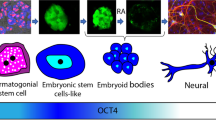
Abstract
The POU domain, class 5, transcription factor 1 (POU5F1), plays a vital role in creating pluripotency and maintaining self-renewal of the spermatogonial stem cells (SSCs). In this experimental research, the gene and protein expression of POU5F1 in two populations of differentiated and undifferentiated spermatogonia were examined, by immunohistochemistry (IMH), immunocytochemistry (ICC) and Fluidigm real-time RT-PCR. Our study was extended with online databases and the creation of PPI networks. The results indicated that the POU5F1 protein was localized in the basal compartment of seminiferous tubules. Under in vitro conditions, isolated SSC colonies were ICC-positive for the POU5F1, but the protein expression level of POU5F1 in the undifferentiated populations was higher than that in differentiated. A significant POU5F1 mRNA expression was seen in passage 4 compared to passage 0 for both populations. POU5F1 has a significantly higher mRNA expression in undifferentiated SSCs than that in differentiated SSCs, also in mESCs than in SSC-like cells. Bioinformatic analysis on POU5F1 shows its impressive connection with other genes involved in spermatogonia differentiation. These results support the advanced investigations of spermatogonia differentiation, both in vitro and in vivo. A better understanding of the POU5F1 gene and its function during differentiation will give the scientific community an open perspective for the development of direct differentiation of SSC to other male germline cells which is very important in infertility treatment.





Similar content being viewed by others
Data availability
All data generated or analyzed during this study are included in this article. Further enquiries can be directed to the corresponding author.
References
Azimi A, Mohaqiq M, Movahedin M, Mazaheri Z (2020) Characterization of embryonic stem-like cells derived from mouse spermatogonial stem cells following low-intensity ultrasound treatment. Rev Int Androl. https://doi.org/10.1016/j.androl.2020.05.010
Azizi H, Asgari B, Skutella T (2019) Pluripotency potential of embryonic stem cell-like cells derived from mouse testis. Cell J 21(3):281. https://doi.org/10.22074/cellj.2019.6068
Azizi H, Conrad S, Hinz U, Asgari B, Nanus D, Peterziel H, Hajizadeh Moghaddam A, Baharvand H, Skutella T (2016) Derivation of pluripotent cells from mouse SSCs seems to be age dependent. Stem Cells Int. https://doi.org/10.1155/2016/8216312
Azizi H, Tabar AN, Skutella T, Govahi M (2020) In vitro and in vivo determinations of the anti-gdnf family receptor alpha 1 antibody in mice by immunochemistry and RT-PCR. Int J Fertil Steril 14(3):228. https://doi.org/10.22074/ijfs.2020.6051
Dann CT, Alvarado AL, Molyneux LA, Denard BS, Garbers DL, Porteus MH (2008) Spermatogonial stem cell self-renewal requires OCT4, a factor downregulated during retinoic acid-induced differentiation. Stem Cells 26(11):2928–2937. https://doi.org/10.1634/stemcells.2008-0134
Fayomi AP, Orwig KE (2018) Spermatogonial stem cells and spermatogenesis in mice, monkeys and men. Stem Cell Res 29:207–214. https://doi.org/10.1016/j.scr.2018.04.009
He Z, Jiang J, Hofmann M-C, Dym M (2007) Gfra1 silencing in mouse spermatogonial stem cells results in their differentiation via the inactivation of RET tyrosine kinase. Biol Reprod 77(4):723–733. https://doi.org/10.1095/biolreprod.107.062513
Ibtisham F, Honaramooz A (2020) Spermatogonial stem cells for in vitro spermatogenesis and in vivo restoration of fertility. Cells 9(3):745. https://doi.org/10.3390/cells9030745
