Abstract
Background
Nasopharyngeal carcinoma (NPC) is a geographically and racially variable disease that has a high incidence in Southeast China. According to previous studies on tumor immunity, we compared multiple clinical parameters and blood indexes with outcomes regarding to Epstein-Barr virus (EBV) status in NPC patients.
Methods
According to the EBV load at diagnosis, 220 NPC patients who received concurrent chemoradiotherapy (CRT) were divided into two groups: EBV DNA ≥ 1500 copies/mL and EBV DNA < 1500 copies/mL, respectively. We compared clinical parameters with peripheral blood mononuclear cells, lymphocyte subsets and biochemical indexes. We also analyzed distant metastases and the overall survival rate regarding to these characteristics.
Results
In most cases, the two groups showed the same trends. Most blood indexes were decreased during CRT and the decrease of the absolute count was more significant than the percentage. Patients with younger age showed the higher CD3+ and CD3 + CD8+ percentages. Patients whose EBV DNA ≥ 1500 copies/mL showed a higher N classification than those with EBV DNA < 1500 copies/mL at first diagnosis. Within patients with EBV DNA ≥ 1500 copies/mL, a higher CD3 + CD8+ percentage or lower CD3-CD56+ percentage had better OS rates, and the CD3 + CD8+ percentage was an independent prognostic factor by multivariate survival analyses.
Conclusions
CRT caused an overall decrease of blood cells in NPC patients. Among all the blood indexes, the CD3 + CD8+ percentage showed a correlation with age and was an independent prognostic factor in patients with EBV DNA ≥ 1500 copies/mL at first diagnosis, which is worthy for further large cohort study.
Similar content being viewed by others
Background
Nasopharyngeal carcinoma (NPC) is one of malignant epithelial cell tumors at the top and lateral wall of the nasal cavity with different etiology and pathology [1]. According to the World Health Organization (WHO) classification, NPC is divided into keratinizing (K) carcinoma (squamous cell carcinoma; SCC), non-keratinizing (NK) carcinoma (including differentiated and undifferentiated variants) and basaloid squamous cell carcinoma. There are great differences in the geographical and ethnic distributions of NPC. The incidence of NPC is significantly higher in South China and Southeast Asia, mainly belonging to NK-NPC (90%). The population is sensitive to radiotherapy and the disease has almost 100% association with Epstein–Barr virus (EBV) [2]. EBV is a common linear double-stranded virus, about 122–180 nm, which contains a double helix DNA wrapped in the protein capsid. EBV infection has been found mainly in B lymphocytes and epithelial cells which can be cleaved and replicated [3]. The current research on other pathogenic factors of NPC is not thorough. Genetic susceptibility and consumption of pickled food are also important pathogenic factors [2]. Previous studies have demonstrated that high EBV DNA levels may indicate poor prognosis and reduced long-term survival in NPC patients [4, 5]. However, tumor immunity at the circulating level in relation to NPC patient outcome remains to be explored.
Radiotherapy is the main treatment for NPC [6]. Radiotherapy alone is effective in the early stage, concurrent chemoradiotherapy (CRT) in the locally advanced stage, and systematic therapy in patients with metastases and recurrence [7]. In recent years, with the progress of molecular biology, targeted therapy and immunotherapy have become new trends, and have determined their positions as second-line treatments for recurrent/metastatic NPC. Moreover, the exploration of first-line combined chemotherapy also shows a good prospect [8, 9].
Immune cell infiltration is a characteristic of almost all malignant tumors. Immune cells mainly include tumor-associated macrophages, lymphocytes, and mast cells. They play important roles in immune monitoring and contribute to the elimination of tumor cells [10]. Given the minimally invasive nature of blood sample collection compared to tumor infiltrating tissues, the regularity of various indexes in peripheral blood is more convenient for clinical application, such as T lymphocyte cells, B lymphocyte cells and NK cells. The inflammation index is the characteristic marker of tumor and plays an auxiliary role in determining the occurrence and progression of tumor [11]. Peripheral blood Neutrophil count/Lymphocyte count (NLR), Lymphocyte count/Monocyte count (LMR), Platelet count/Lymphocyte count (PLR), Platelet count×Neutrophil count/Lymphocyte count (SII) are widely used indexes in the evaluation of inflammation [12]. In addition, albumin (ALB) and lactate dehydrogenase (LDH) are well-studied prognostic biomarkers of NPC [13].
