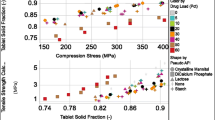
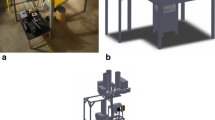
Abstract
The properties of raw materials can affect the properties and quality of intermediates and the final product. The flow behavior of powders, the form of the raw materials for solid oral drug product manufacturing processes, is poorly understood. Thus, the prediction of manufacturability and process performance is difficult. This is further complicated with variability in raw material flow properties. This chapter discusses seven properties typically characterized and the techniques used to measure them. The state of the art in applying this information to define a formulation of manufacturing process is also discussed.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Muzzio FJ et al (2002) Powder technology in the pharmaceutical industry: the need to catch up fast. Powder Technol 124:1–7
Rietema K (1984) Powders, what are they? Powder Technol 37:5–23
Pingali KC et al (2009) Practical methods for improving flow properties of active pharmaceutical ingredients. Drug Dev Ind Pharm 35:1460–1469
Faqih AMN et al (2007) Effect of moisture and magnesium stearate concentration on flow properties of cohesive granular materials. Int J Pharm 336:338–345
Zhou Q et al (2011) Effect of mechanical dry particle coating on the improvement of powder flowability for lactose monohydrate: a model cohesive pharmaceutical powder. Powder Technol 207:414–421
Orband JLR, Geldart D (1995) The use of an antistatic agent to improve powder flowability. Part Part Syst Charact 12:204–206
Pingali KC et al (2009) Use of a static eliminator to improve powder flow. Int J Pharm 369:2–4
Mullarney MP et al (2011) Applying dry powder coatings to pharmaceutical powders using a comil for improving powder flow and bulk density. Powder Technol 212:397–402
Jallo LJ et al (2012) Improvement of flow and bulk density of pharmaceutical powders using surface modification. Int J Pharm 423:213–225
Faqih AN et al (2007) A method for predicting hopper flow characteristics of pharmaceutical powders. Chem Eng Sci 62:1536–1542
Vanarase AU et al (2013) Effects of powder flow properties and shear environment on the performance of continuous mixing of pharmaceutical powders. Powder Technol 246:63–72
Bouffard J et al (2013) Experimental investigation of the effect of particle cohesion on the flow dynamics in a spheronizer. AIChE J 59:1491–1501
He X et al (2013) Assessing powder segregation potential by near infrared (NIR) spectroscopy and correlating segregation tendency to tabletting performance. Powder Technol 236:85–99
Podczeck F (1999) Rheological studies of physical properties of powder used in capsule filling I. Pharm Technol Eur 11:16–24
Podczeck F (1999) Rheological studies of physical properties of powder used in capsule filling II. Pharm Technol Eur 11:34–42
Nase ST et al (2001) Discrete characterization tools for cohesive granular material. Powder Technol 116:214–223
Alexander AW et al (2006) Avalanching flow of cohesive powders. Powder Technol 164:13–21
Faqih A et al (2006) An experimental/computational approach for examining unconfined cohesive powder flow. Int J Pharm 324:116–127
Schulze D (2008) Powders and bulk solids: behavior, characterization, storage and flow. Springer, Berlin
Hancock BC et al (2004) Development of a Robust procedure for assessing powder flow using a commercial avalanche testing instrument. J Pharm Biomed Anal 35:12
Lee YSL et al (2000) Development of a dual approach to assess powder flow from avalanching behavior. AAPS PharmSciTech 1:44
Krantz M et al (2009) Characterization of powder flow: static and dynamic testing. Powder Technol 194:239–245
Vasilenko A et al (2011) Shear and flow behavior of pharmaceutical blends – method comparison study. Powder Technol 208:628–636
Davies CE et al (2004) A new approach to monitoring the movement of particulate material in rotating drums. Dev Chem Eng Miner Process 12:263–275
Jenike AW (1964) Storage and flow of solids. Utah Eng Exp Stat Bull 123:1–194
Carson JW, Wilms H (2006) Development of an international standard for shear testing. Powder Technol 167:1–9
Berry RJ, Bradley MSA (2007) Investigation of the effect of test procedure factors on the failure loci and derived failure functions obtained from annular shear cells. Powder Technol 174:60–63
Pillai JR et al (2007) Comparison between the angles of wall friction measured on an on-line wall friction tester and the Jenike wall friction tester. Powder Technol 174:64–70
Abdullah EC, Geldart D (1999) The use of bulk density measurements as flowability indicators. Powder Technol 102:151–165
Grey RO, Beddow JK (1969) On the Hausner Ratio and its relationship to some properties of metal powders. Powder Technol 2:323–326
Rhodes M (2008) Introduction to particle technology, 2nd edn. John Wiley and Sons, West Sussex
