Abstract
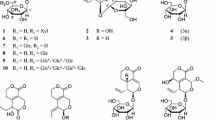
Sixteen known compounds, including three lignan glycosides (1–3), seven phenolic glycosides (4–10), three glycosides (11–13), and three flavonoids (14–16), were isolated from the rhizomes of Actinidia kolomikta (Rupr. & Maxim.) Maxim., Actinidiaceae. Their structures were elucidated through extensive spectroscopic analyses and comparison with those reported in the literature. This study is the first confirmation of the presence of the twelve compounds (1–4, 6–13) isolated from the Actinidiaceae family. The isolated compounds were evaluated for their inhibitory effects on NO, TNF-α, and IL-6 production in LPS-induced RAW264.7 cells. Among them, salidroside (9), D-threo-guaiacylglycerol 7-O-β-D-glucopyranoside (10), and epi-afzelechin (15) exhibited the potent anti-inflammatory effect against NO, TNF-α, and IL-6. In addition, compound 10 showed significant inhibitory effects on COX-2 expression at 50 μM, and compound 15 showed significant inhibitory effects on iNOS expression at 50 μM. These results suggest that the anti-inflammatory activity of A. kolomikta might in part occur via the inhibition of the expression of the pro-inflammatory cytokines.

Similar content being viewed by others
Change history
26 March 2020
This article was updated to include the chemical structures missed during the production process.
References
Abdel-salam NA, Ghazy NM, Sallam SM, Radwan MM, Wanas AS, ElSohly MA, El-Demellawy MA, Abdel-Rahman NM, Piacente S, Shenouda ML (2017) Flavonoids of Alcea rosea L. and their immune stimulant, antioxidant and cytotoxic activities on hepatocellular carcinoma HepG-2 cell line. Nat Prod Res 32:702–706
Binder TP, Robyt JF (1986) Inhibition- and acceptor-reaction studies of Streptococcus mutans 6715 glucosyltransferases with 3-deoxysucrose, 3-deoxy-3-fluorosucrose, and α-D-allopyranosyl β-D-fructofuranoside. Carbohydr Res 154:229–238
Bogačiovienė S, Česonienė L, Ercisli S, Valatavičius A, Jakštys Šatkauskas S, Paulauskaset A (2019) Ploidy levels and genetic diversity of Actinidia arguta (Siebold & Zucc.) Planch. ex Miq., A. kolomikta (Rupr. & Maxim.) Maxim., A. cfallosa Lindl., and A. melanandra Franch., accessions. Genet Resour Crop Evol 66:1107–1118
Cha JM, Suh WS, Lee TH, Subedi L, Kim SY, Lee KR (2017) Phenolic glycosides from Capsella bursa-pastoris (L.) Medik and their anti-inflammatory activity. Molecules 22:1023/1–1023/8
Chesoniene L, Daubaras R, Viskelis P (2004) Biochemical composition of berries of some kolomikta kiwi (Actinidia kolomikta) cultivars and detection of harvest maturity. Acta Hortic 663:305–308
Coussens LM, Werb Z (2002) Inflammation and cancer. Nature 420:860–867
Guan D, Zhang Z, Yang Y, Sugiura N, Hu H, Xing G, Liu J, Zu X, Zhang Y (2011) Antioxidant and antitumor activities of water extracts from the root of Actinidia kolomikta. Exp Ther Med 2:33–39
Hao X, Chen J, Lai Y, Sang M, Yao G, Xue Y, Luo Z, Zhang G, Zhang Y (2015) Chemical constituents from leaves of Cinnamomum subavenium. Biochem Syst Ecol 61:156–160
Hori K, Wada M, Yahara S, Watanabe T, Devkota HP (2017) Antioxidant phenolic compounds from the rhizomes of Astilbe rivularis. Nat Prod Res 32:453–456
Hu JM, Chen JJ, Yu H, Zhao YX, Zhou J (2008) Five new compounds from Dendrobium longicornu. Planta Med 74:535–539
Hu X, Cheng D, Wang L, Li S, Wang Y, Li K, Yang Y, Zhang Z (2015) Evaluation of anti-hyperglycemic effect of Actinidia kolomikta (Maxim. et Rur.) Maxim. root extract. Pak J Pharm Sci 28:1135–1140
Hyun SK, Jung HA, Min BS, Jung JH, Choi JS (2010) Isolation of phenolics, nucleosides, saccharides and an alkaloid from the root of Aralia cordata. Nat Prod Sci 16:20–25
Jiang J, Feng Z, Wang Y, Zhang P (2005) New phenolics from the roots of Symplocos caudate Wall. Chem Pharm Bull 53:110–113
