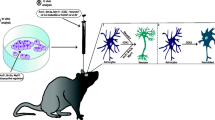
Abstract
Cell transplantation is an attractive therapeutic avenue for injuries of the central nervous system (CNS) and neurodegenerative disorders. Transplanted cells are able to restore the cells that are lost in the injury process, including neurons, oligodendrocytes, and astrocytes. Various clinically relevant cell types and sources have been explored thus far, including induced pluripotent stem cells (iPSCs), which can be used for autologous transplantation. Despite this advantage, differentiation of iPSCs remains time consuming, which may be a limitation in urgent clinical cases. Additionally, the intermediate pluripotent state increases the risk of tumorigenicity when transplanting iPSCs. In this regard, research efforts have shifted toward the transdifferentiation of somatic cells into a variety of neural cell types, including neurons, astrocytes, and oligodendrocytes or their progenitors. This method bypasses the pluripotent stage to reduce the risk of tumorigenicity, thus reducing the induction timeline while still maintaining the patient-specific capacity of the cells. Neural cells or their progenitors can be differentiated in vitro using a number of methods, including transient expression or suppression of certain transcription and chromatin remodeling factors through gene manipulation, or miRNA and small molecule treatment. Recently, research efforts have also focused on in vivo transdifferentiation, in which endogenous cells are targeted for conversion into cell types of interest. The following chapter will focus on the general principles of direct neural lineage conversion, the methods used to derive particular cell types, and their application to injuries of the CNS.
Katarzyna Pieczonka and William Brett Mclntyre have contributed equally to this chapter.
Similar content being viewed by others
Abbreviations
- AD:
-
Alzheimer’s disease
- BMA:
-
Ascl1, Brn2, Mytl1
- CNS:
-
Central nervous system
- CPZ:
-
Cuprizone
- CRISPRa:
-
CRISPR activation
- DCas9:
-
Dead form of Cas9
- DCX:
-
Doublecortin
- DRNPCs:
-
Directly reprogrammed neural progenitor cells
- HDAC:
-
Histone deacetylase
- FACS:
-
Fluorescent activated cell sorting
- IPSCs:
-
Induced pluripotent stem cell
- INSCs:
-
Induced neural stem cells
- miRNA:
-
Micro-RNA
- MACS:
-
Magnetic-activated cell sorting
- MBD2:
-
Methyl-CpG-binding domain protein 2
- MS:
-
Multiple sclerosis
- MSI1:
-
Musashi
- Ngn2:
-
Neurogenin-1
- NSCs:
-
Neural stem/progenitor cells
- OPC:
-
Oligodendrocyte precursor cell
- ORF:
-
Open reading frame
- PD:
-
Parkinson’s disease
- RA:
-
Retinoic acid
- SCI:
-
Spinal cord injury
- SCRNASeq:
-
Single-cell RNA sequencing
- SGRNA:
-
Single guide RNA
- SHH:
-
Sonic hedgehog
- TBI:
-
Traumatic brain injury
- TF:
-
Transcription factor
- 6-OHDA:
-
6-hydroxydopamine
References
Ahlfors J-E, Azimi A, El-Ayoubi R, Velumian A, Vonderwalde I, Boscher C, Mihai O et al (2019) Examining the fundamental biology of a novel population of directly reprogrammed human neural precursor cells. Stem Cell Res Ther 10:166. https://doi.org/10.1186/s13287-019-1255-4