This study was based on the status of EBV load in patients with NPC. We explored not only the relationship between EBV DNA load, lymphocyte subsets and inflammatory indexes in peripheral blood, but also the relationships between EBV DNA load, clinical parameters, distant metastases and OS. The aim was to investigate indicators with prognostic value in NPC patients, and these findings may hence significantly aid the clinical decision-making process.
Materials and methods
Ethics statement and the criteria for patient inclusion
A retrospective single-institution cohort design was used, and a total of 220 patients with NPC who signed an informed consent form and have completed treatment were enrolled in this study. Patients with a history of other malignant tumors were excluded. This study was approved by the local Ethics Committee of the Zhejiang Cancer Hospital (IRB-2021-326). Grading and staging were undertaken according to the 8th edition of the International Union for Cancer Control/American Joint Committee on Cancer (UICC/AJCC) cancer staging system for NPC. Patients who survived were censored at their last date of follow-up. The OS rate was calculated from the date of diagnosis to the date of death.
Therapy regimen
None of the patients underwent surgery. The tissue sent for pathological diagnosis was obtained by nasopharyngeal biopsy. All patients underwent concurrent CRT. Chemotherapy regimen: Nedaplatin plus Docetaxel; Chemotherapy interval: once every 21 days. Radiotherapy dose: 2.0 Gy. Radiotherapy interval: once every 5 days, suspended for 2 days. Circulating blood samples were collected before CRT. The first time was before therapy, named T1; the second time was during therapy, named T2; the third time was before the last therapy, named T3.
Blood samples
Circulating peripheral blood mononuclear cells (PBMCs) were obtained during the period between January 2016 and December 2020. Two milliliters of venous blood was collected in a vacuum tube prefilled with EDTA-K2 (Zhejiang Gongdong Medical Technology Co., Ltd. China) for EBV DNA detection, white blood cell (WBC) count, neutrophil count, total lymphocyte count and platelet count. Two milliliters of venous blood was collected in a vacuum tube prefilled with heparin sodium (Kangshi Medical Inc., China) for lymphocyte subset detection. Two milliliters of venous blood was collected in a vacuum tube prefilled with coagulant (Zhejiang Gongdong Medical Technology Co., Ltd. China) for ALB and LDH. After centrifugation in Biocoll Separating Solution (Merck, Germany), PBMCs were separated and recovered, washed twice in PBS and stored at 4 °C. Lymphocyte subset detection was performed within 24 h. The patient characteristics are summarized in Table 1.
EBV DNA and different blood indexes detection
EBV DNA was analyzed by Roche Lightcycler 480 fluorescence quantitative PCR (Roche, USA) with Daan EBV DNA detection reagent (Daan Gene Co., Ltd. China). WBC count, neutrophil count, total lymphocyte count and platelet count were analyzed by a Mindray CAL 8000 automatic blood cell analysis pipeline (Mindray, China) immediately after blood collection. The vacuum tube prefilled with coagulant was centrifuged at 3500 r/min for 5 min, and serum was detected by a Hitachi 7600 automatic biochemical analyzer (HITACHI, Japan). For flow cytometry analysis, PBMCs in 50 μL PBS supplemented with 0.5% BSA (Thermo Fisher, USA) (1 × 106 cells/mL) were incubated with anti-human monoclonal antibodies (mAbs) for 15 min. The following mAbs were used for flow cytometry: anti-CD45/CD4/CD8/CD3, anti-CD45/CD56/CD19/CD3, anti-CD4, anti-CD45RA, anti-CD45RO, anti-CD8 and anti-CD38 (Beckman Coulter, USA) (Supplementary Table 1). OptiLyse C lysing solution (Beckman Coulter, USA) was used for hemolysis for 15 min, and PBMCs were resuspended in PBS (Beckman Coulter, USA). The results were acquired by a Beckman Coulter FC500 flow cytometer and analyzed by CXP analysis software with the recommended reference ranges [12, 14, 15]. The reference ranges were adjusted in our laboratory (Supplementary Table 2).
