Abstract
Purpose
Ipilimumab is a human monoclonal antibody against cytotoxic T-lymphocyte antigen 4 available as an immunotherapy mainly for advanced melanoma. It induces an activation of T cells, resulting in an immune-mediated anti-tumor response and also immune-related adverse events, including hypophysitis. The aim of this review is to identify and discuss features concerning ipilimumab-induced hypophysitis (IIH).
Design
A MEDLINE research of all years of publication of IIH was conducted. We gathered information regarding clinical, radiologic and laboratory features of 71 cases recorded in the literature.
Results
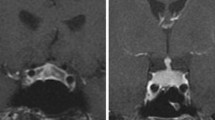
In our review, IIH was more frequent among older and male patients. Fatigue and headache were the most frequent initial clinical manifestations of IIH and enlargement of the pituitary gland at MRI was present in the majority of patients. Those who received more than 3 cycles of ipilimumab had more fatigue (p = 0.04) and arthritis (p = 0.04). Adrenal insufficiency was more prevalent in men (p = 0.007). Glucocorticoid therapy and hormone replacement were required in most patients and pituitary function recovery was uncommon. Low prolactin at diagnosis tended to predict permanent pituitary dysfunction (p = 0.07).
Conclusion
Hypopituitarism as a consequence of IIH, if not promptly recognized, can lead to potentially fatal events, such as adrenal insufficiency. IIH can be easily managed with glucocorticoids and hormonal replacement; therefore, physicians should be familiar with the key aspects of this condition. More studies to develop screening protocols and therapeutic intervention algorithms should be performed to decrease morbidity related to IIH.

Similar content being viewed by others
References
Attia P, Phan GQ, Maker AV, Robinson MR, Quezado MM, Yang JC, Sherry RM, Topalian SL, Kammula US, Royal RE, Restifo NP, Haworth LR, Levy C, Mavroukakis SA, Nichol G, Yellin MJ, Rosenberg SA (2005) Autoimmunity correlates with tumor regression in patients with metastatic melanoma treated with anti-cytotoxic T-lymphocyte antigen-4. J Clin Oncol Off J Am Soc Clin Oncol 23(25):6043–6053. doi:10.1200/JCO.2005.06.205
Phan GQ, Yang JC, Sherry RM, Hwu P, Topalian SL, Schwartzentruber DJ, Restifo NP, Haworth LR, Seipp CA, Freezer LJ, Morton KE, Mavroukakis SA, Duray PH, Steinberg SM, Allison JP, Davis TA, Rosenberg SA (2003) Cancer regression and autoimmunity induced by cytotoxic T lymphocyte-associated antigen 4 blockade in patients with metastatic melanoma. Proc Natl Acad Sci USA 100(14):8372–8377. doi:10.1073/pnas.1533209100
Hodi FS, O’Day SJ, McDermott DF, Weber RW, Sosman JA, Haanen JB, Gonzalez R, Robert C, Schadendorf D, Hassel JC, Akerley W, van den Eertwegh AJ, Lutzky J, Lorigan P, Vaubel JM, Linette GP, Hogg D, Ottensmeier CH, Lebbe C, Peschel C, Quirt I, Clark JI, Wolchok JD, Weber JS, Tian J, Yellin MJ, Nichol GM, Hoos A, Urba WJ (2010) Improved survival with ipilimumab in patients with metastatic melanoma. New Engl J Med 363(8):711–723. doi:10.1056/NEJMoa1003466
Faje AT, Sullivan R, Lawrence D, Tritos NA, Fadden R, Klibanski A, Nachtigall L (2014) Ipilimumab-induced hypophysitis: a detailed longitudinal analysis in a large cohort of patients with metastatic melanoma. J Clin Endocrinol Metab doi:10.1210/jc.2014-2306
Torino F, Barnabei A, De Vecchis L, Salvatori R, Corsello SM (2012) Hypophysitis induced by monoclonal antibodies to cytotoxic T lymphocyte antigen 4: challenges from a new cause of a rare disease. Oncologist 17(4):525–535. doi:10.1634/theoncologist.2011-0404