Ahmed RPH, Haider KH, Buccini S, Shujia J, Ashraf M (2011a) Reprogramming of skeletal myoblasts for induction of pluripotency for tumor free cardiomyogenesis in the infarcted heart. Circ Res 109:60–70. https://doi.org/10.1161/CIRCRESAHA.110.240010
Ahmed RPH, Ashraf M, Buccini S, Shujia J, Haider KH (2011b) Cardiac tumorigenic potential of induced pluripotent stem cells in immunocompetent host with myocardial infarction. Regen Med 6(758):171–178. https://doi.org/10.2217/rme.10.103
Ahmed RPH, Buccini S, Jiang S, Haider KH (2012) Surrogate progenitors for cardiogenesis: direct reprogramming of somatic cells to cross lineage restriction without pluripotency. Circulation (Suppl)
Alizadeh A, Dyck SM, Karimi-Abdolrezaee S (2019) Traumatic spinal cord injury: an overview of pathophysiology, models and acute injury mechanisms. Front Neurol 10:282. https://doi.org/10.3389/fneur.2019.00282
Ambasudhan R, Talantova M, Coleman R, Yuan X, Zhu S, Lipton SA, Ding S (2011) Direct reprogramming of adult human fibroblasts to functional neurons under defined conditions. Cell Stem Cell 9:113–118. https://doi.org/10.1016/j.stem.2011.07.002
Bechler ME, Byrne L, Ffrench-Constant C (2015) CNS myelin sheath lengths are an intrinsic property of oligodendrocytes. Curr Biol 25:2411–2416. https://doi.org/10.1016/j.cub.2015.07.056
Ben-Zwi M, Petrou P, Halimi M, Karussis D, Kassis I (2019) Neuralized mesenchymal stem cells (NMSC) exhibit phenotypical, and biological evidence of neuronal transdifferentiation and suppress EAE more effectively than unmodified MSC. Immunol Lett 212:6–13. https://doi.org/10.1016/j.imlet.2019.05.009
Bradl M, Lassmann H (2010) Oligodendrocytes: biology and pathology. Acta Neuropathol (Berl) 119:37–53. https://doi.org/10.1007/s00401-009-0601-5
Buccini S, Haider KH, Ahmed RPH, Jiang S, Ashraf M (2012) Cardiac progenitors derived from reprogrammed mesenchymal stem cells contribute to angiomyogenic repair of the infarcted heart. Basic Res Cardiol 107(6):301. https://doi.org/10.1007/s00395-012-0301-5
Cagavi E, Akgul Caglar T, Soztekin GI, Haider KH (2018) Patient-specific induced pluripotent stem cells for cardiac disease modelling. In: Haider KH, Aziz S (eds) Stem cells: from hype to real hope. Medicine & Life Sciences, DE GRUYTER, Berlin
Caiazzo M, Dell’Anno MT, Dvoretskova E, Lazarevic D, Taverna S, Leo D, Sotnikova TD et al (2011) Direct generation of functional dopaminergic neurons from mouse and human fibroblasts. Nature 476:224–227. https://doi.org/10.1038/nature10284
Cassady JP, D’Alessio AC, Sarkar S, Dani VS, Fan ZP, Ganz K, Roessler R et al (2014) Direct lineage conversion of adult mouse liver cells and B lymphocytes to neural stem cells. Stem Cell Rep 3:948–956. https://doi.org/10.1016/j.stemcr.2014.10.001
Chen Y-C, Ma N-X, Pei Z-F, Wu Z, Do-Monte FH, Keefe S, Yellin E et al (2020) A NeuroD1 AAV-based gene therapy for functional brain repair after ischemic injury through in vivo astrocyte-to-neuron conversion. Mol Ther J Am Soc Gene Ther 28:217–234. https://doi.org/10.1016/j.ymthe.2019.09.003
D’Alessio AC, Fan ZP, Wert KJ, Baranov P, Cohen MA, Saini JS, Cohick E et al (2015) A systematic approach to identify candidate transcription factors that control cell identity. Stem Cell Rep 5:763–775. https://doi.org/10.1016/j.stemcr.2015.09.016