We divided the patients into two groups: a low-risk group (EBV DNA < 1500 copies/mL at T1) and a high-risk group (EBV DNA ≥ 1500 copies/mL at T1) by a previously defined EBV-DNA cutoff value [16, 17]. We mainly compared the differences between the two groups in the study.
Statistical analysis
Statistical analyses were performed using STATA 14.2 (Stata Corp LLC, Texas, USA). Categorical variables are described as percentages, and numerical variables are represented as the mean ± SD. Qualitative data were compared using the Chi-square test. Univariate and multivariate analyses were performed by the cox proportional hazards model. Survival was determined using the Kaplan–Meier (KM) method. A p value of < 0.05 was regarded as statistically significant.
Results
Patient characteristics
A total of 220 patients were included in this study, with a mean age of 52.31 ± 11.10 (ranging from 17 to 79). There were 166 males and 54 females in the study. It takes an average of 3.6 months from the onset of physical discomfort to diagnosis. The clinicopathological parameters of the patients are summarized in Table 1. Most patient tumors were staged as advanced (UICC TNM stages III/IV, 75/131; 93.64%). The median follow-up time was 41.40 ± 16.24 months (range 10–84 months). Some of the patients had a habit of smoking and drinking, and have family history of cancer.
We divided the patients into two groups by EBV DNA load, 149 cases belonged to low-risk group and 71 cases belonged to high-risk group. The two groups showed different in N classification (p < 0.001), and the high-risk group showed a higher N classification. The data are shown in Table 1.
Distant metastases were found in 44 patients (20.00%). Among all metastases, 17 were bone, 9 were lung, 6 were liver, 4 were brain, 2 were stomach, 2 were mouth, 1 was uterus and 3 were multiple metastases. All patients with metastatic disease had TNM stage IV disease.
The relationships between clinical parameters and blood indexes
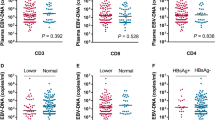
We measured EBV DNA load, peripheral blood indexes such as WBC count, neutrophil count, total lymphocyte count, platelet count; lymphocyte subsets such as CD3+ percentage/count, CD3 + CD4+ percentage/count, CD3 + CD8+ percentage/count; biochemical indexes ALB and LDH. The flow cytometry results are shown in Fig. 1A-J.
Represented images of flow cytometry results and Kaplan-Meier analysis. A. Lymphocyte percentage; B. CD3+ percentage; C. CD3 + CD4+ percentage; D. CD3 + CD8+ percentage; E. CD3 + CD56+ percentage and CD3-CD56+ percentage; F. CD3-CD19+ percentage; G. CD4 + CD45RA+ percentage and CD4 + CD45RA- percentage; H. CD4 + CD45RO+ percentage; I. CD45RA + CD45RO+ percentage; J. CD8 + CD38+ percentage; K. Kaplan-Meier analysis of CD3 + CD8+ percentage in high-risk group; L: Kaplan-Meier analysis of CD3-CD56+ percentage in high-risk group
As shown in Tables 2–3, we studied the relationships between clinical parameters such as age, sex, smoking history, drinking history, family history of cancer, pathological type, clinical stage and the changes of blood indexes in each group. The calculations were carried out at T1.
Among the clinical parameters, age was more correlated with lymphocyte subsets. The significant differences throughout CRT were CD3+ percentage (the younger age, the higher CD3+ percentage), and CD3 + CD8+ percentage (the younger age, the higher CD3 + CD8+ percentage). The other clinical parameters and blood indexes didn’t always show the same rule throughout the therapy. For example, in the high-risk group, patients with higher N classification showed higher WBC count and neutrophil count. In low-risk group, patients with higher N classification showed higher CD4 + CD45RA- count and CD4 + CD45RO+ count.
