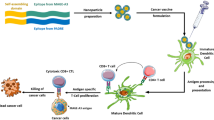
Abstract
Immunotherapy has been considered as one of the most promising strategies for protection against cancer cells due to the tremendous advantages arising from host immune defense. However, establishing versatile strategies with high biosafety and the capability for efficient modulation of immune responses remains challenging. The structural features resembling native proteins of peptides bestow their great potential to address these challenges via either directly eliciting immune responses or improving the efficacy of therapeutics. This review summarizes the progress of cancer immunotherapy achieved based on the strategies utilizing short peptides as therapeutic agents or peptide assemblies as delivery scaffolds, beyond long sequences like proteins and polypeptides. Starting from a brief introduction of cancer immunotherapy, we outline the peptide sequences in terms of their specific functions including immune checkpoint blockades, vaccine antigens and adjuvants. We particularly high-light peptide-based nanomaterials as scaffolds for targeting delivery or co-delivery of multiple therapeutics to enhance immunogenicity. The extraordinary therapeutic efficacy of the limited examples covered here demonstrates the great potency of the peptide-based strategies in modulating immune responses, thus potentially facilitating the clinical translation of cancer immunotherapy in the future.
摘要
基于通过激活宿主天然免疫应答杀死癌症细胞的优势, 免疫治疗被认为有望成为癌症治疗的新方法. 与传统治疗方法相比, 免疫治疗能够诱导长期的免疫记忆以预防癌症复发和转移, 具有更广谱的抗癌效果以及较小的副作用. 然而建立具有高生物安全性和免疫应答能力的策略仍然具有挑战性. 由于其与天然蛋白质类似的结构特征, 多肽分子有望通过直接引发免疫应答或改善药物递送效果来解决这些挑战. 本文总结了过去十年内发现的利用短肽分子作为免疫治疗药物或递送平台的癌症免疫疗法. 从简要介绍癌症免疫治疗开始, 我们概述了多肽分子的特定药物功能, 包括免疫检查点抑制剂、 疫苗抗原和佐剂. 随后着重介绍了基于多肽纳米结构作为递送平台, 用于药物靶向递送或多种药物共同递送以增强免疫原性的进展. 最后对基于多肽的癌症免疫治疗面临的挑战以及未来的发展趋势进行了展望.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Littman DR. Releasing the brakes on cancer immunotherapy. Cell, 2015, 162: 1186–1190
Del Paggio JC. Cancer immunotherapy and the value of cure. Nat Rev Clin Oncol, 2018, 15: 268–270
Waldmann TA. Immunotherapy: Past, present and future. Nat Med, 2003, 9: 269–277
Couzin-Frankel J. Cancer immunotherapy. Science, 2013, 342: 1432–1433
Sahin U, Türeci Ö. Personalized vaccines for cancer immunotherapy. Science, 2018, 359: 1355–1360
Mellman I, Coukos G, Dranoff G. Cancer immunotherapy comes of age. Nature, 2011, 480: 480–489
Byun DJ, Wolchok JD, Rosenberg LM, et al. Cancer immunotherapy—immune checkpoint blockade and associated endocrinopathies. Nat Rev Endocrinol, 2017, 13: 195–207
Ribas A, Wolchok JD. Cancer immunotherapy using checkpoint blockade. Science, 2018, 359: 1350–1355
Park JH, Geyer MB, Brentjens RJ. CD19-targeted CAR T-cell therapeutics for hematologic malignancies: Interpreting clinical outcomes to date. Blood, 2016, 127: 3312–3320
Cheadle EJ, Gornall H, Baldan V, et al. CAR T cells: Driving the road from the laboratory to the clinic. Immunol Rev, 2013, 257: 91–106
Cao Y, Rodgers DT, Du J, et al. Design of switchable chimeric antigen receptor T cells targeting breast cancer. Angew Chem Int Ed, 2016, 55: 7520–7524
Golubovskaya V. CAR-T cell therapy: From the bench to the bedside. Cancers, 2017, 9: 150
Brudno JN, Kochenderfer JN. Chimeric antigen receptor T-cell therapies for lymphoma. Nat Rev Clin Oncol, 2018, 15: 31–46
Zhang S. Discovery and design of self-assembling peptides. Interface Focus, 2017, 7: 20170028
Cui H, Webber MJ, Stupp SI. Self-assembly of peptide amphiphiles: From molecules to nanostructures to biomaterials. Biopolymers, 2010, 94: 1–18