Carr RL (1965) Evaluating flow properties of solids. Chem Eng 72:163–168
Hausner H (1967) Friction conditions in a massive metal powder. Int J Powder Metall 3:7
Freeman R (2007) Measuring the flow properties of consolidated, conditioned and aerated powders — a comparative study using a powder rheometer and a rotational shear cell. Powder Technol 174:25–33
Faqih A et al (2006) Flow - induced dilation of cohesive granular materials. AIChE J 52:4124–4132
Vasilenko A et al (2013) Role of consolidation state in the measurement of bulk density and cohesion. Powder Technol 239:366–373
Trivedi MR, Dave RH (2014) To study physical compatibility between dibasic calcium phosphate and cohesive actives using powder rheometer and thermal methods. Drug Dev Ind Pharm 40:1585
Bemrose CR, Bridgwater J (1987) A review of attrition and attrition test methods. Powder Technol 49:97–126
Lindberg N-O et al (2004) Flowability measurements of pharmaceutical powder mixtures with poor flow using five different techniques. Drug Dev Ind Pharm 30:785–791
Remon JP, Schwartz JB (1987) Effect of raw materials and processing on the quality of granules prepared from microcrystalline cellulose-lactose mixtures. Drug Dev Ind Pharm 13:1–14
Gohel MC, Jogani PD (2003) Exploration of melt granulation technique for the development of coprocessed directly compressible adjuvant containing lactose and microcrystalline cellulose. Pharm Dev Technol 8:175–185
Chevalier E et al (2009) Comparison of low-shear and high-shear granulation processes: effect on implantable calcium phosphate granule properties. Drug Dev Ind Pharm 35:1255–1263
Freeman RE et al (2009) Measuring shear properties and normal stresses generated within a rotational shear cell for consolidated and non-consolidated powders. Powder Technol 190:65–69
Leturia M et al (2014) Characterization of flow properties of cohesive powders: a comparative study of traditional and new testing methods. Powder Technol 253:406–423
Mendez R et al (2012) Effect of feed frame design and operating parameters on powder attrition, particle breakage, and powder properties. Powder Technol 229:253–260
Prestidge CA, Tsatouhas G (2000) Wettability studies of morphine sulfate powders. Int J Pharm 198:201–212
Llusa M et al (2010) Measuring the hydrophobicity of lubricated blends of pharmaceutical excipients. Powder Technol 198:101–107
Washburn EW (1921) The dynamics of capillary flow. Phys Rev 17:273–283
Siebold A et al (1997) Capillary rise for thermodynamic characterization of solid particle surface. J Colloid Interface Sci 186:60–70
Pingali K et al (2011) Evaluation of strain-induced hydrophobicity of pharmaceutical blends and its effect on drug release rate under multiple compression conditions. Drug Dev Ind Pharm 37:428–435
Eilbeck J et al (2000) Effect of contamination of pharmaceutical equipment on powder triboelectrification. Int J Pharm 195:7–11
Harper WR (1951) The Volta effect as a cause of static electrification. Proc Roy Soc Lond Ser A Math Phys Sci 205:83–103
Lowell J, Rose-Innes AC (1980) Contact electrification. Adv Phys 29:947–1023
Jones TB (1995) Electromechanics of particles. Cambridge University Press, Cambridge
Matsusaka S, Masuda H (2003) Electrostatics of particles. Adv Powder Technol 14:143–166
Rowley G (2001) Quantifying electrostatic interactions in pharmaceutical solid systems. Int J Pharm 227:47–55
Jones TB, King JL, Yablonsky JF (1991) Powder handling and electrostatics: understanding and preventing hazards. CRC Press, Boca Raton, FL
Pingali KC et al (2009) An observed correlation between flow and electrical properties of pharmaceutical blends. Powder Technol 192:157–165
Liss ED, Glasser BJ (2001) The influence of clusters on the stress in a sheared granular material. Powder Technol 116:116–132
Alexander A et al (2000) A method to quantitatively describe powder segregation during discharge from vessels. Pharmaceutical Technology Yearbook. Advanstar Communications, Inc., Santa Monica, CA
Prescott JK, Barnum RA (2000) On powder flowability. Pharm Technol 24:60–84
Schulze D (2008) Flow properties of bulk solids. In: Powders and bulk solids. Springer, New York, NY, pp 35–74
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2016 Springer Science+Business Media New York
About this protocol
Cite this protocol
Koynov, S., Muzzio, F.J. (2016). A Quantitative Approach to Understand Raw Material Variability. In: Ierapetritou, M.G., Ramachandran, R. (eds) Process Simulation and Data Modeling in Solid Oral Drug Development and Manufacture. Methods in Pharmacology and Toxicology. Humana, New York, NY. https://doi.org/10.1007/978-1-4939-2996-2_3
Download citation
DOI: https://doi.org/10.1007/978-1-4939-2996-2_3
Publisher Name: Humana, New York, NY
Print ISBN: 978-1-4939-2995-5
Online ISBN: 978-1-4939-2996-2
eBook Packages: Springer Protocols