Dai P, Harada Y, Takamatsu T (2015) Highly efficient direct conversion of human fibroblasts to neuronal cells by chemical compounds. J Clin Biochem Nutr 56:166–170. https://doi.org/10.3164/jcbn.15-39
Davis RL, Weintraub H, Lassar AB (1987) Expression of a single transfected cDNA converts fibroblasts to myoblasts. Cell 51:987–1000. https://doi.org/10.1016/0092-8674(87)90585-x
Dell’Anno MT, Caiazzo M, Leo D, Dvoretskova E, Medrihan L, Colasante G, Giannelli S et al (2014) Remote control of induced dopaminergic neurons in parkinsonian rats. J Clin Invest 124:3215–3229. https://doi.org/10.1172/JCI74664
Dias DO, Kim H, Holl D, Solnestam BW, Lundeberg J, Carlén M, Göritz C et al (2018) Reducing pericyte-derived scarring promotes recovery after spinal cord injury. Cell 173:153–165.e22. https://doi.org/10.1016/j.cell.2018.02.004
Ebrahimi-Barough S, Massumi M, Kouchesfahani HM, Ai J (2013) Derivation of pre-oligodendrocytes from human endometrial stromal cells by using overexpression of MicroRNA 338. J Mol Neurosci 51:337–343. https://doi.org/10.1007/s12031-013-0101-x
Faissner S, Plemel JR, Gold R, Yong VW (2019) Progressive multiple sclerosis: from pathophysiology to therapeutic strategies. Nat Rev Drug Discov 18:905–922. https://doi.org/10.1038/s41573-019-0035-2
Fernandez-Castaneda A, Gaultier A (2016) Adult oligodendrocyte progenitor cells – multifaceted regulators of the CNS in health and disease. Brain Behav Immun 57:1–7. https://doi.org/10.1016/j.bbi.2016.01.005
Gao S, Guo X, Zhao S, Jin Y, Zhou F, Yuan P, Cao L et al (2019) Differentiation of human adipose-derived stem cells into neuron/motoneuron-like cells for cell replacement therapy of spinal cord injury. Cell Death Dis 10:597. https://doi.org/10.1038/s41419-019-1772-1
Ghasemi-Kasman M, Zare L, Baharvand H, Javan M (2018) In vivo conversion of astrocytes to myelinating cells by miR-302/367 and valproate to enhance myelin repair. J Tissue Eng Regen Med 12:e462–e472. https://doi.org/10.1002/term.2276
Gilbert LA, Larson MH, Morsut L, Liu Z, Brar GA, Torres SE, Stern-Ginossar N et al (2013) CRISPR-mediated modular RNA-guided regulation of transcription in eukaryotes. Cell 154:442–451. https://doi.org/10.1016/j.cell.2013.06.044
Gu X, Fu C, Lin L, Liu S, Su X, Li A, Wu Q et al (2018) miR-124 and miR-9 mediated downregulation of HDAC5 promotes neurite development through activating MEF2C-GPM6A pathway. J Cell Physiol 233:673–687. https://doi.org/10.1002/jcp.25927
Guo Z, Zhang L, Wu Z, Chen Y, Wang F, Chen G (2014) In vivo direct reprogramming of reactive glial cells into functional neurons after brain injury and in an Alzheimer’s disease model. Cell Stem Cell 14:188–202. https://doi.org/10.1016/j.stem.2013.12.001
Haider KH, Khan M, Sen C (2015) Chapter 22: MicroRNAs with mega functions in cardiac remodeling and repair: the micromanagement of matters of the heart. In: Sen CK (ed) MicroRNA in regenerative medicine. Elsevier, pp 569–600
Hampel H, Mesulam M-M, Cuello AC, Farlow MR, Giacobini E, Grossberg GT, Khachaturian AS et al (2018) The cholinergic system in the pathophysiology and treatment of Alzheimer’s disease. Brain J Neurol 141:1917–1933. https://doi.org/10.1093/brain/awy132