Barnes CP, Sell SA, Boland ED, et al. Nanofiber technology: Designing the next generation of tissue engineering scaffolds. Adv Drug Deliver Rev, 2007, 59: 1413–1433
Zasloff M. Antimicrobial peptides of multicellular organisms. Nature, 2002, 415: 389–395
Mahlapuu M, Håkansson J, Ringstad L, et al. Antimicrobial peptides: An emerging category of therapeutic agents. Front Cell Infect Microbiol, 2016, 6
Lee S, Xie J, Chen X. Peptides and peptide hormones for molecular imaging and disease diagnosis. Chem Rev, 2010, 110: 3087–3111
Abbas M, Zou Q, Li S, et al. Self-assembled peptide- and protein-based nanomaterials for antitumor photodynamic and photothermal therapy. Adv Mater, 2017, 29: 1605021
Li J, Pu K. Development of organic semiconducting materials for deep-tissue optical imaging, phototherapy and photoactivation. Chem Soc Rev, 2019, 48: 38–71
Miao Q, Yeo DC, Wiraja C, et al. Near-infrared fluorescent molecular probe for sensitive imaging of keloid. Angew Chem Int Ed, 2018, 57: 1256–1260
Yin L, Sun H, Zhang H, et al. Quantitatively visualizing tumor-related protease activity in vivo using a ratiometric photoacoustic probe. J Am Chem Soc, 2019, 141: 3265–3273
Zhang P, Cheetham AG, Lin YA, et al. Self-assembled tat nano-fibers as effective drug carrier and transporter. ACS Nano, 2013, 7: 5965–5977
Zhang N, Zhao F, Zou Q, et al. Multitriggered tumor-responsive drug delivery vehicles based on protein and polypeptide coassembly for enhanced photodynamic tumor ablation. Small, 2016, 12: 5936–5943
Agemy L, Friedmann-Morvinski D, Ramana Kotamraju V, et al. Targeted nanoparticle enhanced proapoptotic peptide as potential therapy for glioblastoma. Proc Natl Acad Sci USA, 2011, 108: 17450–17455
Junttila MR, de Sauvage FJ. Influence of tumour micro-environment heterogeneity on therapeutic response. Nature, 2013, 501: 346–354
Liu Y, Zhang D, Qiao ZY, et al. A peptide-network weaved nanoplatform with tumor microenvironment responsiveness and deep tissue penetration capability for cancer therapy. Adv Mater, 2015, 27: 5034–5042
Gupta B, Levchenko TS, Torchilin VP. Intracellular delivery of large molecules and small particles by cell-penetrating proteins and peptides. Adv Drug Deliver Rev, 2005, 57: 637–651
Curnis F, Sacchi A, Borgna L, et al. Enhancement of tumor necrosis factor a antitumor immunotherapeutic properties by targeted delivery to aminopeptidase N (CD13). Nat Biotechnol, 2000, 18: 1185–1190
Hoshino Y, Kodama T, Okahata Y, et al. Peptide imprinted polymer nanoparticles: A plastic antibody. J Am Chem Soc, 2008, 130: 15242–15243
Rudra JS, Mishra S, Chong AS, et al. Self-assembled peptide nanofibers raising durable antibody responses against a malaria epitope. Biomaterials, 2012, 33: 6476–6484
Cieslewicz M, Tang J, Yu JL, et al. Targeted delivery of proapoptotic peptides to tumor-associated macrophages improves survival. Proc Natl Acad Sci USA, 2013, 110: 15919–15924
Genové E, Shen C, Zhang S, et al. The effect of functionalized self-assembling peptide scaffolds on human aortic endothelial cell function. Biomaterials, 2005, 26: 3341–3351
Pompano RR, Chen J, Verbus EA, et al. Titrating T-cell epitopes within self-assembled vaccines optimizes CD4+ helper T cell and antibody outputs. Adv Healthcare Mater, 2014, 3: 1898–1908
Luo L, Zhu C, Yin H, et al. Laser immunotherapy in combination with perdurable PD-1 blocking for the treatment of metastatic tumors. ACS Nano, 2018, 12: 7647–7662