The relationships between EBV DNA and blood indexes
The relationships between EBV DNA load and lymphocyte subsets at T1 were as follows: a higher EBV DNA load was correlated with higher CD4 + CD45RA- percentage (p = 0.015), CD4 + CD45RO+ percentage (p = 0.019) and lower CD4 + CD45RA+/CD4 + CD45RA- ratio (p = 0.034) (Supplementary Table 3). Additionally, patients with a higher EBV load (high-risk group) had higher NLR (p = 0.009), PLR (p = 0.021) and SII (p = 0.004) compared to the low-risk group. Furthermore, the high-risk group demonstrated higher plasma LDH levels compared to the low-risk group (p < 0.001) (Supplementary Table 3). At T2 and T3, these correlations were not observed. Data were presented as the mean value and were shown in Supplementary Tables 3, 4, 5 and 6.
Expression of blood indexes over time
When we compared the high-risk group and the low-risk group, we found that the two groups were consistent during CRT. The decrease in absolute count was more significant than that in percentage, and all lymphocyte subset counts were decreased at the end of therapy. Additionally, the CD4/CD8 ratio and the CD4 + CD45RA+/CD4 + CD45RA- ratios were significantly decreased during therapy. But NLR (p < 0.001), PLR (p < 0.001) and SII (p < 0.001) showed different patterns with that ratios increased over time. These changes were more significant in the low-risk group. Data were shown in Supplementary Tables 3, 4, 5 and 6.
Expression of blood indexes in patients with or without distant metastases
In 44 patients with distant metastases, 14 belonged to the high-risk group and 30 belonged to the low-risk group. In the high-risk group, monocyte count showed lower in patients with distant metastases at T1. In low-risk group, the expression of CD3+ percentage and PLR were higher while CD3-CD56+ count and CD8 + CD38+ count were lower in patients with distant metastases at T1. The data were shown in Supplementary Tables 7 and 8.
Overall survival
In the high-risk group, after a median follow-up of 39.24 months, the OS rate was 83.10%. In the low-risk group, after a median follow-up of 42.44 months, the OS rate was 87.92%. EBV DNA load was not an independent prognostic factor for NPC in our analysis (data not shown).
We explored the role of clinical parameters and blood indexes in predicting survival before therapy. In the high-risk group, in univariate survival analyses, we found that age (HR = 1.059, 95% CI: 1.000–1.120, p = 0.049), CD3+ percentage (HR = 0.951, 95% CI: 0.915–0.988, p = 0.010), CD3 + CD8+ percentage (HR = 0.902, 95% CI: 0.818–0.995, p = 0.039) and CD3-CD56+ percentage (HR = 1.045, 95% CI: 1.010–1.081, p = 0.011) were significant factors related to survival. In multivariate survival analyses, CD3 + CD8+ percentage (HR = 0.917, 95% CI: 0.810–1.038, p = 0.034) was the only factor associated with survival. The data were shown in Table 4. In the low-risk group, in univariate survival analyses, we found that age (HR = 1.078, 95% CI: 1.022–1.136, p = 0.005), CD4/CD8 ratio (HR = 1.834, 95% CI: 1.054–3.192, p = 0.032), CD4 + CD45RA- % (HR = 1.083, 95% CI: 1.010–1.160, p = 0.025), CD4 + CD45RO+ % (HR = 1.090, 95% CI: 1.015–1.171, p = 0.018) and Platelet count (HR = 1.003, 95% CI: 1.000–1.007, p = 0.042) were significant factors related to survival. In multivariate survival analyses, age (HR = 1.089, 95% CI: 1.026–1.156, p = 0.005) and Platelet count (HR = 1.004, 95% CI: 1.000–1.008, p = 0.030) were significant factors associated with survival. The data were shown in Supplementary Table 9. The EBV status may contribute to the differences of independent factors between the two groups. In addition, our analysis did not show the significance of TNM staging to predict survival. One reason is that the study population consisted of mainly stage III and IV patients in our study. A future study including more stage I and II patients is warranted.
There were differences between the two groups in OS rates before the last therapy. The high-risk group showed a higher CD3 + CD8+ percentage or a lower CD3-CD56+ percentage had better OS rates. The median survival times for the high CD3 + CD8+ percentage group (average 38.50 months) and the normal CD3 + CD8+ percentage group (average 39.53 months) were significantly longer than that for the low CD3 + CD8+ percentage group (average 25.00 months) (p = 0.003). The median survival times for the high CD3-CD56+ percentage group (average 37.60 months) and normal CD3-CD56+ percentage group (average 40.14 months) were significantly longer than those for the low CD3-CD56+ percentage group (average 32.40 months) (p = 0.003). Kaplan-Meier curves of OS for lymphocyte subsets at T3 were plotted and shown in Fig. 1K-L. The prognostic values for other factors were not significant.
