Abstract
Green synthesis is one of the rapid, reliable, and best routes for the synthesis of silver nanoparticles (Ag NPs). The current study revealed that the aqueous leaf extract of Pterocarpus santalinus, which contains steroids, saponins, tannins, phenols, triterpenoids, flavonoids, glycosides, and glycerides, is found to be responsible for bioreduction during the synthesis of spherical Ag nanoparticles. The formed Ag NPs were characterized by ultraviolet-visible (UV-vis), Fourier transform-infrared (FT-IR), X-ray diffraction (XRD), atomic force microscopy (AFM), energy-dispersive X-ray spectroscopy (EDX), and scanning electron microscopy (SEM) analysis. UV-vis spectra of the aqueous medium containing silver nanoparticles showed a surface plasmon resonance peak at 418 nm. FT-IR analysis was performed to analyze the biomolecules responsible for the reduction of Ag NPs. XRD results confirmed the presence of silver nanoparticles with face-centered cubic structure. The EDX analysis showed the completed inorganic composition of the synthesized Ag NPs. AFM analysis exemplified the results of particle sizes (41 nm). The calculated crystallite sizes are in the range of 20 to 50 nm, and the spherical nature of the Ag NPs was ascertained by SEM. The synthesized Ag NPs exhibited good antibacterial potential against gram-positive and gram-negative bacterial strains. The zone of inhibition effect of antibacterial activity depends upon the concentration of Ag NPs.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Background
The application of nanoscale materials and structures, usually ranging from 1 to 100 nm, is an emerging area of nanoscience and nanotechnology. Silver nanoparticles (Ag NPs) have rapidly increased due to their unusual optical, chemical, electronic, photo-electrochemical, catalytic, magnetic, antibacterial, and biological labeling properties [1]. Silver nanoparticles were used in broad range of applications like biomedical [2], drug delivery [3], food industries [4], agriculture [5], textile industries [6], water treatment [7] as an antioxidant [8], antimicrobial [9], anti-cancer [10], cosmetics [11], ointments [12], and larvicides [13]. Nanoparticle synthesis is usually carried out by various physical and chemical methods using various hazardous and toxic chemicals. However, green synthesis approaches of producing Ag NPs are an alternative source of conventional method and possess excellent antimicrobial activity [1]. Recently, the green synthesis of Ag NPs has been reported using the extract of plants such as Artocarpus heterophyllus[14], Sesbania grandiflora[15], Punica granatum[16], Pithecellobium dulce[17], Malva parviflora[18], Iresine herbstii[19], Hibiscus cannabinus[20], Hevea brasiliensis[21], Euphorbia prostrata[22]Cissus quadrangularis[23–25], Catharanthus roseus[26], Coccinia grandis[27], Ixora coccinea[28], Lippia citriodora[29], Manilkara zapota[30], Piper pedicellatum[31], Prosopis juliflora[32], and Semecarpus anacardium[33].
Pterocarpus santalinus belongs to Fabaceae family, and it has been used for treatment of vomiting, treating eye diseases, mental aberrations, and ulcers. In addition, it has shown to have antipyretic, anti-inflammatory, anthelmintic, tonic, hemorrhage, dysentery, aphrodisiac, and diaphoretic activities [34]. In this present study, we have reported the green synthesis and characterization of silver nanoparticles using P. santalinus leaf extract. To the best of our knowledge, this is the first report for the synthesis of Ag NPs using P. santalinus leaf extract.
Results and discussion
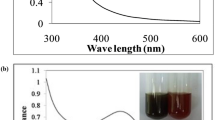
A reduction of Ag NPs was clearly observed when P. santalinus leaf extract was added with AgNO3 solution within 20 min. The colorless solution was changed to brown color which indicates the formation of silver nanoparticles.