Peng J, Yang Q, Xiao Y, et al. Tumor microenvironment responsive drug-dye-peptide nanoassembly for enhanced tumor-targeting, penetration, and photo-chemo-immunotherapy. Adv Funct Mater, 2019, 29: 1900004
Kuai R, Ochyl LJ, Bahjat KS, et al. Designer vaccine nanodiscs for personalized cancer immunotherapy. Nat Mater, 2016, 16: 489–496
Kakwere H, Ingham ES, Allen R, et al. Toward personalized peptide-based cancer nanovaccines: A facile and versatile synthetic approach. Bioconjugate Chem, 2017, 28: 2756–2771
Chen J, Pompano RR, Santiago FW, et al. The use of self-adjuvanting nanofiber vaccines to elicit high-affinity B cell responses to peptide antigens without inflammation. Biomaterials, 2013, 34: 8776–8785
Koutsopoulos S, Unsworth LD, Nagai Y, et al. Controlled release of functional proteins through designer self-assembling peptide nanofiber hydrogel scaffold. Proc Natl Acad Sci USA, 2009, 106: 4623–4628
Li Y, Wang F, Cui H. Peptide-based supramolecular hydrogels for delivery of biologics. Bioeng Translat Med, 2016, 1: 306–322
Koutsopoulos S, Zhang S. Two-layered injectable self-assembling peptide scaffold hydrogels for long-term sustained release of human antibodies. J Control Release, 2012, 160: 451–458
Liu R, Yu M, Yang X, et al. Linear chimeric triblock molecules self-assembled micelles with controllably transformable property to enhance tumor retention for chemo-photodynamic therapy of breast cancer. Adv Funct Mater, 2019, 29: 1808462
Jin P, Sha R, Zhang Y, et al. Blood circulation-prolonging peptides for engineered nanoparticles identified via phage display. Nano Lett, 2019, 19: 1467–1478
Ng CW, Li J, Pu K. Recent progresses in phototherapy-synergized cancer immunotherapy. Adv Funct Mater, 2018, 28: 1804688
Wang D, Wang T, Liu J, et al. Acid-activatable versatile micelleplexes for PD-L1 blockade-enhanced cancer photodynamic immunotherapy. Nano Lett, 2016, 16: 5503–5513
He C, Duan X, Guo N, et al. Core-shell nanoscale coordination polymers combine chemotherapy and photodynamic therapy to potentiate checkpoint blockade cancer immunotherapy. Nat Commun, 2016, 7: 12499
Xu J, Xu L, Wang C, et al. Near-infrared-triggered photodynamic therapy with multitasking upconversion nanoparticles in combination with checkpoint blockade for immunotherapy of colorectal cancer. ACS Nano, 2017, 11: 4463–4474
Yu S, Wang C, Yu J, et al. Injectable bioresponsive gel depot for enhanced immune checkpoint blockade. Adv Mater, 2018, 30: 1801527
Buchbinder EI, Desai A. CTLA-4 and PD-1 pathways. Am J Clin Oncol, 2016, 39: 98–106
Sharma P, Allison JP. The future of immune checkpoint therapy. Science, 2015, 348: 56–61
Gubin MM, Zhang X, Schuster H, et al. Checkpoint blockade cancer immunotherapy targets tumour-specific mutant antigens. Nature, 2014, 515: 577–581
Pardoll DM. The blockade of immune checkpoints in cancer immunotherapy. Nat Rev Cancer, 2012, 12: 252–264
Parry RV, Chemnitz JM, Frauwirth KA, et al. CTLA-4 and PD-1 receptors inhibit T-cell activation by distinct mechanisms. Mol Cellular Biol, 2005, 25: 9543–9553
Schwartz RH. Costimulation of t lymphocytes: The role of CD28, CTLA-4, and B7/BB1 in interleukin-2 production and immunotherapy. Cell, 1992, 71: 1065–1068
Triebel F, Jitsukawa S, Baixeras E, et al. LAG-3, a novel lymphocyte activation gene closely related to CD4. J Exp Med, 1990, 171: 1393–1405
Huang CT, Workman CJ, Flies D, et al. Role of LAG-3 in regulatory T cells. Immunity, 2004, 21: 503–513
Grosso JF, Kelleher CC, Harris TJ, et al. LAG-3 regulates CD8+ T cell accumulation and effector function in murine self- and tumor-tolerance systems. J Clin Invest, 2007, 117: 3383–3392