Kitadate Y, Jörg DJ, Tokue M et al (2019) Competition for mitogens regulates spermatogenic stem cell homeostasis in an open niche. Cell Stem Cell 24(1):79-92.e6. https://doi.org/10.1016/j.stem.2018.11.013
Kubota H (2019) Heterogeneity of spermatogonial stem cells. In: Birbrair A (ed) Stem Cells Heterogeneity in Different Organs. Springer International Publishing, Cham, pp 225–242. https://doi.org/10.1007/978-3-030-24108-7_12
Kubota H, Avarbock MR, Brinster RL (2004) Growth factors essential for self-renewal and expansion of mouse spermatogonial stem cells. Proc Natl Acad Sci U S A 101(47):16489–16494. https://doi.org/10.1073/pnas.0407063101
Kubota H, Brinster RL (2018) Spermatogonial stem cells. Biol Reprod 99(1):52–74. https://doi.org/10.1093/biolre/ioy077
Lavial F, Acloque H, Bertocchini F, MacLeod DJ, Boast S, Bachelard E, Montillet G, Thenot S, Sang HM, Stern CD (2007) The Oct4 homologue PouV and Nanog regulate pluripotency in chicken embryonic stem cells. Dev 134(19):3549–3563. https://doi.org/10.1242/dev.006569
Lee S-W, Wu G, Choi NY, Lee HJ, Bang JS, Lee Y, Lee M, Ko K, Schöler HR, Ko K (2018) Self-reprogramming of spermatogonial stem cells into pluripotent stem cells without microenvironment of feeder cells. Mol Cells 41(7):631. https://doi.org/10.14348/molcells.2018.2294
Li R, Vannitamby A, Zhang J-G, Fehmel EL, Southwell BR, Hutson JM (2015) Oct4-GFP expression during transformation of gonocytes into spermatogonial stem cells in the perinatal mouse testis. J Pediatr Surg 50(12):2084–2089. https://doi.org/10.1016/j.jpedsurg.2015.08.031
Looijenga LH, Stoop H, De Leeuw HP, de Gouveia Brazao CA, Gillis AJ, Van Roozendaal KE, Van Zoelen EJ, Weber RF, Wolffenbuttel KP, Van Dekken H (2003) POU5F1 (OCT3/4) identifies cells with pluripotent potential in human germ cell tumors. Cancer Res 63(9):2244–2250
Lord T, Nixon B (2020) Metabolic changes accompanying spermatogonial stem cell differentiation. Dev Cell 52(4):399–411. https://doi.org/10.1016/j.devcel.2020.01.014
Malik V, Glaser LV, Zimmer D, Velychko S, Weng M, Holzner M, Arend M, Chen Y, Srivastava Y, Veerapandian V (2019) Pluripotency reprogramming by competent and incompetent POU factors uncovers temporal dependency for Oct4 and Sox2. Nat Commun 10(1):1–16. https://doi.org/10.1038/s41467-019-11054-7
Masui S, Nakatake Y, Toyooka Y, Shimosato D, Yagi R, Takahashi K, Okochi H, Okuda A, Matoba R, Sharov AA (2007) Pluripotency governed by Sox2 via regulation of Oct3/4 expression in mouse embryonic stem cells. Nat Cell Biol 9(6):625–635. https://doi.org/10.1038/ncb1589
Niknejad P, Azizi H, Sojoudi K (2021) POU5F1 protein and gene expression analysis in neonate and adult mouse testicular germ cells by immunohistochemistry and immunocytochemistry. Cell Reprogram 23(6):349–358. https://doi.org/10.1089/cell.2021.0108
Oatley JM, Brinster RL (2008) Regulation of spermatogonial stem cell self-renewal in mammals. Annu Rev Cell Dev Biol 24:263–286. https://doi.org/10.1146/annurev.cellbio.24.110707.175355
Picot T, Kesr S, Wu Y, Aanei CM, Flandrin-Gresta P, Tondeur S, Tavernier E, Wattel E, Guyotat D, Campos L (2017) Potential role of OCT4 in leukemogenesis. Stem Cells Dev 26(22):1637–1647. https://doi.org/10.1089/scd.2017.0134
Rasouli-Gharehsaghal K, Shakeri M, Zhandi M, Amini H-R, Yousefi AR, AsadiRad M (2020) Improvement of in vitro proliferation of cockerel spermatogonial stem cells using different combinations of growth factors. Br Poult Sci 61(6):660–668. https://doi.org/10.1080/00071668.2020.1808187
Simmet K, Zakhartchenko V, Philippou-Massier J, Blum H, Klymiuk N, Wolf E (2018) OCT4/POU5F1 is required for NANOG expression in bovine blastocysts. Proc Natl Acad Sci U S A 115(11):2770–2775. https://doi.org/10.1073/pnas.1718833115