UV-vis spectroscopy and Fourier transform-infrared spectroscopy analysis
The mixture of leaf extract and AgNO3 solution was subjected to ultraviolet-visible (UV-vis) spectroscopy analysis in the recorded spectra, and it showed a observable peak at 418 nm which corresponds to the wavelength of the surface plasmon resonance of Ag NPs (Figure 1). Various reports have established that the resonance peak of silver nanoparticles appears around this region [22]. Fourier transform-infrared (FT-IR) analysis was performed to identify the possible biomolecules responsible for the reduction of the Ag+ ions and capping of the reduced Ag NPs synthesized using P. santalinus leaf extract. The strong IR bands were observed at 3,382, 2,922, 2,337, 1,614, 1,384, 1,070, and 590 cm−1. The bands which appeared at 3,382 and 2,922 cm−1 correspond to -OH stretching and aliphatic -C-H stretching, respectively [35]. The bands at 2,337 and 1,613 cm−1 are due to the CO2 and C=C stretching, respectively. The IR bands observed at 1,384 and 1,070 cm−1 may be ascribed to -C-O and -C-O-C stretching modes, respectively. The low band at 590 cm−1 corresponds to C-Cl stretching. Hence, the main components such as steroids, saponins, tannins, phenols, triterpenoids, flavonoids, glycosides, and glycerides present in the leaf extract of P. santalinus are prime responsible for the observed reduction and capping during the synthesis of Ag NPs. The two new strong bands recorded at 825 and 463 cm−1 in the spectra of the synthesized material were assigned to C-H bending and metal (Ag), respectively. The C-H bending peak may be raised due to the reduction of AgNO3 to Ag nanoparticles (Figure 2).
X-ray diffraction and energy dispersive X-ray spectroscopy analysis
X-ray diffraction pattern (XRD) was recorded for the synthesized Ag NPs (Figure 3). Three distinct diffraction peaks at 38.04°, 44.23°, and 64.37° were indexed with the planes (111), (200), and (220) for the face-centered cubic silver as per the JCPDS card no. 89–3722 [36]. The well-resolved and intense XRD pattern clearly showed that the Ag NPs formed by the reduction of Ag+ ions using P. santalinus leaf extract are crystalline in nature. Similar results were reported for Ag NPs in the literature [14–22]. The low intense peak at 77.34° belongs to (311) plane. Energy-dispersive X-ray spectroscopy (EDX) (Figure 4) illustrated the chemical nature of synthesized silver nanoparticles using P. santalinus leaf extract. The peak was obtained at the energy of 3 keV for silver, and also some of the weak peaks for C, O, Cl, Al, and Na were found. The emission energy at 3 keV indicates the reduction of silver ions to element of silver. Similarly, C. quadrangularis stem extract derived silver nanoparticles showed an EDX spectrum, emission energy at 3 keV for silver, and some of the weak signals from Cl, K, O, Ca, Mg, and S were observed [25].
Atomic force microscopy and scanning electron microscopy
Surface topology of the formulated silver nanoparticles was studied by atomic force microscopy (AFM) analysis (Figure 5a, b). The micrographs clearly indicate that the formulated Ag NPs possess spherical shape and have the calculated sizes in the range of 20 to 50 nm. The scanning electron microscopy (SEM) image (Figure 6) further ascertains that the silver nanoparticles are predominantly spherical in morphology with their sizes ranging from 20 to 50 nm and have an average size of about 20 nm.