Monney L, Sabatos CA, Gaglia JL, et al. Th1-specific cell surface protein TIM-3 regulates macrophage activation and severity of an autoimmune disease. Nature, 2002, 415: 536–541
Zhu C, Anderson AC, Schubart A, et al. The TIM-3 ligand galectin-9 negatively regulates t helper type 1 immunity. Nat Immunol, 2005, 6: 1245–1252
Fourcade J, Sun Z, Benallaoua M, et al. Upregulation of Tim-3 and PD-1 expression is associated with tumor antigen-specific CD8+ T cell dysfunction in melanoma patients. J Exp Med, 2010, 207: 2175–2186
Fife BT, Bluestone JA. Control of peripheral T-cell tolerance and autoimmunity via the CTLA-4 and PD-1 pathways. Immunological Rev, 2010, 224: 166–182
Yu X, Harden K, C Gonzalez L, et al. The surface protein TIGIT suppresses T cell activation by promoting the generation of mature immunoregulatory dendritic cells. Nat Immunol, 2008, 10: 48–57
Joller N, Lozano E, Burkett PR, et al. Treg cells expressing the coinhibitory molecule TIGIT selectively inhibit proinflammatory Th1 and Th17 cell responses. Immunity, 2014, 40: 569–581
Walker LSK, Sansom DM. The emerging role of CTLA4 as a cell-extrinsic regulator of T cell responses. Nat Rev Immunol, 2011, 11: 852–863
Walunas TL, Lenschow DJ, Bakker CY, et al. CTLA-4 can function as a negative regulator of T cell activation. Immunity, 1994, 1: 405–413
Leach DR, Krummel MF, Allison JP. Enhancement of antitumor immunity by CTLA-4 blockade. Science, 1996, 271: 1734–1736
Peggs KS, Quezada SA, Chambers CA, et al. Blockade of CTLA-4 on both effector and regulatory T cell compartments contributes to the antitumor activity of anti-CTLA-4 antibodies. J Exp Med, 2009, 206: 1717–1725
Krummel MF, Allison JP. CD28 and CTLA-4 have opposing effects on the response of T cells to stimulation. J Exp Med, 1995, 182: 459–465
Wing K, Onishi Y, Prieto-Martin P, et al. CTLA-4 control over Foxp3+ regulatory T cell function. Science, 2008, 322: 271–275
Zou W, Wolchok JD, Chen L. PD-L1 (B7-H1) and PD-1 pathway blockade for cancer therapy: Mechanisms, response biomarkers, and combinations. Sci Transl Med, 2016, 8: 328rv4
Francisco LM, Sage PT, Sharpe AH. The PD-1 pathway in tolerance and autoimmunity. Immunological Rev, 2010, 236: 219–242
Latchman Y, Wood CR, Chernova T, et al. PD-L2 is a second ligand for PD-1 and inhibits T cell activation. Nat Immunol, 2001, 2: 261–268
Chen L. Co-inhibitory molecules of the B7-CD28 family in the control of T-cell immunity. Nat Rev Immunol, 2004, 4: 336–347
Tumeh PC, Harview CL, Yearley JH, et al. PD-1 blockade induces responses by inhibiting adaptive immune resistance. Nature, 2014, 515: 568–571
Gordon SR, Maute RL, Dulken BW, et al. PD-1 expression by tumour-associated macrophages inhibits phagocytosis and tumour immunity. Nature, 2017, 545: 495–499
Boutros C, Tarhini A, Routier E, et al. Safety profiles of anti-CTLA-4 and anti-PD-1 antibodies alone and in combination. Nat Rev Clin Oncol, 2016, 13: 473–486
Reilly RM, Sandhu J, Alvarez-Diez TM, et al. Problems ofdelivery of monoclonal antibodies. Clin Pharm, 1995, 28: 126–142
Chen T, Li Q, Liu Z, et al. Peptide-based and small synthetic molecule inhibitors on PD-1/PD-L1 pathway: A new choice for immunotherapy? Eur J Medicinal Chem, 2019, 161: 378–398
Chang HN, Liu BY, Qi YK, et al. Blocking of the PD-1/PD-L1 interaction by a D-peptide antagonist for cancer immunotherapy. Angew Chem Int Ed, 2015, 54: 11760–11764
Maute RL, Gordon SR, Mayer AT, et al. Engineering high-affinity PD-1 variants for optimized immunotherapy and immuno-pet imaging. Proc Natl Acad Sci USA, 2015, 112: E6506–E6514