Discussion
With the improved understanding of the pathogenesis of EBV associated NPC and plasma EBV DNA test for population screening as well as personalized therapy strategies, the mortality rate of NPC has been greatly reduced. However, the early diagnosis of NPC remains a challenge. Therefore, exploring therapeutic and prognostic biomarkers for NPC is necessary. In this study, NPC patients were divided into a high-risk group and a low-risk group according to the EBV DNA load before the first CRT. By comparing clinicopathological data and peripheral blood indexes between the high-risk group and the low-risk group, we hope to find potential biomarkers that can help to predict prognosis and guide NPC therapy.
In our study, the initial EBV DNA load was related to N classification that is patients with a higher initial EBV load had a higher N classification. This finding was consistent to previous studies [4, 18]. In addition, we found that EBV DNA load was associated with a variety of lymphocyte subsets, especially CD3+ percentage and CD3 + CD8+ percentage. Similar to our results, Mo et al [19] reported the relationship between EBV DNA load and CD4 + CD25+ T cells and CD8+ cells in patients with NPC. A higher CD8+ percentage tends to have a better prognosis, which may due to the fact that CD8+ cells play an important role in establishing effective immune surveillance.
Previous studies have shown that EBV DNA and LDH are independent prognostic factors for locoregionally advanced NPC [16, 17]. The circulating levels of EBV DNA and LDH in combination with other risk factors can predict recurrence and overall survival for NPC patients in established statistic nomogram models [13, 20]. In our analysis, patients with higher initial EBV load referring to high-risk group had a higher LDH level compared to the low-risk group. However, multivariate survival analyses of overall survival combining circulating lymphocyte subsets did not show the significant prognostic value of LDH in high-risk patients. Thus, additional studies are warranted to explore the role of circulating level of LDH when NPC patients are stratified by plasma EBV DNA load, ideally from large prospective studies.
During CRT, we also found that lymphocyte subset counts were decreased before last therapy compared to initial time point before therapy in both high-risk group and low-risk group. This phenomenon has been observed during CRT in other tumors. For example, Lee et al reported pancytopenia after CRT in rectal cancer [21]. It may relate to myelosuppression [22]. Schuler et al [23] reported that the number of CD4+ T cells declined after CRT in patients with head and neck cancer, because CD4+ T cells were sensitive to CRT.
Inflammation plays a crucial role in cancer development [24]. Peripheral blood neutrophils and lymphocytes not only constitute the predominant proportion, but also play an important role in the immune response. The blood neutrophil-to-lymphocyte ratio has been found to be associated with disease progression in a number of malignancies [25]. During CRT, we found NLR, PLR and SII were increased, mainly because lymphocytes were more sensitive to CRT and the decrease of lymphocytes was greater than that in other white blood cells [24].
Cancer metastases are the main cause of cancer mortality. One important question regarding which patients are likely to develop distant metastases remains to be answered. We found differences of lymphocyte subsets before therapy between the high-risk group and the low-risk group when comparing patients who developed metastases or not. For example, patients who developed metastases in the high-risk group had lower monocyte counts while CD3+ percentage, CD3-CD56+ count and CD8 + CD38+ count were decreased in the low-risk group. These differences changed after therapy. EBV may attack immune cells and interfere with the immune system cell function [26]. EBV induces the apoptosis of monocyte precursors and therefore inhibit the development of dendritic cells [27]. Our findings warrant further investigation.
CD8+ lymphocytes have been shown to play an important role in the host’s defense against malignancies, and a high infiltration of CD8+ lymphocytes was associated with good clinical outcome in many cancers [28,29,30]. Consistent to these findings, in the high-risk group, patients with a higher CD3 + CD8+ percentage had better OS rates and the CD3 + CD8+ percentage was an independent prognostic factor. Moreover, after treatment, patients in the high-risk group at T3 with a higher CD3-CD56+ percentage referring to the natural killer cell percentage, had poor survival compared to the low and normal CD3-CD56+ percentages. These findings suggest that the change of circulating lymphocyte subsets during treatment has the potential to predict the patient’s outcome.