Antibacterial assay
The antibacterial assay was performed against three gram-positive and gram-negative bacterial pathogens using green-synthesized Ag NPs. The concentration of silver nanoparticles varied between 10, 50, and 100 μL (Table 1). Staphylococcus aureus and Streptococcus pneumoniae exhibit similar zone of inhibition for all three concentrations. The most significant effect of silver nanoparticles at low concentration of 10 μL per disc against S. dysenteriae is to produce a 2-mm inhibition zone of gram-negative bacteria, and at the same concentration, the nanoparticles did not show any significant effect on Bacillus subtilis, Pseudomonas aeruginosa, and Proteus vulgaris (Figure 7). The zone of inhibition increases with increasing concentration of silver nanoparticles as the Ag NPs bind with cytoplasmic membrane and killed the bacterial cell. Electrostatic attraction of silver nanoparticles causes damage of bacterial cell membrane to the formation of pits on the surface, and these structural changes take place due to cell expiration [37]. The prokaryotic bacteria have a mesosome cell organelle, and they are present in the inside of plasma membrane. It produced more enzymes as well as major function of cellular respiration, DNA replication, cell division, and increased the surface area of the bacterial cell membrane. Ag NPs interfere with the bacterial cell membrane and bind with mesosome cell organelle and after that reduce the mesosomal function and increase the ROS generation. Ag NPs interact with thiol groups in protein which induced the inactivation of the bacterial protein synthesis as well as DNA replication [38]. Similarly, oxygen associates with silver and reacts with the sulfhydryl (−S-H) groups on cell wall to remove the hydrogen atoms (as water), causing the sulfur atoms to form an R-S-S-R bond, blocked the respiration, and causing the lethal effect of bacterial cells [39]. Ag NPs naturally interact with the membrane of bacteria and disrupt the membrane integrity; silver ions bind to sulfur, oxygen, and nitrogen of essential biological molecules and inhibit bacterial growth [40].
Conclusions
The present work indicates the green-synthesized Ag NPs using P. santalinus leaf extract. The AFM and SEM images suggested that the particles are spherical shaped with average size of 20 nm. The antimicrobial activity depends upon the concentration of Ag NPs to produce the most significant effects against the gram-positive and gram-negative bacteria. This green-synthesized method is rapid, facile, convenient, less time consuming, environmentally safe, and can be applied in a variety of existing applications. This plant leaf extract compounds can be extended to the synthesis of other metal and metal oxide nanoparticles.
Methods
Synthesis of silver nanoparticles using P. santalinus leaf extract
Fresh P. santalinus plant leaves were collected from Karaikudi (Tamil Nadu, India) and used to retrieve their extraction. First, the leaves were cleaned with tap water, followed by distilled water and then finely cut into small pieces. Ten grams of finely cut leaves was added with 100 mL of double distilled water and boiled at 50°C to 60°C for 5 min. The obtained extraction was filtered using Whatman No. 1 filter paper, and the filtrate was collected in 250-mL Erlenmeyer flask and stored at room temperature for further usage. Then, 1 mL of P. santalinus leaf extract was added to 100 mL of 1 mM AgNO3 solution at room temperature, and the reduction of Ag NPs was clearly observed within the next 20 min.
Characterization
The synthesized silver nanoparticles were subjected to UV-vis analysis in the wavelength range of 350 to 800 nm using Shimadzu spectrophotometer (model UV-1800, Shimadzu, Kyoto, Japan) operating at a resolution of 1 nm. Also, FT-IR analysis was carried out in the range of 400 to 4,000 cm−1. XRD pattern was recorded using Cu Kα radiation (λ = 1.54060 Å) with nickel monochromator in the range of 2θ from 10° to 80°. The average crystallite size of the synthesized Ag NPs was calculated using Scherrer’s formula D = 0.9λ/β cosθ. EDX analysis for a thin film of the sample prepared on a aluminum foil (1 cm × 1 cm) by dropping 100 μL of the sample on the foil and allowed to dry for 30 min for further use. AFM analysis for a thin-film sample was prepared on a glass slide (1 cm × 1 cm) by dropping 100 μL of the sample on the slide and allowed to dry for 30 min. The slides were then scanned with AFM (APE Research-model no: A100SGS). The AFM characterization was carried out in ambient temperature in non-contact mode using silicon nitrite tips with varying resonance frequencies. The morphology of the synthesized Ag NPs was examined by SEM.