Li C, Zhang N, Zhou J, et al. Peptide blocking of PD-1/PD-L1 interaction for cancer immunotherapy. Cancer Immunol Res, 2018, 6: 178–188
Munir Ahmad S, Martinenaite E, Hansen M, et al. PD-L1 peptide co-stimulation increases immunogenicity ofa dendritic cell-based cancer vaccine. Oncoimmunology, 2016, 5: e1202391
Magiera-Mularz K, Skalniak L, Zak KM, et al. Bioactive macro-cyclic inhibitors of the PD-1/PD-L1 immune checkpoint. Angew Chem Int Ed, 2017, 56: 13732–13735
Molino NM, Anderson AKL, Nelson EL, et al. Biomimetic protein nanoparticles facilitate enhanced dendritic cell activation and cross-presentation. ACS Nano, 2013, 7: 9743–9752
Black M, Trent A, Kostenko Y, et al. Self-assembled peptide amphiphile micelles containing a cytotoxic T-cell epitope promote a protective immune response in vivo. Adv Mater, 2012, 24: 3845–3849
Qian Y, Jin H, Qiao S, et al. Targeting dendritic cells in lymph node with an antigen peptide-based nanovaccine for cancer immunotherapy. Biomaterials, 2016, 98: 171–183
Jäger E, Chen YT, Drijfhout JW, et al. Simultaneous humoral and cellular immune response against cancer-testis antigen NY-ESO-1: definition of human histocompatibility leukocyte antigen (HLA)-A2-binding peptide epitopes. J Exp Med, 1998, 187: 265–270
Neek M, Tucker JA, Kim TI, et al. Co-delivery of human cancer-testis antigens with adjuvant in protein nanoparticles induces higher cell-mediated immune responses. Biomaterials, 2018, 156: 194–203
Jäeger E, Bernhard H, Romero P, et al. Generation of cytotoxic T-cell responses with synthetic melanoma-associated peptides in vivo: Implications for tumor vaccines with melanoma-associated antigens. Int J Cancer, 1996, 66: 162–169
Jäger E, Höhn H, Necker A, et al. Peptide-specific CD8+ T-cell evolution in vivo: Response to peptide vaccination with Melan-A/MART-1. Int J Cancer, 2002, 98: 376–388
Slingluff, CLJ, Yamshchikov G, Neese P, et al. Phase I trial of a melanoma vaccine with gp100(280–288) peptide and tetanus helper peptide in adjuvant: Immunologic and clinical outcomes. Clin Cancer Res, 2001, 7: 3012–3024
Tarhini AA, Leng S, Moschos SJ, et al. Safety and immunogenicity of vaccination with MART-1 (26–35, 27L), gp100 (209–217, 210M), and tyrosinase (368–376, 370D) in adjuvant with PF-3512676 and GM-CSF in metastatic melanoma. J ImmunoTher, 2012, 35: 359–366
Guo Y, Wang D, Song Q, et al. Erythrocyte membrane-enveloped polymeric nanoparticles as nanovaccine for induction of anti-tumor immunity against melanoma. ACS Nano, 2015, 9: 6918–6933
Wakabayashi R, Sakuragi M, Kozaka S, et al. Solid-in-oil peptide nanocarriers for transcutaneous cancer vaccine delivery against melanoma. Mol Pharm, 2018, 15: 955–961
Haugen A. Cancer, genes and gender. Carcinogenesis, 2002, 23: 1089
Peoples GE, Holmes JP, Hueman MT, et al. Combined clinical trial results of a HER2/neu (E75) vaccine for the prevention of recurrence in high-risk breast cancer patients: U.S. military cancer institute clinical trials group study I-01 and I-02. Clinical Cancer Res, 2008, 14: 797–803
Villa CH, Dao T, Ahearn I, et al. Single-walled carbon nanotubes deliver peptide antigen into dendritic cells and enhance IgG responses to tumor-associated antigens. ACS Nano, 2011, 5: 5300–5311
Ressing ME, Sette A, Brandt RMP, et al. Human CTL epitopes encoded by human papillomavirus type 16 E6 and E7 identified through in vivo and in vitro immunogenicity studies of HLA-A*0201-binding peptides. J Immunol, 1995, 154: 5934–5943