Jung J, Park S, Hong S, Ha M, Park HG, Park Y, Lee HJ, Park Y (2014) Synthesis of gold nanoparticles with glycosides: synthetic trends based on the structures of glycones and aglycones. Carbohydr Res 386:57–61
Jung YW, Lee BM, Ha MT, Tran MH, Kim JA, Lee S, Lee JH, Woo MH, Min BS (2019) Lignans from Saururus chinensis exhibit anti-inflammatory activity by influencing the Nrf2/HO-1 activation pathway. Arch Pharm Res 42:332–343
Lee CK, Birch GG (1976) Structural functions of taste in the sugar series VII: taste properties of ketoses. J Pharm Sci 65:1222–1225
Li G, Wang J, Jin M, Jin C, Ye C, Zhou Y, Wang R, Cui H, Zhou W (2019) A new pentacyclic triterpenoid from the leaves of Rhododendron dauricum L. with inhibition of NO production in LPS-induced RAW 264.7 cells. Nat Prod Res. https://doi.org/10.1080/14786419.2019.1566822
Ngoc TM, Khoi NM, Ha DT, Nhiem NX, Tai BH, Don DV, Luong HV, Son DC, Bae KH (2012) Xanthine oxidase inhibitory activity of constituents of Cinnamomum cassia twigs. Bioorg Med Chem Lett 22:4625–4628
Santos CCS, Masullo M, Cerulli A, Mari A, Estevam CDS, Pizza C, Piacente S (2017) Isolation of antioxidant phenolics from Schinopsis brasiliensis based on a preliminary LC-MS profiling. Phytochemistry 140:45–51
Su XD, Ali I, Arooj M, Koh YS, Yang SY, Kim YH (2018a) Chemical constituents from Sanguisorba officinalis L. and their inhibitory effects on LPS-stimulated pro-inflammatory cytokine production in bone marrow-derived dendritic cells. Arch Pharm Res 41:497–505
Su XD, Guo RH, Li HX, Ma JY, Kim YH (2018b) Anti-allergic inflammatory components from Sanguisorba officinalis L. Bioorg Med Chem Lett 28:2210–2216
Tran HNK, Cao TQ, Kim JA, Woo MH, Min BS (2019) Anti-inflammatory and cytotoxic activities of constituents isolated from the fruits of Ziziphus jujuba var. inermis Rehder. Fitoterapia 137:104261. https://doi.org/10.1016/j.fitote.2019.104261
Wang C, Xiao Y, Yang B, Wang Z, Wu L, Su X, Brantner A, Kuang H, Wang Q (2014) Isolation and screened neuroprotective active constituents from the roots and rhizomes of Valeriana amurensis. Fitoterapia 96:48–55
Xu F, Fan W, Zi C, Dong F, Yang D, Zhou J, Hu J (2016) Four new glycosides from the stems of Dendrobium fimbriatum Hook. Nat Prod Res 31:797–801
Ye C, Jin M, Jin C, Wang R, Wang J, Zhang Y, Li S, Sun J, Zhou W, Li G (2019) Two novel flavonoids from the leaves of Rhododendron dauricum L. with their inhibition of TNF-α production in LPS-induced RAW 264.7 cells. Nat Prod Res. https://doi.org/10.1080/14786419.2019.1648455
Zhou Y, Jin M, Zhou W, Gao L (2018) Chemical constituents of the stem barks of Quercus mongolica. Chem Nat Compd 54:973–974
Zuo L, Wang Z, Fan Z, Tian S, Liu J (2012) Evaluation of antioxidant and antiproliferative properties of three Actinidia (Actinidia kolomikta, Actinidia arguta, Actinidia chinensis) extracts in vitro. Int J Mol Sci 13:5506–5518
Acknowledgments
We thank Alison McGonagle, PhD, from Liwen Bianji, Edanz Group China (www.liwenbianji.cn/ac), for editing the English text of a draft of this manuscript.
Funding
This work was supported by the National Natural Science Foundation of China under Grant numbers (81660699, 81660579, and 81760627).
Author information
Authors and Affiliations
Contributions
CY and JS carried out the extract, isolation, purification, and running the laboratory work. MJ, RL, CJ, LJ, and YJM contributed to biological studies. WZ and GL contributed equally to this paper on structural determination and writing this paper.
Corresponding authors
Electronic Supplementary Material
ESM 1
(DOCX 2002 kb)
Rights and permissions
About this article
Cite this article
Ye, C., Jin, M., Jin, C. et al. Inhibitory Effects of Chemical Constituents from Actinidia kolomikta on LPS-Induced Inflammatory Responses. Rev. Bras. Farmacogn. 30, 127–131 (2020). https://doi.org/10.1007/s43450-020-00004-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s43450-020-00004-w