Abstract
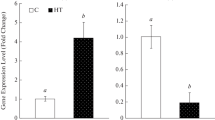
DNA methylation is an important part of epigenetics, which can regulate gene expression, cell differentiation, growth and development in plants. In this study, the salt-sensitive variety Chuanqiao No.2 of Tartary buckwheat was used as experimental material. Different concentrations of 5-azaC were used to infect Tartary buckwheat leaves and combined with gene expression analysis, it is determined that the appropriate concentration of 5-azaC treatment is 100 µM. DNA methylation often occurs in the gene promoter region, causing gene transcription changes, and thus regulating the response to salt stress. By CpG island prediction and promoter region methylation site analysis of FtNHX1 gene, it was found that FtNHX1 gene expression was significantly regulated by DNA methylation. Moreover, after salt stress for 6 h, DNA methylation rate of FtNHX1 gene was increased. This increase of DNA methylation rate in a short time may inhibit over-expression of FtNHX1 gene.






Similar content being viewed by others
References
Bonafaccia G, Marocchini M, Kreft I (2003) Composition and technological properties of the flour and bran from common and tartary buckwheat. Food Chem 80(1):9–15. https://doi.org/10.1016/S0308-8146(02)00228-5
Chen F, Wang ZC (2011) Effects of 5-azaC on development and DNA methylation in wheat. J Henan Univ 41(1):61–66. https://doi.org/10.3969/j.issn.1003-4978.2011.01.013
Chen FS (2017) Investigating the correlation of CpG density and methylation. Dalian Med University
Choi Y, Gehring M, Johnson L, Hannon M, Harada JJ, Goldberg RB, Jacobsen SE, Fischer RL (2002) Demeter, a DNA glycosylase domain protein, is required for endosperm gene imprinting and seed viability in Arabidopsis. Cell 110(1):33–42. https://doi.org/10.1016/S0092-8674(02)00807-3
Choi J, Lyons DB, Kim MY, Moore JD, Zilberman D (2020) DNA methylation and histone H1 jointly repress transposable elements and aberrant intragenic transcripts. Mol Cell 77(2):310–323. https://doi.org/10.1016/j.molcel.2019.10.011
Cokus SJ, Feng S, Zhang X, Chen Z, Merriman B, Haudenschild CD, Pradhan S, Nelson SF, Pellegrini M, Jacobsen SE (2008) Shotgun bisulphite sequencing of the Arabidopsis genome reveals DNA methylation patterning. Nature 452(7184):215–219. https://doi.org/10.1038/nature06745
Cortellino S, Xu J, Sannai M, Moore R, Caretti E, Cigliano A, Coz ML, Devarajan K, Wessels A, Soprano D, Abramowitz LK, Bartolomei MS, Rambow F, Bassi MR, Bruno T, Fanciulli M, Renner C, Klein-Szanto AJ, Matsumoto Y, Kobi D, Davidson I, Alberti C, Larue L, Bellacosa A (2011) Thymine DNA glycosylase is essential for active DNA demethylation by linked deamination-base excision repair. Cell 146(1):67–79. https://doi.org/10.1016/j.cell.2011.06.020
Fabjan N, Rode J, Kosir IJ, Wang Z, Kreft I (2003) Tartary buckwheat (Fagopyrum tataricum Gaertn.) as a source of dietary rutin and quercitin. J Agric Food Chem 51(22):6452–6455. https://doi.org/10.1021/jf034543e
Fu L, Xia Y, He J, Liu X, Chen X, Wang Y, Ding Y (2012) Data analysis and its analytical softs application on DNA methylation in tumor research. Chongqing Med. https://doi.org/10.3969/j.issn.1671-8348.2012.17.018
Galbally M, Watson SJ, van IJzendoorn M, Saffery R, Ryan J, de Kloet ER, Oberlander TF, Lappas M, Lewis AJ (2020) The role of glucocorticoid and mineralocorticoid receptor DNA methylation in antenatal depression and infant stress regulation. Psychoneuroendocrino 115:104611. https://doi.org/10.1016/j.psyneuen.2020.104611
Gehring M, Jin HH, Hsieh TF, Penterman J, Choi Y, Harada JJ, Goldberg RB, Fischer RL (2006) DEMETER DNA glycosylase establishes MEDEA polycomb gene self-imprinting by allele-specific demethylation. Cell 124:495–506. https://doi.org/10.1016/j.cell.2005.12.034
Hughes AL, Kelley JR, Klose RJ (2020) Understanding the interplay between CpG island-associated gene promoters and H3K4 methylation. BBA-Gene Regul Mech 1863(8):194567. https://doi.org/10.1016/j.bbagrm.2020.194567
Kim JK, Kim SK (2006) Physiochemical properties of buckwheat starches from different areas. Korean J Food Sci Technol 36(4):598–603. https://doi.org/10.1134/S0020168513080165
Kinoshita T, Miura A, Choi Y, Kinoshital Y, Cao X, Jacobsen SE, Fischer RL, Kakutani T (2004) One-way control of FWA imprintingin Arabidopsis endosperm by DNA methylation. Science 303(5657):521–523. https://doi.org/10.1126/science.1089835
Lang Z, Wang Y, Tang K, Tang D, Datsenka T, Cheng J, Zhang Y, Handa AK, Zhu JK (2017) Critical roles of DNA demethylation in the activation of ripening-induced genes and inhibition of ripening-repressed genes in tomato fruit. P Natl Acad Sci USA 114(22):E4511–E4519. https://doi.org/10.1073/pnas.1705233114
Li LC (2007) Designing PCR primer for DNA methylation mapping. Methods Mol Biol. https://doi.org/10.1007/978-1-59745-528-2_19