Antibacterial activity of Ag NPs
The minimum inhibitory concentration of the green-synthesized Ag NPs was examined using three gram-positive (B. subtilis, S. aureus, S. pneumoniae) and three gram-negative bacteria (P. aeruginosa, P. vulgaris, S. dysenteriae) were assayed by disk diffusion method. These six bacteria were grown in nutrient broth medium for 24 h. Approximately 20 mL of molten and cooled nutrient agar was poured into the petri dishes. The six tested organisms were swapped over the nutrient agar medium, and the disks containing silver nanoparticles were kept over the medium using sterile forceps. The silver nanoparticle-loaded disks were prepared at different concentrations of 10, 50, and 100 μL/6-mm disc. The plates were incubated for 24 h at 37°C, and the inhibition zone diameters were measured.
References
Sharma VK, Yngard RA, Lin Y: Silver nanoparticles: green synthesis and their antimicrobial activities. Adv. Colloid Interface Sci. 2009, 145: 83–96.
Chaloupka K, Malam Y, Seifalian AM: Nanosilver as a new generation of nanoproduct in biomedical applications. Trends Biotechnol. 2010, 28: 580–588.
Prow TW, Grice JE, Lin LL, Faye R, Butler M, Becker W, Wurm EMT, Yoong C, Robertson TA, Soyer HP, Roberts MS: Nanoparticles and microparticles for skin drug delivery. Adv. Drug Deliv. Rev. 2011, 63: 470–491.
Chaudhry Q, Castle L: Food applications of nanotechnologies: an overview of opportunities and challenges for developing countries. Trends Food Sci. Tech. 2011,2011(22):595–603.
Nair R, Varghese SH, Nair BG, Maekawa T, Yoshida Y, Sakthi Kumar D: Nanoparticulate material delivery to plants. Plant Sci. 2010, 179: 154–163.
Kelly FM, Johnston JH: Colored and functional silver nanoparticle wool fiber composites. ACS Appl. Mater. Interfaces 2011, 3: 1083–1092.
Dankovich TA, Gray DG: Bactericidal paper impregnated with silver nanoparticles for point-of-use water treatment. Environ. Sci. Technol. 2011, 45: 1992–1998.
Niraimathi KL, Sudha V, Lavanya R, Brindha P: Biosynthesis of silver nanoparticles using Alternanthera sessilis (Linn.) extract and their antimicrobial, antioxidant activities. Colloid. Surface B 2013, 102: 288–291.
Sankar R, Karthik A, Prabu A, Karthik S, Shivashangari KS, Ravikumar V: Origanum vulgare mediated biosynthesis of silver nanoparticles for its antibacterial and anticancer activity. Colloid. Surface B. 2013, 108: 80–84.
Boca SC, Potara M, Gabudean AM, Juhem A, Baldeck PL, Astilean S: Chitosan-coated triangular silver nanoparticles as a novel class of biocompatible, highly effective photothermal transducers for in vitro cancer cell therapy. Cancer Lett. 2011, 31: 131–140.
Jain J, Arora S, Rajwade JM, Omray P, Khandelwal S, Paknikar KM: Silver nanoparticles in therapeutics: development of an antimicrobial gel formulation for topical use. Mol. Pharm. 2009, 6: 1388–1401.
Murphy CJ: Sustainability as a design criterion in nanoparticle synthesis and applications. J. Mater. Chem. 2008, 18: 2173–2176.
Roopan SM, Rohit , Madhumitha G, Abdul Rahuman A, Kamaraj C, Bharathi A, Surendra TV: Low-cost and eco-friendly phyto-synthesis of silver nanoparticles using Cocos nucifera coir extract and its larvicidal activity. Ind. Crop Prod. 2013, 43: 631–635.
Jagtap U, Bapa VA: Green synthesis of silver nanoparticles using Artocarpus heterophyllus Lam. Seed extract and its antibacterial activity. Ind. Crop Prod. 2013, 46: 132–137.