Torino F, Barnabei A, Paragliola RM, Marchetti P, Salvatori R, Corsello SM (2013) Endocrine side-effects of anti-cancer drugs: mAbs and pituitary dysfunction: clinical evidence and pathogenic hypotheses. Eur J Endocrinol Eur Federation Endocr Soc 169(6):R153–R164. doi:10.1530/eje-13-0434
Corsello SM, Barnabei A, Marchetti P, De Vecchis L, Salvatori R, Torino F (2013) Endocrine side effects induced by immune checkpoint inhibitors. J Clin Endocrinol metabolism 98(4):1361–1375. doi:10.1210/jc.2012-4075
Dillard T, Yedinak CG, Alumkal J, Fleseriu M (2010) Anti-CTLA-4 antibody therapy associated autoimmune hypophysitis: serious immune related adverse events across a spectrum of cancer subtypes. Pituitary 13(1):29–38. doi:10.1007/s11102-009-0193-z
Di Giacomo AM, Biagioli M, Maio M (2010) The emerging toxicity profiles of anti-CTLA-4 antibodies across clinical indications. Semin Oncol 37(5):499–507. doi:10.1053/j.seminoncol.2010.09.007
Patel SP, Woodman SE (2011) Profile of ipilimumab and its role in the treatment of metastatic melanoma. Drug design, development and therapy 5:489–495. doi:10.2147/DDDT.S10945
Della Vittoria Scarpati G, Fusciello C, Perri F, Sabbatino F, Ferrone S, Carlomagno C, Pepe S (2014) Ipilimumab in the treatment of metastatic melanoma: management of adverse events. OncoTargets and therapy 7:203–209. doi:10.2147/ott.s57335
Tarhini A (2013) Immune-mediated adverse events associated with ipilimumab ctla-4 blockade therapy: the underlying mechanisms and clinical management. Scientifica 2013:857519. doi:10.1155/2013/857519
Kaehler KC, Piel S, Livingstone E, Schilling B, Hauschild A, Schadendorf D (2010) Update on immunologic therapy with anti-CTLA-4 antibodies in melanoma: identification of clinical and biological response patterns, immune-related adverse events, and their management. Semin Oncol 37(5):485–498. doi:10.1053/j.seminoncol.2010.09.003
Boasberg P, Hamid O, O’Day S (2010) Ipilimumab: unleashing the power of the immune system through CTLA-4 blockade. Semin Oncol 37(5):440–449. doi:10.1053/j.seminoncol.2010.09.004
Yang JC, Hughes M, Kammula U, Royal R, Sherry RM, Topalian SL, Suri KB, Levy C, Allen T, Mavroukakis S, Lowy I, White DE, Rosenberg SA (2007) Ipilimumab (anti-CTLA4 antibody) causes regression of metastatic renal cell cancer associated with enteritis and hypophysitis. J Immunother 30(8):825–830. doi:10.1097/CJI.0b013e318156e47e
Maker AV, Yang JC, Sherry RM, Topalian SL, Kammula US, Royal RE, Hughes M, Yellin MJ, Haworth LR, Levy C, Allen T, Mavroukakis SA, Attia P, Rosenberg SA (2006) Intrapatient dose escalation of anti-CTLA-4 antibody in patients with metastatic melanoma. J Immunother 29(4):455-463. doi:10.1097/01.cji.0000208259.73167.58
Blansfield JA, Beck KE, Tran K, Yang JC, Hughes MS, Kammula US, Royal RE, Topalian SL, Haworth LR, Levy C, Rosenberg SA, Sherry RM (2005) Cytotoxic T-lymphocyte-associated antigen-4 blockage can induce autoimmune hypophysitis in patients with metastatic melanoma and renal cancer. J Immunother 28(6):593–598
Marlier J, Cocquyt V, Brochez L, Van Belle S, Kruse V (2014) Ipilimumab, not just another anti-cancer therapy: hypophysitis as side effect illustrated by four case-reports. Endocrine. doi:10.1007/s12020-014-0199-9
Iwama S, De Remigis A, Callahan MK, Slovin SF, Wolchok JD, Caturegli P (2014) Pituitary expression of CTLA-4 mediates hypophysitis secondary to administration of CTLA-4 blocking antibody. Science translational medicine 6(230):230ra245. doi:10.1126/scitranslmed.3008002
Caturegli P, Newschaffer C, Olivi A, Pomper MG, Burger PC, Rose NR (2005) Autoimmune hypophysitis. Endocr Rev 26(5):599–614. doi:10.1210/er.2004-0011