Heinrich C, Bergami M, Gascón S, Lepier A, Viganò F, Dimou L, Sutor B et al (2014) Sox2-mediated conversion of NG2 glia into induced neurons in the injured adult cerebral cortex. Stem Cell Rep 3:1000–1014. https://doi.org/10.1016/j.stemcr.2014.10.007
Heng BC, Haider KH, Sim EKW, Cao T, Tong GO, Ng SC (2005) Reprogramming autologous skeletal myoblasts to express cardiomyogenic function: challenges and possible approaches. Int J Cardiol 100(3):355–362. https://doi.org/10.1016/j.ijcard.2004.06.009
Horiuchi M, Suzuki-Horiuchi Y, Akiyama T, Itoh A, Pleasure D, Carstens E, Itoh T (2017) Differing intrinsic biological properties between forebrain and spinal oligodendroglial lineage cells. J Neurochem 142:378–391. https://doi.org/10.1111/jnc.14074
Hu W, Qiu B, Guan W, Wang Q, Wang M, Li W, Gao L et al (2015) Direct conversion of normal and Alzheimer’s disease human fibroblasts into neuronal cells by small molecules. Cell Stem Cell 17:204–212. https://doi.org/10.1016/j.stem.2015.07.006
Hu X, Qin S, Huang X, Yuan Y, Tan Z, Gu Y, Cheng X et al (2019) Region-restrict astrocytes exhibit heterogeneous susceptibility to neuronal reprogramming. Stem Cell Rep 12:290–304. https://doi.org/10.1016/j.stemcr.2018.12.017
Ibrahim AY, Mehdi Q, Abbas AO, Alashkar A, Haider KH (2016) Induced pluripotent stem cells: next generation cells for tissue regeneration. J Biomed Sci Eng 9(4):226–244
Karow M, Sánchez R, Schichor C, Masserdotti G, Ortega F, Heinrich C, Gascón S et al (2012) Reprogramming of pericyte-derived cells of the adult human brain into induced neuronal cells. Cell Stem Cell 11:471–476. https://doi.org/10.1016/j.stem.2012.07.007
Karow M, Camp JG, Falk S, Gerber T, Pataskar A, Gac-Santel M, Kageyama J et al (2018) Direct pericyte-to-neuron reprogramming via unfolding of a neural stem cell-like program. Nat Neurosci 21:932–940. https://doi.org/10.1038/s41593-018-0168-3
Kumar A, Dudhal S, Sundari TA, Sunkara M, Usman H, Varshney A, Mukhopadhyay A (2016) Dopaminergic-primed fetal liver mesenchymal stromal-like cells can reverse parkinsonian symptoms in 6-hydroxydopamine-lesioned mice. Cytotherapy 18:307–319. https://doi.org/10.1016/j.jcyt.2015.11.007
Kuriakose D, Xiao Z (2020) Pathophysiology and treatment of stroke: present status and future perspectives. Int J Mol Sci 21:7609. https://doi.org/10.3390/ijms21207609
Ladewig J, Mertens J, Kesavan J, Doerr J, Poppe D, Glaue F, Herms S et al (2012) Small molecules enable highly efficient neuronal conversion of human fibroblasts. Nat Methods 9:575–578. https://doi.org/10.1038/nmeth.1972
Laredo F, Plebanski J, Tedeschi A (2019) Pericytes: problems and promises for CNS repair. Front Cell Neurosci 13:546. https://doi.org/10.3389/fncel.2019.00546
Li X, Zuo X, Jing J, Ma Y, Wang J, Liu D, Zhu J et al (2015) Small-molecule-driven direct reprogramming of mouse fibroblasts into functional neurons. Cell Stem Cell 17:195–203. https://doi.org/10.1016/j.stem.2015.06.003
Liang X-G, Tan C, Wang C-K, Tao R-R, Huang Y-J, Ma K-F, Fukunaga K et al (2018) Myt1l induced direct reprogramming of pericytes into cholinergic neurons. CNS Neurosci Ther 24:801–809. https://doi.org/10.1111/cns.12821