Das J, Paul Das M, Velusamy P: Sesbania grandiflora leaf extract mediated green synthesis of antibacterial silver nanoparticles against selected human pathogens. Spectrochim. Acta. A. 2013, 104: 265–270.
Edison TJ, Sethuraman MG: Biogenic robust synthesis of silver nanoparticles using Punica granatum peel and its application as a green catalyst for the reduction of an anthropogenic pollutant 4-nitrophenol. Spectrochim. Acta. A. 2013, 104: 262–264.
Raman N, Sudharsan S, Veerakumar V, Pravin N, Vithiya K: Pithecellobium dulce mediated extra cellular green synthesis of larvicidal silver nanoparticles. Spectrochim. Acta. A. 2012, 96: 1031–1037.
Zayed MF, Eisa WH, Shabaka AA: Malva parviflora extract assisted green synthesis of silver nanoparticles. Spectrochim. Acta. A. 2012, 98: 423–428.
Dipankar C, Murugan S: The green synthesis, characterization and evaluation of the biological activities of silver nanoparticles synthesized from Iresine herbstii leaf aqueous extracts. Colloid. Surface B. 2012, 98: 112–119.
Bindhu MR, Umadevi M: Synthesis of monodispersed silver nanoparticles using Hibiscus cannabinus leaf extract and its antimicrobial activity. Spectrochim. Acta. A. 2013, 101: 184–190.
Guidelli EJ, Ramos AP, Zaniquelli ME, Baffa O: Green synthesis of colloidal silver nanoparticles using natural rubber latex extracted from Hevea brasiliensis . Spectrochim. Acta. A. 2011, 82: 140–145.
Zahir AA, Rahuman AA: Evaluation of different extracts and synthesised silver nanoparticles from leaves of Euphorbia prostrata against Haemaphysalis bispinosa and Hippobosca maculata . Vet. Parasitol. 2012, 187: 511–520.
Valli JS, Vaseeharan B: Biosynthesis of silver nanoparticles by Cissus quadrangularis extracts. Mater. Lett. 2012, 82: 171–173.
Santhoshkumar T, Rahuman AA, Bagavan A, Marimuthu S, Jayaseelan C, Kirthi AV, Kamaraj C, Rajakumar G, Zahir AA, Elango G, Velayutham K, Iyappan M, Siva C, Karthik L, Rao KV: Evaluation of stem aqueous extract and synthesized silver nanoparticles using Cissus quadrangularis against Hippobosca maculata and Rhipicephalus (Boophilus) microplus . Exp. Parasitol. 2012, 132: 156–165.
Vanaja M, Gnanajobitha G, Paulkumar K, Rajeshkumar S, Malarkodi C, Annadura G: Phytosynthesis of silver nanoparticles by Cissus quadrangularis : influence of physicochemical factors. J. Nanostructure Chem. 2013, 3: 17.
Kotakadi VS, Rao YS, Gaddam SA, Prasad TNVKV, Reddy AV, Gopal DVRS: Simple and rapid biosynthesis of stable silver nanoparticles using dried leaves of Catharanthus roseus. Linn. G. Donn and its anti microbial activity. Colloid. Surface B. 2013, 105: 194–198.
Arunachalam R, Dhanasingh S, Kalimuthu B, Uthirappan M, Rose C, Mandal AB: Phytosynthesis of silver nanoparticles using Coccinia grandis leaf extract and its application in the photocatalytic degradation. Colloid. Surface B. 2012, 94: 226–230.
Karuppiah M, Rajmohan R: Green synthesis of silver nanoparticles using Ixora coccinea leaves extract. Mater. Lett. 2013, 97: 141–143.
Cruz D, Falé PL, Mourato A, Vaz PD, Serralheiro ML, Lino ARL: Preparation and physicochemical characterization of Ag nanoparticles biosynthesized by Lippia citriodora (Lemon Verbena). Colloid. Surface B. 2010, 81: 67–73.
