Abstract
Ever since its outbreak, Corona Virus Disease 2019(COVID-19) caused by SARS-CoV-2 has affected more than 26 million individuals in more than 200 countries. Although the mortality rate of COVID-19 is low, but several clinical studies showed, patients with diabetes mellitus (DM) or other major complication at high risk of COVID-19 and reported more severe disease and increased fatality. The angiotensin-converting-enzyme 2 (ACE2), a component of renin–angiotensin-system (RAS); acts on ACE/Ang-II/AT1recptor axis, and regulates pathological processes like hypertension, cardiac dysfunction, Acute Respiratory Distress Syndrome (ARDS) etc. The progression of T2DM and hypertension show decreased expression and activity of ACE2. There are several treatment strategies for controlling diabetes, hypertension, etc; like ACE2 gene therapies, endogenous ACE2 activators, human recombinant ACE2 (hrACE2), Angiotensin-II receptor blockers (ARBs) and ACE inhibitors (ACEi) medications. ACE2, the receptors for SARS-CoV2, facilitates virus entry inside host cell. Clinicians are using two classes of medications for the treatment of COVID-19; one targets the SARS-CoV-2-ACE2 interaction, while other targets human immune system. The aim of this review is to discuss the role of ACE2 in diabetes and in COVID-19 and to provide an analysis of data proposing harm and benefit of RAS inhibitor treatment in COVID-19 infection as well as showing no association whatsoever. This review also highlights some candidate vaccines which are undergoing clinical trials.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
1 Introduction
Ever since the outbreak of Corona Virus Disease 2019 (COVID-19), caused by Severe Acute Respiratory Syndrome Coronavirus 2 (SARS-CoV-2) has been reported to affect more than 21 million individuals and claimed over 7.5 lakhs lives in more than 200 countries around the globe (COVID-19 situation report-209). Although the mortality rate of COVID-19 is low, patients with diabetes mellitus (DM) and hypertension have shown more severe disease and increased fatality [1, 2]. One of the initial studies done in Wuhan before 2 January, 2020, reported that out of 41 patients infected and identified with COVID-19, 8 were diabetes patients (20%), 6 were hypertensive (15%) and 6 had cardiovascular diseases (15%) [3]. Another study including 26 deceased COVID-19 infected patients reported that, 53.8% were hypertensive, 42.3% diabetes patients, and 19.2% had coronary heart disease. In addition, these significant comorbidities were related with an expanded danger of mortality [4]. A study done in China comprising of 72,314 cases reported that DM patients show higher mortality rate (7.3%) when compared to other patients [5]. Of all COVID-19 patients who died in Italy, 20.3% were diabetes patients [6]. A cross-sectional survey (NCT04331574) with 1581 COVID-19 patients was performed in Italy, where except hypertension age, diabetes mellitus, chronic kidney disease and chronic obstructive pulmonary disease predicted mortality [7]. It was suggested that sex and gender disparities played a role in COVID-19 vulnerability [8]. Global Health 50/50 demonstrated nearly equal number of cases in men and women, but increased fatality in men [9]. Several factors like male-female differences in immune response to vaccines, increased adverse drug reaction to antiviral treatment, influence the action of drugs and vaccines in COVID-19 patients [8].
Older age, diabetes, hypertension, coronary artery disease and smoking are major risk factors for severe COVID-19, and all these conditions relate with vascular endothelial dysfunction [10]. This effects the vascular equilibrium and causes vasoconstriction, thrombosis and inflammation. Angiopoietin-2, a biomarker for endothelial dysfunction was found significantly increased in critical COVID-19 patients, it suggests COVID-19-associated microvascular dysfunction [8]. The role of endothelial-dysfunction in COVID-19 pathogenesis and its complications should be the focus of future studies.
The role of renin–angiotensin-system (RAS) has been suggested in the progression of diabetes. Angiotensin-II (Ang-II) prevents elevated insulin secretion from pancreatic islets in hyperglycemic condition by disrupting β-cell function, and Angiotensin-converting enzyme 2 (ACE2) gene therapy reduces this damaging effect of Ang-II and insulin is released accordingly from β-cells during hyperglycemic condition. Furthermore, both Angiotensin-II receptor blockers (ARBs) and ACE inhibitors (ACEi) control glucose levels by blocking overactive RAS and improve the morphology and function of islets. In spite of large data obtained from studies in animal models, facts on humans are yet not clear. This suggests for future studies on exploring the medications targeting RAS as a potent therapeutic for diabetes and comorbidities. ACE2 was found to be one of the main receptors of both SARS-CoV and SARS-CoV-2. ACE2 receptors are expressed widely on heart, respiratory tract, intestines, kidneys and pancreas [11].