van Driel WJ, Ressing ME, Kenter GG, et al. Vaccination with HPV16 peptides of patients with advanced cervical carcinoma: clinical evaluation of a phase I–II trial. Eur J Cancer, 1999, 35: 946–952
Liu TY, Hussein WM, Jia Z, et al. Self-adjuvanting polymer-peptide conjugates as therapeutic vaccine candidates against cervical cancer. Biomacromolecules, 2013, 14: 2798–2806
Liu TY, Hussein WM, Giddam AK, et al. Polyacrylate-based delivery system for self-adjuvanting anticancer peptide vaccine. J Med Chem, 2015, 58: 888–896
Tindle RW, Croft S, Herd K, et al. A vaccine conjugate of ‘ISCAR’ immunocarrier and peptide epitopes of the E7 cervical cancer-associated protein of human papillomavirus type 16 elicits specific Th1- and Th2-type responses in immunized mice in the absence of oil-based adjuvants. Clin Exp Immunol, 1995, 101: 265–271
Tsoras AN, Champion JA. Cross-linked peptide nanoclusters for delivery of oncofetal antigen as a cancer vaccine. Bioconj Chem, 2018, 29: 776–785
Rudra JS, Tian YF, Jung JP, et al. A self-assembling peptide acting as an immune adjuvant. Proc Natl Acad Sci USA, 2010, 107: 622–627
Rudra JS, Sun T, Bird KC, et al. Modulating adaptive immune responses to peptide self-assemblies. ACS Nano, 2012, 6: 1557–1564
Luo Z, Wu Q, Yang C, et al. A powerful CD8+ T-cell stimulating D-tetra-peptide hydrogel as a very promising vaccine adjuvant. Adv Mater, 2017, 29: 1601776
Yang C, Shi F, Li C, et al. Single dose of protein vaccine with peptide nanofibers as adjuvants elicits long-lasting antibody titer. ACS Biomater Sci Eng, 2017, 4: 2000–2006
Wang Z, Liang C, Shi F, et al. Cancer vaccines using supramolecular hydrogels of NSAID-modified peptides as adjuvants abolish tumorigenesis. Nanoscale, 2017, 9: 14058–14064
Melero I, Gaudernack G, Gerritsen W, et al. Therapeutic vaccines for cancer: An overview of clinical trials. Nat Rev Clin Oncol, 2014, 11: 509–524
van Duin D, Medzhitov R, Shaw AC. Triggering TLR signaling in vaccination. Trends Immunol, 2006, 27: 49–55
Parkin J, Cohen B. An overview of the immune system. Lancet, 2001, 357: 1777–1789
Zheng Y, Wen Y, George AM, et al. A peptide-based material platform for displaying antibodies to engage T cells. Biomaterials, 2011, 32: 249–257
König R, Huang LY, Germain RN. MHC class II interaction with CD4 mediated by a region analogous to the MHC class I binding site for CD8. Nature, 1992, 356: 796–798
Plotkin SA. Vaccines: Past, present and future. Nat Med, 2005, 11: S5–S11
Matsumura M, Fremont DH, Peterson PA, et al. Emerging principles for the recognition of peptide antigens by MHC class I molecules. Science, 1992, 257: 927–934
Skwarczynski M, Toth I. Recent advances in peptide-based subunit nanovaccines. Nanomedicine, 2014, 9: 2657–2669
Zhu G, Zhang F, Ni Q, et al. Efficient nanovaccine delivery in cancer immunotherapy. ACS Nano, 2017, 11: 2387–2392
Rosenberg SA, Yang JC, Schwartzentruber DJ, et al. Immunologic and therapeutic evaluation of a synthetic peptide vaccine for the treatment of patients with metastatic melanoma. Nat Med, 1998, 4: 321–327
Bakker AB, Schreurs MW, de Boer AJ, et al. Melanocyte lineage-specific antigen gp100 is recognized by melanoma- derived tumor-infiltrating lymphocytes. J Exp Med, 1994, 179: 1005–1009
Speiser D, Liénard D, Pittet M, et al. In vivo activation of melanoma-specific CD8+ T cells by endogenous tumor antigen and peptide vaccines. A comparison to virus-specific T cells. Eur J Immunol, 2002, 32: 731
Skakuj K, Wang S, Qin L, et al. Conjugation chemistry-dependent T-cell activation with spherical nucleic acids. J Am Chem Soc, 2018, 140: 1227–1230