Liu Y, Yu C, Daley TP, Wang F, Cao WS, Bhate S, Lin X et al (2018) CRISPR activation screens systematically identify factors that drive neuronal fate and reprogramming. Cell Stem Cell 23:758–771.e8. https://doi.org/10.1016/j.stem.2018.09.003
Ma Y, Xie H, Du X, Wang L, Jin X, Zhang Q, Han Y et al (2021) In vivo chemical reprogramming of astrocytes into neurons. Cell Discov 7:1–13. https://doi.org/10.1038/s41421-021-00243-8
Mandai M, Watanabe A, Kurimoto Y, Hirami Y, Morinaga C, Daimon T, Fujihara M et al (2017) Autologous induced stem-cell–derived retinal cells for macular degeneration. N Engl J Med 376:1038–1046. https://doi.org/10.1056/NEJMoa1608368
Marro S, Pang ZP, Yang N, Tsai M-C, Qu K, Chang HY, Südhof TC et al (2011) Direct lineage conversion of terminally differentiated hepatocytes to functional neurons. Cell Stem Cell 9:374–382. https://doi.org/10.1016/j.stem.2011.09.002
McGinn MJ, Povlishock JT (2016) Pathophysiology of traumatic brain injury. Neurosurg Clin N Am 27:397–407. https://doi.org/10.1016/j.nec.2016.06.002
Mertens J, Paquola ACM, Ku M, Hatch E, Böhnke L, Ladjevardi S, McGrath S et al (2015) Directly reprogrammed human neurons retain aging-associated transcriptomic signatures and reveal age-related nucleocytoplasmic defects. Cell Stem Cell 17:705–718. https://doi.org/10.1016/j.stem.2015.09.001
Moreno-Luna R, Esteban PF, Paniagua-Torija B, Arevalo-Martin A, Garcia-Ovejero D, Molina-Holgado E (2021) Heterogeneity of the endocannabinoid system between cerebral cortex and spinal cord oligodendrocytes. Mol Neurobiol 58:689–702. https://doi.org/10.1007/s12035-020-02148-1
Mothe AJ, Zahir T, Santaguida C, Cook D, Tator CH (2011) Neural stem/progenitor cells from the adult human spinal cord are multipotent and self-renewing and differentiate after transplantation. PLoS One 6:e27079. https://doi.org/10.1371/journal.pone.0027079
Nagoshi N, Khazaei M, Ahlfors J-E, Ahuja CS, Nori S, Wang J, Shibata S et al (2018) Human spinal oligodendrogenic neural progenitor cells promote functional recovery after spinal cord injury by axonal remyelination and tissue sparing: oligodendrogenic NPCs for spinal cord injury. Stem Cells Transl Med 7:806–818. https://doi.org/10.1002/sctm.17-0269
Niu W, Zang T, Zou Y, Fang S, Smith DK, Bachoo R, Zhang C-L (2013) In vivo reprogramming of astrocytes to neuroblasts in the adult brain. Nat Cell Biol 15:1164–1175. https://doi.org/10.1038/ncb2843
Niu W, Zang T, Smith DK, Vue TY, Zou Y, Bachoo R, Johnson JE et al (2015) SOX2 reprograms resident astrocytes into neural progenitors in the adult brain. Stem Cell Rep 4:780–794. https://doi.org/10.1016/j.stemcr.2015.03.006
Pang ZP, Yang N, Vierbuchen T, Ostermeier A, Fuentes DR, Yang TQ, Citri A et al (2011) Induction of human neuronal cells by defined transcription factors. Nature 476:220–223. https://doi.org/10.1038/nature10202
Pereira M, Birtele M, Shrigley S, Benitez JA, Hedlund E, Parmar M, Ottosson DR (2017) Direct reprogramming of resident NG2 glia into neurons with properties of fast-spiking Parvalbumin-containing interneurons. Stem Cell Rep 9:742–751. https://doi.org/10.1016/j.stemcr.2017.07.023