Min L, Vaidya A, Becker C (2012) Association of ipilimumab therapy for advanced melanoma with secondary adrenal insufficiency: a case series. Endocr Pract Off J Am Coll Endocrinol Am Assoc Clin Endocrinol 18(3):351–355. doi:10.4158/ep11273.or
Weber JS, Kahler KC, Hauschild A (2012) Management of immune-related adverse events and kinetics of response with ipilimumab. J Clin Oncol Off J Am Soc Clin Oncol 30(21):2691–2697. doi:10.1200/jco.2012.41.6750
Bronstein Y, Ng CS, Hwu P, Hwu WJ (2011) Radiologic manifestations of immune-related adverse events in patients with metastatic melanoma undergoing anti-CTLA-4 antibody therapy. AJR Am J Roentgenol 197 (6):w992–w1000. doi:10.2214/ajr.10.6198
Fassett DR, Couldwell WT (2004) Metastases to the pituitary gland. Neurosurg Focus 16(4):E8
Wang YY, Norris A, du Plessis D, Gnanalingham KK (2011) Melanoma of the sellar region. J Clin Neurosci Off J Neurosurg Soc Australas 18(1):154–156. doi:10.1016/j.jocn.2010.07.111
Chodakiewitz Y, Brown S, Boxerman JL, Brody JM, Rogg JM (2014) Ipilimumab treatment associated pituitary hypophysitis: clinical presentation and imaging diagnosis. Clin Neurol Neurosurg 125:125–130. doi:10.1016/j.clineuro.2014.06.011
Glezer A, Bronstein MD (2012) Pituitary autoimmune disease: nuances in clinical presentation. Endocrine 42(1):74–79. doi:10.1007/s12020-012-9654-7
Carpinteri R, Patelli I, Casanueva FF, Giustina A (2009) Pituitary tumours: inflammatory and granulomatous expansive lesions of the pituitary. Best Pract Res Clin Endocrinol Metab 23(5):639–650. doi:10.1016/j.beem.2009.05.009
Downey SG, Klapper JA, Smith FO, Yang JC, Sherry RM, Royal RE, Kammula US, Hughes MS, Allen TE, Levy CL, Yellin M, Nichol G, White DE, Steinberg SM, Rosenberg SA (2007) Prognostic factors related to clinical response in patients with metastatic melanoma treated by CTL-associated antigen-4 blockade. Clin Cancer Res Off J Am Assoc Cancer Res 13(22 Pt 1):6681–6688. doi:10.1158/1078-0432.CCR-07-0187
Abe T (2008) Lymphocytic infundibulo-neurohypophysitis and infundibulo-panhypophysitis regarded as lymphocytic hypophysitis variant. Brain Tumor Pathol 25(2):59–66. doi:10.1007/s10014-008-0234-8
Beressi N, Beressi JP, Cohen R, Modigliani E (1999) Lymphocytic hypophysitis. A review of 145 cases. Ann Med Interne 150(4):327–341
Weber JS, O’Day S, Urba W, Powderly J, Nichol G, Yellin M, Snively J, Hersh E (2008) Phase I/II study of ipilimumab for patients with metastatic melanoma. J Clin Oncol Off J Am Soc Clin Oncol 26(36):5950–5956. doi:10.1200/JCO.2008.16.1927
Weber J, Thompson JA, Hamid O, Minor D, Amin A, Ron I, Ridolfi R, Assi H, Maraveyas A, Berman D, Siegel J, O’Day SJ (2009) A randomized, double-blind, placebo-controlled, phase II study comparing the tolerability and efficacy of ipilimumab administered with or without prophylactic budesonide in patients with unresectable stage III or IV melanoma. Clin Cancer Res Off J Am Assoc Cancer Res 15(17):5591–5598. doi:10.1158/1078-0432.CCR-09-1024
Acknowledgments
This study received no grant from any funding agency in the public, commercial or not-for-profit sectors.
Conflict of interest
The authors declare that they have no conflict of interest in this study.
Ethical approval
This is a retrospective study and all patient data were collected from the chart.
Informed consent
Informed consent was obtained from the patient.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Araujo, P.B., Coelho, M.C.A., Arruda, M. et al. Ipilimumab-induced hypophysitis: review of the literature. J Endocrinol Invest 38, 1159–1166 (2015). https://doi.org/10.1007/s40618-015-0301-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40618-015-0301-z