Xie J, Yang C, Liu Q, et al. Encapsulation of hydrophilic and hydrophobic peptides into hollow mesoporous silica nano-particles for enhancement of antitumor immune response. Small, 2017, 13: 1701741
Tao Y, Zhang Y, Ju E, et al. Gold nanocluster-based vaccines for dual-delivery of antigens and immunostimulatory oligonucleotides. Nanoscale, 2015, 7: 12419–12426
Gendler SJ. MUC1, the renaissance molecule. J Mammary Gland Biol Neoplasia, 2001, 6: 339–353
Huang ZH, Shi L, Ma JW, et al. A totally synthetic, self-assembling, adjuvant-free MUC1 glycopeptide vaccine for cancer therapy. J Am Chem Soc, 2012, 134: 8730–8733
Liu Y, Wang Y, Yu F, et al. Potentiating the immune response of MUC1-based antitumor vaccines using a peptide-based nano-vector as a promising vaccine adjuvant. Chem Commun, 2017, 53: 9486–9489
de Faria PCB, dos Santos LI, Coelho JP, et al. Oxidized multi-walled carbon nanotubes as antigen delivery system to promote superior CD8+ T cell response and protection against cancer. Nano Lett, 2014, 14: 5458–5470
Rad-Malekshahi M, Fransen MF, Krawczyk M, et al. Self-assembling peptide epitopes as novel platform for anticancer vaccination. Mol Pharm, 2017, 14: 1482–1493
Feltkamp MCW, Vreugdenhil GR, Vierboom MPM, et al. Cytotoxic T lymphocytes raised against a subdominant epitope offered as a synthetic peptide eradicate human papillomavirus type 16-induced tumors. Eur J Immunol, 1995, 25: 2638–2642
Acar H, Srivastava S, Chung EJ, et al. Self-assembling peptide-based building blocks in medical applications. Adv Drug Deliver Rev, 2017, 110–111: 65–79
Reed SG, Orr MT, Fox CB. Key roles of adjuvants in modern vaccines. Nat Med, 2013, 19: 1597–1608
Appavu R, Chesson CB, Koyfman AY, et al. Enhancing the magnitude of antibody responses through biomaterial stereochemistry. ACS Biomater Sci Eng, 2015, 1: 601–609
Wang H, Luo Z, Wang Y, et al. Enzyme-catalyzed formation of supramolecular hydrogels as promising vaccine adjuvants. Adv Funct Mater, 2016, 26: 1822–1829
Gazit E. Self-assembled peptide nanostructures: The design of molecular building blocks and their technological utilization. Chem Soc Rev, 2007, 36: 1263–1269
Hendricks MP, Sato K, Palmer LC, et al. Supramolecular assembly of peptide amphiphiles. Acc Chem Res, 2017, 50: 2440–2448
Cui H, Muraoka T, Cheetham AG, et al. Self-assembly of giant peptide nanobelts. Nano Lett, 2009, 9: 945–951
Hamley IW. Peptide fibrillization. Angew Chem Int Ed, 2007, 46: 8128–8147
De Santis E, Ryadnov MG. Peptide self-assembly for nanomaterials: The old new kid on the block. Chem Soc Rev, 2015, 44: 8288–8300
Hu X, Hu J, Tian J, et al. Polyprodrug amphiphiles: Hierarchical assemblies for shape-regulated cellular internalization, trafficking, and drug delivery. J Am Chem Soc, 2013, 135: 17617–17629
Hudalla GA, Sun T, Gasiorowski JZ, et al. Gradated assembly of multiple proteins into supramolecular nanomaterials. Nat Mater, 2014, 13: 829–836
Demaria S, Kawashima N, Yang AM, et al. Immune-mediated inhibition of metastases after treatment with local radiation and CTLA-4 blockade in a mouse model of breast cancer. Clin Cancer Res, 2005, 11: 728–734
Sharabi AB, Lim M, DeWeese TL, et al. Radiation and checkpoint blockade immunotherapy: Radiosensitisation and potential mechanisms of synergy. Lancet Oncol, 2015, 16: e498–e509
Fan W, Yung B, Huang P, et al. Nanotechnology for multimodal synergistic cancer therapy. Chem Rev, 2017, 117: 13566–13638
Sharma P, Allison JP. Immune checkpoint targeting in cancer therapy: Toward combination strategies with curative potential. Cell, 2015, 161: 205–214