In conclusion, we demonstrated that the CRT treatment caused overall decreases of blood cells in NPC patients, especially lymphocytes. We also demonstrated that patients with EBV DNA ≥ 1500 copies/mL had a higher CD3 + CD8+ percentage or a lower CD3-CD56+ percentage had better OS rates and the CD3 + CD8+ percentage was an independent prognostic factor by multivariate survival analyses. With the introduction of tumor immunotherapy as a promising therapeutic approach in NPC, our findings of the changes of circulating lymphocyte subsets during treatment may add to the current understanding of EBV-associated NPC immunity and advance the management of NPC patients.
Availability of data and materials
The datasets generated and/or analyzed during the present study are available from the corresponding author upon reasonable request.
References
Lee HM, Okuda KS, González FE, Patel V. Current perspectives on nasopharyngeal carcinoma. Adv Exp Med Biol. 2019;1164:11–34. https://doi.org/10.1007/978-3-030-22254-3_2. PMID: 31576537.
Petersson F. Nasopharyngeal carcinoma: a review. Semin Diagn Pathol. 2015;32(1):54–73. https://doi.org/10.1053/j.semdp.2015.02.021. Epub 2015 Feb 25. PMID: 25769204.
Kanda T, Yajima M, Ikuta K. Epstein-Barr virus strain variation and cancer. Cancer Sci. 2019;110(4):1132–9. https://doi.org/10.1111/cas.13954. Epub 2019 Feb 21. PMID: 30697862; PMCID: PMC6447851.
Chen YP, Chan ATC, Le QT, Blanchard P, Sun Y, Ma J. Nasopharyngeal carcinoma. Lancet. 2019;394(10192):64–80. https://doi.org/10.1016/S0140-6736(19)30956-0. Epub 2019 Jun 6. PMID: 31178151.
Liu TB, Zheng ZH, Pan J, Pan LL, Chen LH. Prognostic role of plasma Epstein-Barr virus DNA load for nasopharyngeal carcinoma: a meta-analysis. Clin Invest Med. 2017;40(1):E1–E12. https://doi.org/10.25011/cim.v40i1.28049. PMID: 28218577.
Sun XS, Li XY, Chen QY, Tang LQ, Mai HQ. Future of Radiotherapy in Nasopharyngeal Carcinoma. Br J Radiol. 2019;92(1102):20190209. https://doi.org/10.1259/bjr.20190209. Epub 2019 Jul 9. PMID: 31265322; PMCID: PMC6774595.
Lee AWM, Ngan RKC, Ng WT, Tung SY, Cheng AAC, Kwong DLW, et al. NPC-0501 trial on the value of changing chemoradiotherapy sequence, replacing 5-fluorouracil with capecitabine, and altering fractionation for patients with advanced nasopharyngeal carcinoma. Cancer. 2020;126(16):3674–88. https://doi.org/10.1002/cncr.32972. Epub 2020 Jun 4. PMID: 32497261.
Kang Y, He W, Ren C, Qiao J, Guo Q, Hu J, et al. Advances in targeted therapy mainly based on signal pathways for nasopharyngeal carcinoma. Signal Transduct Target Ther. 2020;5(1):245. https://doi.org/10.1038/s41392-020-00340-2. Erratum in: Signal Transduct Target Ther 2020 Nov 13;5(1):265. PMID: 33093441; PMCID: PMC7582884.
Hong M, Tang K, Qian J, Deng H, Zeng M, Zheng S, et al. Immunotherapy for EBV-associated nasopharyngeal carcinoma. Crit Rev Oncog. 2018;23(3–4):219–34. https://doi.org/10.1615/CritRevOncog.2018027528. PMID: 30311576.
Picard E, Verschoor CP, Ma GW, Pawelec G. Relationships between immune landscapes, genetic subtypes and responses to immunotherapy in colorectal Cancer. Front Immunol. 2020;6(11):369. https://doi.org/10.3389/fimmu.2020.00369. PMID: 32210966; PMCID: PMC7068608.
