
Online first articles
Articles not assigned to an issue 23 articles
-

-
Factors Associated with Inclusion of Japan in Phase I Multiregional Clinical Trials in Oncology
Authors
- Akio Maki
- Mamoru Narukawa
- Content type: Original Research
- Published: 23 April 2024

-
Unlocking the Potential: A Systematic Review of Master Protocol in Pediatrics
Authors (first, second and last of 10)
- Yimei Li
- Robert Nelson
- Jingjing Ye
- Content type: Review
- Open Access
- Published: 23 April 2024

-
A Survey to Assess the Current Status of Structured Benefit-Risk Assessment in the Global Drug and Medical Device Industry
Authors (first, second and last of 9)
- Martin Gebel
- Cheryl Renz
- Michael Colopy
- Content type: Original Research
- Published: 22 April 2024
-
Detection Algorithms for Gastrointestinal Perforation Cases in the Medical Information Database Network (MID-NET®) in Japan
Authors (first, second and last of 10)
- Masatoshi Tanigawa
- Mei Kohama
- Hideto Yokoi
- Content type: Original Research
- Published: 21 April 2024

-
Safety of Linagliptin in Patients with Type 2 Diabetes Mellitus: A Systematic Review and Meta-analysis of Randomized Clinical Trials
Authors (first, second and last of 4)
- Hadir Aljohani
- Fares S. Alrubaish
- Fawaz Al-Harbi
- Content type: Review
- Published: 18 April 2024
-
Pathway for Development and Validation of Multi-domain Endpoints for Amyloid Light Chain (AL) Amyloidosis
Authors (first, second and last of 9)
- James Signorovitch
- Jialu Zhang
- Isabelle Lousada
- Content type: Analytical Report
- Open Access
- Published: 17 April 2024
-
Correction: Comparative Assessment of Drug Lag for Approved Oncology Targeted Therapies Between Saudi Arabia, the United States, and the European Union
Authors (first, second and last of 9)
- Mohammed Alnuhait
- Abdullah Alshammari
- Abdullah M. Alrajhi
- Content type: Correction
- Published: 16 April 2024

-
The FDA Reclassification of Cervical Pedicle and Lateral Mass Screws: A Case Study in Regulatory History
Authors (first, second and last of 9)
- Jonathan H. Sussman
- Ahmed Albayar
- William C. Welch
- Content type: Review
- Open Access
- Published: 15 April 2024
-
Improvement of Midpoint Imputation for Estimation of Median Survival Time for Interval-Censored Time-to-Event Data
Authors
- Yuki Nakagawa
- Takashi Sozu
- Content type: Original Research
- Published: 10 April 2024

-
Reflections on the Saudi FDA Regulatory Experience with Smart GxP Inspections
Authors (first, second and last of 10)
- Ali M. Alhomaidan
- Nawaf G. Al-Murikhi
- Mohammed A. Dahhas
- Content type: Original Research
- Published: 10 April 2024
-
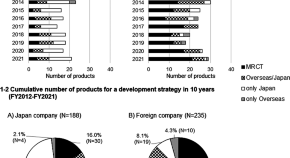
Different Development Strategies Affecting Japan’s Drug lag between Japan-Based and Foreign-Based Companies
Authors
- Masayasu Hidaka
- Hideki Hanaoka
- Yoshiaki Uyama
- Content type: Original Research
- Published: 04 April 2024
-
Benchmarking Site Activation and Patient Enrollment
Authors (first, second and last of 5)
- Mary Jo Lamberti
- Abigail Dirks
- Kenneth Getz
- Content type: Original Research
- Published: 03 April 2024
-
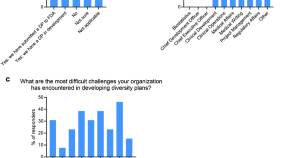
Diversity Plans and Postmarketing Studies: First Impressions of Anticipated Diversity Requirements in the United States
Authors
- Blake Schouest
- Krithi Rao Bindal
- Content type: Original Research
- Published: 03 April 2024
-
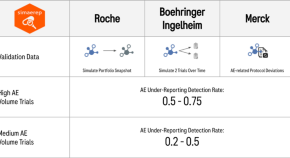
An Open-Source R Package for Detection of Adverse Events Under-Reporting in Clinical Trials: Implementation and Validation by the IMPALA (Inter coMPany quALity Analytics) Consortium
Authors (first, second and last of 10)
- Björn Koneswarakantha
- Ronojit Adyanthaya
- On behalf of the IMPALA (Inter coMPany quALity Analytics) Consortium
- Content type: Analytical Report
- Open Access
- Published: 02 April 2024
-
How Aggregate Safety Assessment Planning Supports Investigational New Drug Safety Reporting Decisions
Authors (first, second and last of 4)
- Barbara A. Hendrickson
- Cynthia McShea
- Susan Talbot
- Content type: Analytical Report
- Published: 30 March 2024

-
Human Medicines Recall Announcements in Saudi Arabia Between 2017 and 2022: An Analysis of Saudi Food and Drug Authority (SFDA) Reports
Authors (first, second and last of 5)
- Malak Almutairi
- Aljoharah Algabbani
- Amani S. Alqahtani
- Content type: Original Research
- Published: 28 March 2024
-
Adoption of Decentralization: Are Our Perceptions Holding Us Back?
Authors (first, second and last of 4)
- Lindsay Kehoe
- Sara Bristol Calvert
- Morgan Hanger
- Content type: Commentary
- Open Access
- Published: 28 March 2024
-
Comparative Assessment of Drug Lag for Approved Oncology Targeted Therapies Between Saudi Arabia, the United States, and the European Union
Authors (first, second and last of 9)
- Mohammed Alnuhait
- Abdullah Alshammari
- Abdullah M. Alrajhi
- Content type: Original Research
- Published: 27 March 2024

-
International Comparison of Qualification Process for Medical Product Development Tools
Authors
- Daichi Uchijima
- Shingo Kano
- Content type: Original Research
- Open Access
- Published: 27 March 2024
-
Recent Status of Phase I Clinical Trials for Brain Tumors: A Regulatory Science Study of Exploratory Efficacy Endpoints
Authors (first, second and last of 10)
- Shinya Watanabe
- Takahiro Nonaka
- Eiichi Ishikawa
- Content type: Original Research
- Open Access
- Published: 26 March 2024

-
New Benchmarks on Protocol Amendment Experience in Oncology Clinical Trials
Authors
- Emily Botto
- Zachary Smith
- Kenneth Getz
- Content type: Original Research
- Published: 26 March 2024
-
Decentralized Clinical Trials in the Development of Drugs and Biological Products
Authors
- Ryan Robinson
- Leonard Sacks
- Content type: Commentary
- Open Access
- Published: 02 February 2024
This is part of 1 collection:
For authors
Submit manuscriptWorking on a manuscript?
Avoid the most common mistakes and prepare your manuscript for journal editors.
Learn more