Mahoney KM, Rennert PD, Freeman GJ. Combination cancer immunotherapy and new immunomodulatory targets. Nat Rev Drug Discov, 2015, 14: 561–584
Cheng K, Ding Y, Zhao Y, et al. Sequentially responsive therapeutic peptide assembling nanoparticles for dual-targeted cancer immunotherapy. Nano Lett, 2018, 18: 3250–3258
Wang T, Wang D, Yu H, et al. A cancer vaccine-mediated postoperative immunotherapy for recurrent and metastatic tumors. Nat Commun, 2018, 9: 1532
Yang P, Song H, Qin Y, et al. Engineering dendritic-cell-based vaccines and PD-1 blockade in self-assembled peptide nanofibrous hydrogel to amplify antitumor T-cell immunity. Nano Lett, 2018, 18: 4377–4385
Song W, Kuang J, Li CX, et al. Enhanced immunotherapy based on photodynamic therapy for both primary and lung metastasis tumor eradication. ACS Nano, 2018, 12: 1978–1989
Kuang J, Song W, Yin J, et al. iRGD modified chemo-immunotherapeutic nanoparticles for enhanced immunotherapy against glioblastoma. Adv Funct Mater, 2018, 28: 1800025
Skwarczynski M, Toth I. Peptide-based synthetic vaccines. Chem Sci, 2016, 7: 842–854
Parmiani G, Russo V, Maccalli C, et al. Peptide-based vaccines for cancer therapy. Human Vaccines Immunotherapeutics, 2014, 10: 3175–3178
Zacco E, Anish C, Martin CE, et al. A self-assembling peptide scaffold for the multivalent presentation of antigens. Biomacromolecules, 2015, 16: 2188–2197
Eskandari S, Guerin T, Toth I, et al. Recent advances in self-assembled peptides: Implications for targeted drug delivery and vaccine engineering. Adv Drug Deliver Rev, 2017, 110–111: 169–187
Acknowledgements
This work was supported by the National Natural Science Foundation of China (21774065), the Fundamental Research Funds for the Central Universities, and the Natural Science Foundation of Tianjin (18JCQNJC14100).
Author information
Authors and Affiliations
Contributions
Li M prepared the manuscript under the guidance of Yu Z. Li M and Zhao X designed and prepared the figures. Yu Z and Dai J revised the manuscript. All authors contributed to the general discussion and revision of the manuscript.
Corresponding author
Additional information
Conflict of interest
The authors declare no conflict of interest.
Mingming Li received her Master degree in chemical engineering from China University of Petroleum (Beijing) in 2018. Currently she is a PhD candidate under the supervision of Prof. Zhilin Yu at the Institute of Polymer Chemistry of Nankai University. Her current research interest lies in the field of stimulus-responsive peptide self-assembly and the biomaterials including drug delivery and disease therapy.
Zhilin Yu was awarded his PhD degree under the supervision of Prof. Stefan Hecht at the Humboldt-Universität zu Berlin in 2013. He conducted his postdoctoral training with Prof. Samuel I. Stupp at Northwestern University focusing on self-assembly of peptide-based amphiphilic molecules. In 2017, he started his independent career at the Institute of Polymer Science of Nankai University. His current research interests focus on the self-assembly of peptides into dynamic nanostructures and their broad applications as biomaterials including disease diagnosis and therapy.
Rights and permissions
About this article
Cite this article
Li, M., Zhao, X., Dai, J. et al. Peptide therapeutics and assemblies for cancer immunotherapy. Sci. China Mater. 62, 1759–1781 (2019). https://doi.org/10.1007/s40843-019-9451-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40843-019-9451-7