Till date there is no specific vaccine or medication against COVID-19 [12] and clinicians are using two classes of medications; one which acts directly on coronavirus (CoV) and the other that targets human immune system [13]. The most effective therapeutics could be targeting the interaction of host-cell receptor and the virus itself, which will stop the binding of SARS-CoV-2 with ACE2 receptor and ultimately terminate the entry of the virus into the host cell [14]. This approach may enhance diabetes complications, cardiovascular diseases and other comorbidities by affecting normal functioning of ACE2, which ultimately increases Ang-II levels and leads to inflammation and oxidative stress in islets.
Results of some studies have suggested that boosting passive immunity could be an effective approach for the treatment of severe COVID-19. The trials of a recombinant adeno vaccine (AZD1222), developed by Oxford University’s Jenner Institute have begun last month and if data from the trial show positive results, late-stage trials would begin in a number of countries. After vaccination, the spike proteins are produced, which prepares the immune system to attack COVID-19 if it infects body in future.
Apart from these approaches there could be some other strategies that could prove to be potent therapeutics for COVID-19 in context of ACE2. On the other hand, there is a huge controversy regarding the use of ARBs and ACEi medications, as they can increase ACE2 levels and thus make subjects more susceptible to SARS-CoV infection. The present knowledge lacks any clinical data to support the hypothesis that use of ARBs/ACEi increases patients’ susceptibility to COVID-19 infection and suggests both benefit as well as harm. Therefore, it’s better to continue ARBs and ACEi medications until some strong evidence claims such hypothesis to be true.
Exactly how DM and cardiovascular comorbidities are related with increased complications and significantly increased death rates for patients infected with COVID-19 is not known. As COVID-19 is profoundly transmissible and extremely pathogenic, understanding these mechanisms can help in the development of potential and successful treatments for diabetes patients susceptible to COVID-19 infection. In this review we have discussed the role of ACE2 in diabetes and in COVID-19 and analysed the data proposing harm and benefit of RAS inhibitor treatment in COVID-19 infection as well as showing no association whatsoever. This review also highlights some candidate vaccines which are undergoing clinical trials.
2 ACE2: Overview and Function
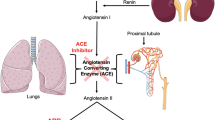
ACE2 is a component of RAS which maintains blood pressure, fluid and electrolyte balance and reabsorption of sodium in kidney [15] by controlling vasoconstriction. As a usual physiological feedback to low renal blood flow, juxtaglomerular cells convert inactive prorenin to active renin, which is secreted directly into the circulation. Now, plasma renin converts angiotensinogen, from liver, to Angiotensin-I (Ang-I). Angiotensin converting enzyme (ACE) converts Ang-I to its active form Ang-II, a vasoconstrictor (Fig.1). ACE2 counterbalances the whole mechanism by targeting Ang-II and converting it to Ang 1–7, which is a vasodilator [15]. ACE2 thus regulates several pathological processes like fibrosis, inflammation, oxidative stress and vasoconstriction. There are many drugs that interrupts this system in different steps to control the abnormally active RAS. The inhibitors of ACE enzyme (ACEi) are often used to downregulate the production of potent Ang-II (Fig. 1). Furthermore, ARBs are also used to control the overactive RAS by preventing the binding of Ang-II to its receptors Angiotensin-II Type 1 Receptor (AT1R) (Fig. 1).
3 ACE2: Role in Diabetes
ACE2 has been known to exist in many tissues, including heart, respiratory tract, intestines, kidneys and pancreas [11]. ACE2 controls RAS by acting on ACE/Ang-II/AT1R axis, and by this it regulates several pathological processes like acute respiratory distress syndrome (ARDS) [16], fibrosis [17], hypertension and cardiac dysfunction [17].
Ang-I and Ang-II being the main targets of ACE2 are degraded to Ang-(1–9) and Ang-(1–7) respectively (Fig. 1). Ang-(1–7), a vasodilator peptide, is a ligand for the Mitochondrial assembly receptor (MasR) which opposes the function of vasoconstrictor Ang-II [18]. Thus, ACE2 can be considered as a protective agent, as it increases and decreases the levels of Ang-(1–7) and Ang-II respectively, which prevents the harmful effects of overactive RAS. ACE2 is gaining attention, due to this therapeutic property given the extensive role of RAS in controlling different functions in our body.
