Science (JSPS) KAKENHI Grant Number T25930017.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by: Maciej Szydlowski
Rights and permissions
About this article
Cite this article
Zempo, H., Suzuki, Ji., Ogawa, M. et al. A different role of angiotensin II type 1a receptor in the development and hypertrophy of plantaris muscle in mice. J Appl Genetics 57, 91–97 (2016). https://doi.org/10.1007/s13353-015-0291-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13353-015-0291-8