Pfisterer U, Kirkeby A, Torper O, Wood J, Nelander J, Dufour A, Björklund A et al (2011) Direct conversion of human fibroblasts to dopaminergic neurons. Proc Natl Acad Sci U S A 108:10343–10348. https://doi.org/10.1073/pnas.1105135108
Pfisterer U, Ek F, Lang S, Soneji S, Olsson R, Parmar M (2016) Small molecules increase direct neural conversion of human fibroblasts. Sci Rep 6:38290. https://doi.org/10.1038/srep38290
Qi R, Yang C (2018) Renal tubular epithelial cells: the neglected mediator of tubulointerstitial fibrosis after injury. Cell Death Dis 9:1–11. https://doi.org/10.1038/s41419-018-1157-x
Qin H, Zhao A, Zhang C, Fu X (2016) Epigenetic control of reprogramming and transdifferentiation by histone modifications. Stem Cell Rev Rep 12:708–720. https://doi.org/10.1007/s12015-016-9682-4
Qiu X-C, Jin H, Zhang R-Y, Ding Y, Zeng X, Lai B-Q, Ling E-A et al (2015) Donor mesenchymal stem cell-derived neural-like cells transdifferentiate into myelin-forming cells and promote axon regeneration in rat spinal cord transection. Stem Cell Res Ther 6:105. https://doi.org/10.1186/s13287-015-0100-7
Rackham OJL, Firas J, Fang H, Oates ME, Holmes ML, Knaupp AS, FANTOM Consortium et al (2016) A predictive computational framework for direct reprogramming between human cell types. Nat Genet 48:331–335. https://doi.org/10.1038/ng.3487
Ringsrud KM (2001) Cells in the urine sediment. Lab Med 32:153–155. https://doi.org/10.1309/60JQ-6F5F-1V9E-AGGB
Rinn JL, Wang JK, Liu H, Montgomery K, van de Rijn M, Chang HY (2008) A systems biology approach to anatomic diversity of skin. J Invest Dermatol 128:776–782. https://doi.org/10.1038/sj.jid.5700986
Rivetti di Val Cervo P, Romanov RA, Spigolon G, Masini D, Martín-Montañez E, Toledo EM, La Manno G et al (2017) Induction of functional dopamine neurons from human astrocytes in vitro and mouse astrocytes in a Parkinson’s disease model. Nat Biotechnol 35:444–452. https://doi.org/10.1038/nbt.3835
Rowe RG, Daley GQ (2019) Induced pluripotent stem cells in disease modelling and drug discovery. Nat Rev Genet 20:377–388. https://doi.org/10.1038/s41576-019-0100-z
Shibata M, Kurokawa D, Nakao H, Ohmura T, Aizawa S (2008) MicroRNA-9 modulates Cajal–Retzius cell differentiation by suppressing Foxg1 expression in mouse medial pallium. J Neurosci 28:10415–10421. https://doi.org/10.1523/JNEUROSCI.3219-08.2008
Simon DK, Tanner CM, Brundin P (2020) Parkinson disease epidemiology, pathology, genetics and pathophysiology. Clin Geriatr Med 36:1–12. https://doi.org/10.1016/j.cger.2019.08.002
Song B, Cha Y, Ko S, Jeon J, Lee N, Seo H, Park K-J et al (2020) Human autologous iPSC–derived dopaminergic progenitors restore motor function in Parkinson’s disease models. J Clin Invest 130:904–920. https://doi.org/10.1172/JCI130767
Strano A, Tuck E, Stubbs VE, Livesey FJ (2020) Variable outcomes in neural differentiation of human PSCs arise from intrinsic differences in developmental signaling pathways. Cell Rep 31:107732. https://doi.org/10.1016/j.celrep.2020.107732
Su Z, Niu W, Liu M-L, Zou Y, Zhang C-L (2014) In vivo conversion of astrocytes to neurons in the injured adult spinal cord. Nat Commun 5:3338. https://doi.org/10.1038/ncomms4338