Singh SR, Burnicka-Turek O, Chauhan C, Hou SX (2011) Spermatogonial stem cells, infertility and testicular cancer. J Cell Mol Med 15(3):468–483. https://doi.org/10.1111/j.1582-4934.2010.01242.x
Sohn EJ, Moon HJ, Lim JK, Kim DS, Kim JH (2021) Regulation of the protein stability and transcriptional activity of OCT4 in stem cells. Adv Biol Regul 79:100777. https://doi.org/10.1016/j.jbior.2020.100777
Song HW, Wilkinson MF (2014) Transcriptional control of spermatogonial maintenance and differentiation. Semin Cell Dev Biol. https://doi.org/10.1016/j.semcdb.2014.02.005
Tabar AN, Sojoudi K, Henduei H, Azizi H (2022) Review of sertoli cell dysfunction caused by COVID-19 that could affect male fertility. Zygote 30(1):17–24. https://doi.org/10.1017/S0967199421000320
Takashima S, Shinohara T (2018) Culture and transplantation of spermatogonial stem cells. Stem Cell Res 29:46–55. https://doi.org/10.1016/j.scr.2018.03.006
Wang X, Dai J (2010) Concise review: isoforms of OCT4 contribute to the confusing diversity in stem cell biology. Stem Cells 28(5):885–893. https://doi.org/10.1002/stem.419
Wu J, Vallenius T, Ovaska K, Westermarck J, Mäkelä TP, Hautaniemi S (2009) Integrated network analysis platform for protein-protein interactions. Nat Methods 6(1):75–77. https://doi.org/10.1038/nmeth.1282
Yoshimizu T, Sugiyama N, De Felice M, Yeom YI, Ohbo K, Masuko K, Obinata M, Abe K, Schöler HR, Matsui Y (1999) Germline-specific expression of the Oct-4/green fluorescent protein (GFP) transgene in mice. Dev Growth Differ 41(6):675–684. https://doi.org/10.1046/j.1440-169x.1999.00474.x
Zhang J, Tam W-L, Tong GQ, Wu Q, Chan H-Y, Soh B-S, Lou Y, Yang J, Ma Y, Chai L (2006) Sall4 modulates embryonic stem cell pluripotency and early embryonic development by the transcriptional regulation of Pou5f1. Nat Cell Biol 8(10):1114–1123. https://doi.org/10.1038/ncb1481
Zhang X, Zhang J, Wang T, Esteban MA, Pei D (2008) Esrrb activates Oct4 transcription and sustains self-renewal and pluripotency in embryonic stem cells. J Biol Chem 283(51):35825–35833. https://doi.org/10.1074/jbc.M803481200
Acknowledgements
The present article extracted from master dissertation of microbial biotechnology at Amol University of Special Modern Technologies, Iran, in 2021. I wish to express my sincere thanks to Amol University of special modern technologies, Principal of the Faculty, for providing me with all the necessary facilities for the research. I take this opportunity to express gratitude to all of the Department faculty members for their help and support. I also acknowledge my colleagues for their wonderful collaboration.
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Author information
Authors and Affiliations
Contributions
MM wrote the manuscript and analyzed data. HA carried out and designed the experiment, assembled data and analyzed data. KS designed PPI network analysis, wrote the manuscript, analyzed data and edited the final document. MY wrote the manuscript and edited the final document. DGH provided critical feedback and analyzed data. The authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
We have no competing interests.
Ethical approval
In the current investigation, animal experiments were approved (Ir.ausmt.rec.1400.03) by the Ethics Committee of Amol University of Special Modern Technologies.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Masoudi, M., Azizi, H., sojoudi, K. et al. Comparison of POU5F1 gene expression and protein localization in two differentiated and undifferentiated spermatogonial stem cells. BIOLOGIA FUTURA 73, 503–512 (2022). https://doi.org/10.1007/s42977-022-00149-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s42977-022-00149-w