Abstract
Background
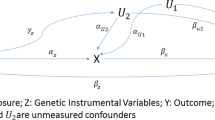
Properly adjusting for unmeasured confounders is critical for health studies in order to achieve valid testing and estimation of the exposure’s causal effect on outcomes. The instrumental variable (IV) method has long been used in econometrics to estimate causal effects while accommodating the effect of unmeasured confounders. Mendelian randomization (MR), which uses genetic variants as the instrumental variables, is an application of the instrumental variable method to biomedical research fields, and has become popular in recent years. One often-used estimator of causal effects for instrumental variables and Mendelian randomization is the two-stage least square estimator (TSLS). The validity of TSLS relies on the accurate prediction of exposure based on IVs in its first stage.
Results
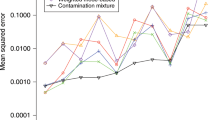
In this note, we propose to model the link between exposure and genetic IVs using the least-squares kernel machine (LSKM). Some simulation studies are used to evaluate the feasibility of LSKM in TSLS setting.
Conclusions
Our results show that LSKM based on genotype score or genotype can be used effectively in TSLS. It may provide higher power when the association between exposure and genetic IVs is nonlinear.

Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Rosenbaum, P. R. and Rubin, D. B. (1983) The central role of the propensity score in observational studies for causal effects. Biometrika. 70, 41–55
Rosenbaum, P. R. and Rubin, D. B. (1984) Reducing bias in observational studies using subclassification on the propensity score. J. Am. Stat. Assoc., 79, 516–524
Rosenbaum, P. R. and Rubin, D. B. (1985) Constructing a control group using multivariate matched sampling methods that incorporate the propensity score. Am. Stat. 39, 33–38
Robins, J. M., Mark, S. D. and Newey, W. K. (1992) Estimating exposure effects by modelling the expectation of exposure conditional on confounders. Biometrics, 48, 479–495
Wright, P. G. (1928) The Tariff on Animal and Vegetable Oils. New York: The Macmillan company
Katan, M. B. (2004) Apolipoprotein E isoforms, serum cholesterol, and cancer. Int. J. Epidemiol., 33, 9
Hillemacher, T., Frieling, H., Moskau, S., Muschler, M. A., Semmler, A., Kornhuber, J., Klockgether, T., Bleich, S. and Linnebank, M. (2008) Global DNA methylation is influenced by smoking behaviour. Eur. Neuropsychopharmacol., 18, 295–298
Bouwland-Both, M. I., van Mil, N. H., Tolhoek, C. P., Stolk, L., Eilers, P. H., Verbiest, M. M., Heijmans, B. T., Uitterlinden, A. G., Hofman, A., van Ijzendoorn, M. H., et al. (2015) Prenatal parental tobacco smoking, gene specific DNA methylation, and newborns size: the Generation R study. Clin. Epigenetics, 7, 83
Crider, K. S., Yang, T. P., Berry, R. J. and Bailey, L. B. (2012) Folate and DNA methylation: a review of molecular mechanisms and the evidence for folate’s role. Adv. Nutr., 3, 21–38
Geach, T. (2017) Obesity: methylation a consequence not a cause. Nat. Rev. Endocrinol., 13, 127
Relton, C. L. and Davey Smith, G. (2012) Two-step epigenetic Mendelian randomization: a strategy for establishing the causal role of epigenetic processes in pathways to disease. Int. J. Epidemiol., 41, 161–176
Lin, W., Feng, R., Li, H. (2015) Regularization methods for highdimensional instrumental variables regression with an application to genetical genomics. J. Am. Stat. Assoc., 110, 270–288
Kang, H., Zhang, A., Cai, T. and Small, D. (2016) Instrumental variables estimation with some invalid instruments and its application to Mendelian randomization. J. Am. Stat. Assoc., 111, 132–144
Hall, P., Horowitz, J. (2005) Nonparametric methods for inference in the presence of instrumental variables. Ann. Stat., 33, 2904–2929
Laurain, V., Toth, R., Piga, D. and Zheng, W. (2015) An instrumental least squares support vector machine for nonlinear system identification. Automatica. 54, 340–347
White, H. (1982) Instrumental variables regression with independent observations. Econometrica, 50, 483–99
Liu, D., Lin, X. and Ghosh, D. (2007) Semiparametric regression of multidimensional genetic pathway data: least-squares kernel machines and linear mixed models. Biometrics, 63, 1079–1088
Kwee, L. C., Liu, D., Lin, X., Ghosh, D. and Epstein, M. P. (2008) A powerful and flexible multilocus association test for quantitative traits. Am. J. Hum. Genet., 82, 386–397
Wu, M. C., Lee, S., Cai, T., Li, Y., Boehnke, M. and Lin, X. (2011) Rare-variant association testing for sequencing data with the sequence kernel association test. Am. J. Hum. Genet., 89, 82–93
Lee, S., Emond, M. J., Bamshad, M. J., Barnes, K. C., Rieder, M. J., Nickerson, D. A., Christiani, D. C., Wurfel, M. M. Lin, X., and the NHLBI GO Exome Sequencing Project—ESP Lung Project Team. (2012) Optimal unified approach for rare-variant association testing with application to small-sample case-control whole-exome sequencing studies. Am. J. Hum. Genet., 91, 224–237
Lee, S., Wu, M. C. and Lin, X. (2012) Optimal tests for rare variant effects in sequencing association studies. Biostatistics, 13, 762–775
Ionita-Laza, I., Lee, S., Makarov, V., Buxbaum, J. D. and Lin, X. (2013) Sequence kernel association tests for the combined effect of rare and common variants. Am. J. Hum. Genet., 92, 841–853
Lee, S., Teslovich, T. M., Boehnke, M. and Lin, X. (2013) General framework for meta-analysis of rare variants in sequencing association studies. Am. J. Hum. Genet., 93, 42–53
Zhang, W., Epstein, M. P., Fingerlin, T. E. and Ghosh, D. (2017) Links between the sequence kernel association and the kernelbased adaptive cluster tests. Stat. Biosci., 9, 246–258
Burgess, S. and Thompson, S. G. (2011) Bias in causal estimates from Mendelian randomization studies with weak instruments. Stat. Med., 30, 1312–1323
Burgess, S. and Thompson, S. G. (2015) Mendelian Randomization: Methods for Using Genetic Variants in Causal Estimation. Boca Raton: CRC Press
Bound J., Jaeger, D. and Baker, R. (1995) Problems with instrumental variables estimation when the correlation between the instruments and the endogenous explanatory variable is weak. J. Am. Stat. Assoc., 90, 443–450
Wang, F., Meyer, N. J., Walley, K. R., Russell, J. A. and Feng, R. (2016) Causal genetic inference using haplotypes as instrumental variables. Genet. Epidemiol., 40, 35–44
Acknowledgements
This research was supported by the National Science Foundation under Grant (No. NSF ABI 1457935) and the National Institutes of Health under Grant (No. R01 GM117946).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zhang, W., Ghosh, D. On the use of kernel machines for Mendelian randomization. Quant Biol 5, 368–379 (2017). https://doi.org/10.1007/s40484-017-0124-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40484-017-0124-3