Sullivan GM, Knutsen AK, Peruzzotti-Jametti L, Korotcov A, Bosomtwi A, Dardzinski BJ, Bernstock JD et al (2020) Transplantation of induced neural stem cells (iNSCs) into chronically demyelinated corpus callosum ameliorates motor deficits. Acta Neuropathol Commun 8:84. https://doi.org/10.1186/s40478-020-00960-3
Tai W, Xu X-M, Zhang C-L (2020) Regeneration through in vivo cell fate reprogramming for neural repair. Front Cell Neurosci 14:107. https://doi.org/10.3389/fncel.2020.00107
Tang Y, Liu M-L, Zang T, Zhang C-L (2017) Direct reprogramming rather than iPSC-based reprogramming maintains aging hallmarks in human motor neurons. Front Mol Neurosci 10:359. https://doi.org/10.3389/fnmol.2017.00359
Thier MC, Hommerding O, Panten J, Pinna R, García-González D, Berger T, Wörsdörfer P et al (2019) Identification of embryonic neural plate border stem cells and their generation by direct reprogramming from adult human blood cells. Cell Stem Cell 24:166–182.e13. https://doi.org/10.1016/j.stem.2018.11.015
Torper O, Ottosson DR, Pereira M, Lau S, Cardoso T, Grealish S, Parmar M (2015) In vivo reprogramming of striatal NG2 glia into functional neurons that integrate into local host circuitry. Cell Rep 12:474–481. https://doi.org/10.1016/j.celrep.2015.06.040
Toyokuni S (2015) Chemical conversion of human fibroblasts into neuronal cells: dawn of future clinical trials. J Clin Biochem Nutr 56:165. https://doi.org/10.3164/jcbn.56-3-editorial
Tsunemoto RK, Eade KT, Blanchard JW, Baldwin KK (2015) Forward engineering neuronal diversity using direct reprogramming. EMBO J 34:1445–1455. https://doi.org/10.15252/embj.201591402
Tsunemoto R, Lee S, Szűcs A, Chubukov P, Sokolova I, Blanchard JW, Eade KT et al (2018) Diverse reprogramming codes for neuronal identity. Nature 557:375–380. https://doi.org/10.1038/s41586-018-0103-5
Ullah M, Iqbal H, Khan I, Salim A (2021) Chapter 9: Chromatin remodeling and cardiac differentiation of stem cells. In: Haider KH (ed) Stem cells: from potential to promise. Springer NATURE, pp 251–271
Vierbuchen T, Ostermeier A, Pang ZP, Kokubu Y, Südhof TC, Wernig M (2010) Direct conversion of fibroblasts to functional neurons by defined factors. Nature 463:1035–1041. https://doi.org/10.1038/nature08797
Vonderwalde I, Azimi A, Rolvink G, Ahlfors J-E, Shoichet MS, Morshead CM (2020) Transplantation of directly reprogrammed human neural precursor cells following stroke promotes synaptogenesis and functional recovery. Transl Stroke Res 11:93–107. https://doi.org/10.1007/s12975-019-0691-x
Wang L, Wang L, Huang W, Su H, Xue Y, Su Z, Liao B et al (2013) Generation of integration-free neural progenitor cells from cells in human urine. Nat Methods 10:84–89. https://doi.org/10.1038/nmeth.2283
Wang H, Song G, Chuang H, Chiu C, Abdelmaksoud A, Ye Y, Zhao L (2018) Portrait of glial scar in neurological diseases. Int J Immunopathol Pharmacol 31. https://doi.org/10.1177/2058738418801406
Wang L-L, Garcia CS, Zhong X, Ma S, Zhang C-L (2020) Rapid and efficient in vivo astrocyte-to-neuron conversion with regional identity and connectivity? bioRxiv 2020.08.16.253195. https://doi.org/10.1101/2020.08.16.253195
Wapinski OL, Lee QY, Chen AC, Li R, Corces MR, Ang CE, Treutlein B et al (2017) Rapid chromatin switch in the direct reprogramming of fibroblasts to neurons. Cell Rep 20:3236–3247. https://doi.org/10.1016/j.celrep.2017.09.011