Liu X, Song J, Zhang Y, Hou L, Yu Y, Zhao F, Liu C, Dong C, Yang H (2017) Cloning and expression analysis of FtNHX1 in Tartary buckwheat. Acta Agric Boreali-Sin 32(4):49–54. https://doi.org/10.7668/hbnxb.2017.04.008
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2-△△CT method. Methods 25:402–408. https://doi.org/10.1006/meth.2001.1262
McCord JM (2000) The evolution of free radicals and oxidative stress. Am J Med 108(8):652–659. https://doi.org/10.1016/S0002-9343(00)00412-5
Nass R, Cunningham KW, Rao R (1997) Intracellular sequestration of sodium by a novel Na+/H+ exchanger in yeast is enhanced by mutations in the plasma membrane H+-ATPase. J Biol Chem 272(42):26145–26152. https://doi.org/10.1074/JBC.272.42.26145
Peng C, Sui Z, Zhou W, Hu Y, Mi P, Jiang M, Li X, Ruan X (2018) Analysis of DNA methylation of Gracilariopsis lemaneiformis under temperature stress using the methylation sensitive amplification polymorphism (MSAP) technique. J Ocean Univ China 17(3):623–631. https://doi.org/10.1007/s11802-018-3426-9
Penterman J, Zilberman D, Huh JH, Ballinger T, Henikoff S, Fischer RL (2007) DNA demethylation in the Arabidopsis genome. P Natl Acad Sci USA 104(16):6752–6757. https://doi.org/10.1073/pnas.0701861104
Robertson KD (2005) DNA methylation and human disease. Nat Rev Genet 6(8):597–610. https://doi.org/10.1038/nrg1655
Stenz L, Schechter DS, Serpa SR, Paoloni-Giacobino A (2018) Intergenerational transmission of DNA methylation signatures associated with early life stress. Curr Genomics 19(8):665–675. https://doi.org/10.2174/1389202919666171229145656
Su D (2012) Studies on physiological and biochemical responses of Halocnermum strobilaceum to salt stress and the cloning of its betaine aldehyde dehydrogenase gene. Gansu Agric Univ
Sun XF, Zheng QS, Liu YL (2000) Salinity injury to germination and growth of cotton (Gossypium hirsutum L.) at emergence and seedling stages. J Plant Resour Environ 9(3):22–25. https://doi.org/10.3969/j.issn.1674-7895.2000.03.007
Wang XA, Yang HB, Qiu NW (2010) Application of paired-sample design in plant physiology experiment. Plant Physiol Commun 46:161–164
Wang D, Zhao Y, Zhang F (2020) Screening and identification of salt-tolerant plant growth-promoting bacteria and its promotion effect on wheat seedling under salt stress. J Tritic Crops 40(1):110–117. https://doi.org/10.7606/j.issn.1009-1041.2020.01.13
Wu Y (2017) Effects of 5-azaC on flowering time and physiological indexes of Rhododendron occidentalis. Fujian Agric Forest Univ
Yang Q, Wu M, Wang P, Kang J, Zhou X (2005) Cloning and expression analysis of a vacuolar Na+/H+ antiporter gene from alfalfa. DNA Seq 16(5):352–357. https://doi.org/10.1080/10425170500272742
Yang S, Ji J, Wang G (2006) Effects of salt stress on plants and the mechanism of salt resistance in plants. World Sci Technol Res Dev 28(4):70–76
Zeng Y, Wu N, Wang NN (2018) Research progress on gene expression variation in rice under heavy metal stress. Mol Pant Beed 16(24):8179–8184
Zhang X, Jacobsen SE (2006) Genetic analyses of DNA methyltransferases in Arabidopsis thaliana. Cold Spr Harb Symp Quant Biol 71:439–447. https://doi.org/10.1101/sqb.2006.71.047
Zhang L, Li X, Ma B, Gao Q, Du H, Han Y, Li Y, Cao Y, Qi M, Zhu Y, Lu H, Ma M, Liu L, Zhou J, Nan C, Qin Y, Wang J, Cui L, Liu H, Liang C, Qiao Z (2017) The Tartary buckwheat genome provides insights into rutin biosynthesis and abiotic stress tolerance. Mol Plant 10(9):1224–1237. https://doi.org/10.1016/j.molp.2017.08.013
Zhang H, Lang Z, Zhu JK (2018) Dynamics and function of DNA methylation in plants. Nat Rev Mol Cell Biol 19(8):489–506. https://doi.org/10.1038/s41580-018-0016-z
Zhang F, Zhang Y, Yuan F, Liu Y, Chen Y, Yilidana S, Liu L, Li H (2019a) The clinical significance and methylation level of RASSF1A and survivin promoter CpG island in Kazakh esophageal squa-mous cell carcinoma. J Shanxi Med Univ 50(9):1210–1213
Zhang Y, Ruan Y, Zhao C, Xue M, Li B, Wang J, Liu Y, Wang K, Wang H (2019b) Analysis of genomic DNA methylation level in foxtail millet by Methylation Sensitive Amplified Polymorphism. J Biol Eng 35(2):263–269
Acknowledgements
We acknowledge the financial support from the National Natural Science Foundation of China (Grant No. 31371552). The research material Chuanqiao No.2 was furnished by the Research Station of Alpine Crop, Department of Agriculture and Science in Liangshan State, Sichuan Province, China.
Author information
Authors and Affiliations
Contributions
WYQ analyzed the DNA methylation and wrote the manuscript. SJN determined the relative expression of FtNHX1 gene. YHB designed the experiment and revised the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no competing interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Wang, YQ., Song, JN. & Yang, HB. DNA methylation regulates the expression of salt tolerance gene FtNHX1 in Tartary buckwheat. Theor. Exp. Plant Physiol. 34, 185–195 (2022). https://doi.org/10.1007/s40626-022-00241-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40626-022-00241-5