Rajakumar G, Rahuman AA: Acaricidal activity of aqueous extract and synthesized silver nanoparticles from Manilkara zapota against Rhipicephalus (Boophilus) microplus . Res. Vet. Sci. 2012, 93: 303–309.
Tamuly C, Hazarika M, Borah SC, Das MR, Boruah MP: In situ biosynthesis of Ag, Au and bimetallic nanoparticles using Piper pedicellatum C.DC: Green chemistry approach. Colloid. Surface B 2013, 102: 627–634.
Raja K, Saravanakumar A, Vijayakumar R: Efficient synthesis of silver nanoparticles from Prosopis juliflora leaf extract and its antimicrobial activity using sewage. Spectrochim. Acta. A. 2012, 97: 490–494.
Raju D, Hazra S, Hazra S, Mehta UJ: Phytosynthesis of silver nanoparticles by Semecarpus anacardium L. leaf extract. Mater. Lett. 2013, 102–103: 5–7.
Arunakumara KKIU, Walpola BC, Sibasinghe S, Yoon MH: Pterocarpus santalinus Linn. f. (Rath handun): a review of its botany, uses, phytochemistry and pharmacology. J. Korean Soc. Appl. Biol. Chem. 2011, 54: 495–500.
Kumar KM, Mandal BK, Sinha M, Krishnakumar V: Terminalia chebula mediated green and rapid synthesis of gold nanoparticles. Spectrochim. Acta. A. 2012, 86: 490–494.
Vivek R, Thangam R, Muthuchelian K, Gunasekaran P, Kaveri K, Kannan S: Green biosynthesis of silver nanoparticles from Annona squamosa leaf extract and its in vitro cytotoxic effect on MCF-7 cells. Process Biochem. 2012, 47: 2405–2410.
Sondi I, Salopek-Sondi B: Silver nanoparticles as antimicrobial agent: a case study on E.coli as a model for Gram-negative bacteria. J. Colloid Interface Sci. 2004, 275: 177–182.
Feng QL, Wu J, Chen GQ, Cui FZ, Kim TN, Kim JO: A mechanistic study of the antibacterial effect of silver ions on Escherichia coli and Staphylococcus aureus . J. Biomed. Mater. Res. 2000, 52: 662–668.
Siva Kumar V, Nagaraja BM, Shashikala V, Padmasri AH, Madhavendra SS, Raju BD, Rama Rao KS: Highly efficient Ag/C catalyst prepared by electro-chemical deposition method in controlling microorganisms in water. J. Mol. Catal. A. Chem. 2004, 223: 313–319.
Juan L, Zhimin Z, Anchun M, Lei L, Jingchao Z: Deposition of silver nanoparticles on titanium surface for antibacterial effect. Int. J. Nanomed. 2010, 5: 261–267.
Acknowledgments
The authors gratefully thank School of Physics, Alagappa University for extending the XRD facility and also the Department of Industrial Chemistry, Alagappa University for providing the SEM with EDX facilities.
Author information
Authors and Affiliations
Corresponding author
Additional information
Competing interests
The authors declare that they have no competing interests.
Authors’ contributions
KG and SG carried out the nanoparticles synthesis, characterization, and antimicrobial activity. AA carried out the manuscript preparation. All authors read and approved the final manuscript.
Authors’ original submitted files for images
Below are the links to the authors’ original submitted files for images.
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 2.0 International License ( https://creativecommons.org/licenses/by/2.0 ), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
About this article
Cite this article
Gopinath, K., Gowri, S. & Arumugam, A. Phytosynthesis of silver nanoparticles using Pterocarpus santalinus leaf extract and their antibacterial properties. J Nanostruct Chem 3, 68 (2013). https://doi.org/10.1186/2193-8865-3-68
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/2193-8865-3-68