Wu J, Sheng C, Liu Z, Jia W, Wang B, Li M, Fu L et al (2015) Lmx1a enhances the effect of iNSCs in a PD model. Stem Cell Res 14:1–9. https://doi.org/10.1016/j.scr.2014.10.004
Wuttke TV, Markopoulos F, Padmanabhan H, Wheeler AP, Murthy VN, Macklis JD (2018) Developmentally primed cortical neurons maintain fidelity of differentiation and establish appropriate functional connectivity after transplantation. Nat Neurosci 21:517–529. https://doi.org/10.1038/s41593-018-0098-0
Xie X, Fu Y, Liu J (2017) Chemical reprogramming and transdifferentiation. Curr Opin Genet Dev 46:104–113. https://doi.org/10.1016/j.gde.2017.07.003
Xue Y, Ouyang K, Huang J, Zhou Y, Ouyang H, Li H, Wang G et al (2013) Direct conversion of fibroblasts to neurons by reprogramming PTB-regulated microRNA circuits. Cell 152:82–96. https://doi.org/10.1016/j.cell.2012.11.045
Yang H, Hao D, Liu C, Huang D, Chen B, Fan H, Liu C et al (2019) Generation of functional dopaminergic neurons from human spermatogonial stem cells to rescue parkinsonian phenotypes. Stem Cell Res Ther 10:195. https://doi.org/10.1186/s13287-019-1294-x
Yildirim T, Okutan O, Akpinar E, Yilmaz A, Isik HS (2018) Neuroprotective effects of high-dose human albumin against traumatic spinal cord injury in rats. Bratisl Med J 119:86–91. https://doi.org/10.4149/BLL_2018_016
Yoo AS, Sun AX, Li L, Shcheglovitov A, Portmann T, Li Y, Lee-Messer C et al (2011) MicroRNA-mediated conversion of human fibroblasts to neurons. Nature 476:228–231. https://doi.org/10.1038/nature10323
Yoon H, Walters G, Paulsen AR, Scarisbrick IA (2017) Astrocyte heterogeneity across the brain and spinal cord occurs developmentally, in adulthood and in response to demyelination. PLoS One 12:e0180697. https://doi.org/10.1371/journal.pone.0180697
Zhang L, Yin J-C, Yeh H, Ma N-X, Lee G, Chen XA, Wang Y et al (2015a) Small molecules efficiently reprogram human Astroglial cells into functional neurons. Cell Stem Cell 17:735–747. https://doi.org/10.1016/j.stem.2015.09.012
Zhang S-Z, Ma L-X, Qian W-J, Li H-F, Wang Z-F, Wang H-X, Wu Z-Y (2015b) Modeling neurological disease by rapid conversion of human urine cells into functional neurons. Stem Cells Int 2016:e2452985. https://doi.org/10.1155/2016/2452985
Author information
Authors and Affiliations
Corresponding authors
Editor information
Editors and Affiliations
Section Editor information
Rights and permissions
Copyright information
© 2022 Springer Nature Singapore Pte Ltd.
About this entry
Cite this entry
Pieczonka, K., McIntyre, W.B., Khazaei, M., Fehlings, M.G. (2022). Direct Reprogramming Strategies for the Treatment of Nervous System Injuries and Neurodegenerative Disorders. In: Haider, K.H. (eds) Handbook of Stem Cell Therapy. Springer, Singapore. https://doi.org/10.1007/978-981-16-6016-0_14-1
Download citation
DOI: https://doi.org/10.1007/978-981-16-6016-0_14-1
Received:
Accepted:
Published:
Publisher Name: Springer, Singapore
Print ISBN: 978-981-16-6016-0
Online ISBN: 978-981-16-6016-0
eBook Packages: Springer Reference Biomedicine and Life SciencesReference Module Biomedical and Life Sciences