Tomita M, Maeda R, Ayabe T, Nakamura K. Prognostic impact of a novel tumor marker and inflammation index for patients with non-small-cell lung Cancer. Anticancer Res. 2020;40(7):4023–7. https://doi.org/10.21873/anticanres.14397. PMID: 32620647.
Meng X, Chang Q, Liu Y, Chen L, Wei G, Yang J, et al. Determinant roles of gender and age on SII, PLR, NLR, LMR and MLR and their reference intervals defining in Henan, China: A posteriori and big-data-based. J Clin Lab Anal. 2018;32(2):e22228. https://doi.org/10.1002/jcla.22228. Epub 2017 Apr 5. PMID: 28378887; PMCID: PMC6817185.
Zhang LL, Xu F, Song D, Huang MY, Huang YS, Deng QL, et al. Development of a nomogram model for treatment of nonmetastatic nasopharyngeal carcinoma. JAMA Netw Open. 2020;3(12):e2029882. https://doi.org/10.1001/jamanetworkopen.2020.29882. PMID: 33306119; PMCID: PMC7733160.
Qin L, Jing X, Qiu Z, Cao W, Jiao Y, Routy JP, et al. Aging of immune system: immune signature from peripheral blood lymphocyte subsets in 1068 healthy adults. Aging (Albany NY). 2016;8:848–59.
Luo H, He L, Zhang G, Yu J, Chen Y, Yin H, et al. Normal reference intervals of neutrophil-to-lymphocyte ratio, platelet-to-lymphocyte ratio, lymphocyte-to-monocyte ratio, and systemic immune inflammation index in healthy adults: a large multi-center study from Western China. Clin Lab. 2019;65(3). https://doi.org/10.7754/Clin.Lab.2018.180715. PMID: 30868857.
Liu SL, Sun XS, Xie HJ, Chen QY, Lin HX, Liang H, et al. Comparing three induction chemotherapy regimens for patients with locoregionally advanced nasopharyngeal carcinoma based on TNM stage and plasma Epstein-Barr virus DNA level. BMC Cancer. 2020;20(1):89. https://doi.org/10.1186/s12885-020-6555-7. PMID: 32013967; PMCID: PMC6998839.
Jin YN, Yao JJ, Zhang F, Wang SY, Zhang WJ, Zhou GQ, et al. Is pretreatment Epstein-Barr virus DNA still associated with 6-year survival outcomes in locoregionally advanced nasopharyngeal carcinoma? J Cancer. 2017;8(6):976–82. https://doi.org/10.7150/jca.18124. PMID: 28529609; PMCID: PMC5436249.
Liu LT, Chua MLK, Tao Y, Tang LQ, Mai HQ. Optimal sequencing of chemotherapy with chemoradiotherapy based on TNM stage classification and EBV DNA in locoregionally advanced nasopharyngeal carcinoma. Cancer Commun (Lond). 2019;39(1):64. https://doi.org/10.1186/s40880-019-0398-0. PMID: 31653270; PMCID: PMC6814970.
Mo WN, Tang AZ, Zhou L, Huang GW, Wang Z, Zeng Y. Analysis of Epstein-Barr viral DNA load, EBV-LMP2 specific cytotoxic T-lymphocytes and levels of CD4+CD25+ T cells in patients with nasopharyngeal carcinomas positive for IgA antibody to EBV viral capsid antigen. Chin Med J. 2009;122(10):1173–8. PMID: 19493466.
Tang LQ, Li CF, Li J, Chen WH, Chen QY, Yuan LX, et al. Establishment and Validation of Prognostic Nomograms for Endemic Nasopharyngeal Carcinoma. J Natl Cancer Inst. 2015;108(1):djv291. https://doi.org/10.1093/jnci/djv291. PMID: 26467665.
Lee YJ, Lee SB, Beak SK, Han YD, Cho MS, Hur H, et al. Temporal changes in immune cell composition and cytokines in response to chemoradiation in rectal cancer. Sci Rep. 2018;8(1):7565. https://doi.org/10.1038/s41598-018-25970-z. PMID: 29765096; PMCID: PMC5953940.
Yang TJ, Oh JH, Apte A, Son CH, Deasy JO, Goodman KA. Clinical and dosimetric predictors of acute hematologic toxicity in rectal cancer patients undergoing chemoradiotherapy. Radiother Oncol. 2014;113(1):29–34. https://doi.org/10.1016/j.radonc.2014.09.002. Epub 2014 Oct 7. PMID: 25304718; PMCID: PMC4822505.
Schuler PJ, Harasymczuk M, Schilling B, Saze Z, Strauss L, Lang S, et al. Effects of adjuvant chemoradiotherapy on the frequency and function of regulatory T cells in patients with head and neck cancer. Clin Cancer Res. 2013;19:6585–96.
Singh N, Baby D, Rajguru JP, Patil PB, Thakkannavar SS, Pujari VB. Inflammation and cancer. Ann Afr Med. 2019;18(3):121–6. https://doi.org/10.4103/aam.aam_56_18. PMID: 31417011; PMCID: PMC6704802.
Roxburgh C, Mcmillan D. Role of systemic inflammatory response in predicting survival in patients with primary operable cancer. Future Oncol. 2010;6:149–63.
Thorley-Lawson DA. EBV Persistence--Introducing the Virus. Curr Top Microbiol Immunol. 2015;390(Pt 1):151–209. https://doi.org/10.1007/978-3-319-22822-8_8. PMID: 26424647; PMCID: PMC5125397.
Li L, Liu D, Hutt-Fletcher L, Morgan A, Masucci MG, Levitsky V. Epstein-Barr virus inhibits the development of dendritic cells by promoting apoptosis of their monocyte precursors in the presence of granulocyte macrophage-colony-stimulating factor and interleukin-4. Blood. 2002;99(10):3725–34. https://doi.org/10.1182/blood.v99.10.3725. PMID: 11986229.
Wang YQ, Chen L, Mao YP, Li YQ, Jiang W, Xu SY, et al. Prognostic value of immune score in nasopharyngeal carcinoma using digital pathology. J Immunother Cancer. 2020;8(2):e000334. https://doi.org/10.1136/jitc-2019-000334. PMID: 32690665; PMCID: PMC7371227.
Sangro B, Melero I, Wadhawan S, Finn RS, Abou-Alfa GK, Cheng AL, et al. Association of inflammatory biomarkers with clinical outcomes in nivolumab-treated patients with advanced hepatocellular carcinoma. J Hepatol. 2020;73(6):1460–9. https://doi.org/10.1016/j.jhep.2020.07.026. Epub 2020 Jul 22. PMID: 32710922; PMCID: PMC7751218.
Lu J, Chen XM, Huang HR, Zhao FP, Wang F, Liu X, et al. Detailed analysis of inflammatory cell infiltration and the prognostic impact on nasopharyngeal carcinoma. Head Neck. 2018;40(6):1245–53. https://doi.org/10.1002/hed.25104. Epub 2018 Mar 1. PMID: 29493822.
Funding
This study was supported by Guangji Talents Foundation Award (E) and Internal Talent Program (under Forty) of Zhejiang Cancer Hospital. The funder played no role in the design of the study and collection, analysis, and interpretation of data and in writing the manuscript.
Author information
Authors and Affiliations
Contributions
J.Z. designed the experiments and wrote the paper. R.F. performed the experiments. Z.P. analyzed the data. X.Q. conceived the study and revised the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
The study was conducted according to the guidelines of the Declaration of Helsinki and approved by the local Ethics Committee of the Zhejiang Cancer Hospital (IRB-2021-326). Informed consent was obtained from all subjects involved in the study.
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/. The Creative Commons Public Domain Dedication waiver (http://creativecommons.org/publicdomain/zero/1.0/) applies to the data made available in this article, unless otherwise stated in a credit line to the data.
About this article
Cite this article
Zhu, J., Fang, R., Pan, Z. et al. Circulating lymphocyte subsets are prognostic factors in patients with nasopharyngeal carcinoma. BMC Cancer 22, 716 (2022). https://doi.org/10.1186/s12885-022-09438-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s12885-022-09438-y