Chhabra and his colleagues (in 2013) for the first time demonstrated a direct role of ACE2 in insulin secretion by culturing mouse islets in presence of Ang-II [19]. Ang-II prevents elevated insulin secretion from pancreatic islets in hyperglycemic condition by disrupting β-cell function, and ACE2 gene therapy reduces this damaging effect of Ang-II and insulin is released accordingly from β-cells during hyperglycemic condition [19]. In β-cells, ACE2 seems to obstruct Ang-II signalling thereby showing its therapeutic effects. Indeed, Ang-II mediated upregulation of AT1R in mice islets was attenuated upon treatment with ACE2 adenovirus [19]. Therefore, ACE2 is thought to have potential of improving AT1R-mediated harmful effects, like oxidative stress, inflammation, fibrosis and blood flow in the β-cells (Fig. 1).
Ang-II induces the production of NADPH oxidase which produces superoxide radicals and thereby increases oxidative stress [20] which finally results in β-cell dysfunction [21]. Some studies have reported role of ACE2 in reducing Ang-II-mediated oxidative stress [19], thus improving pancreatic dysfunction.
β-Cell function is significantly hampered by islet fibrosis and inflammation [22, 23], thereby effecting glucose homeostasis. The beneficial role of ACE2 in reducing inflammation and fibrosis in islets is yet to be explored. The glucose levels have been found to be improved by agents that reduce fibrosis and inflammation [23]. Ang-II mediates islets fibrosis and inflammation via AT1R [22], therefore it becomes important to investigate the role of ACE2 in lowering fibrosis and inflammation islets.
Carlsson et al. [24] suggested that glucose-homeostasis is affected by modulating blood flow to the pancreatic islets. Although, ACE2 therapy in Ang-II treated mice models exhibited no increase in pancreatic blood flow [19], but ACE2 as a vasodilator may affect insulin secretion [19] by increasing the blood flow in islets suggesting that further studies should be done.
A novel mechanism that has been shown to promote chronic diseases like diabetes and hypertension [25, 26] is Endoplasmic reticulum (ER) stress. ER stress is found to be induced by elevated Ang-II level [19], therefore hypertension can be reduced by drugs which limits RAS activity [27]. Although, ER stress has been thought to effect β-cell function and cause hyperglycemia [28], further studies should be performed to investigate the role of Ang-II and ACE2 reducing ER stress [27].
Several studies have demonstrated that with the progression of T2DM and hypertension, there is a decrease in the expression level and activity of ACE2 in various tissues [19, 29, 30]. In type 2 diabetes mellitus (T2DM) cases that have developed atherosclerosis plaque, the ADAM metallopeptidase domain 17 (ADAM17) expression was found upregulated [31] and this tumour necrosis factor-α-converting enzyme (TACE), is reported to cleave and remove the catalytic domain of membrane-bound ACE2 [32]. This could be an explanation for ACE2 downregulation in mouse islets [33]. Interestingly, expression of ADAM17 is upregulated by Ang-II in pancreas, brain, adipose tissue and smooth muscle cells of blood vessels [33]. On the other hand, ACE2 treatment can decrease the expression of ADAM17, which suggests that modulating Ang-II levels, effects the level of ADAM17 [33]. Hence, these preliminary studies suggest that shedding of ACE2 in the islet links overactive RAS and hyperglycemia, and this could be normalized by restoring shed ACE2.
3.1 ACE2: Role in Comorbidities of Diabetes
3.1.1 Hypertension
ACE2 is present in vascular endothelium; therefore, reduction in expression of ACE2 results in an increase in the levels of Ang-II, which leads to increased blood pressure [34]. A study done on rats demonstrated that increased expression of ACE2 in blood vessels reduced blood pressure, on the other hand, deficiency of ACE2 contributed to elevated systolic blood pressure [34, 35]. Furthermore, levels of ACE2 have been shown to affect central hypertension as well [35]. These studies suggested that maintaining the concentration of ACE2 may prove to be an important therapeutic approach against diabetes induced hypertension.
3.1.2 Cardiomyopathy
The patients with diabetes are more prone to develop cardiovascular disease compared to normal population. An imbalance in ACE2-Ang-(1–7) and ACE-Ang-II and activation of RAS are mainly responsible for this process [15]. Hyperglycemia promotes the local production of Ang-II in heart tissue. In diabetes models, upregulation of RAS leads to Ang-II pathway activation and subsequently increase oxidative stress, fibrosis, inflammation, cell proliferation and even apoptosis. These changes cause cardiac remodelling and lead to development of atherosclerosis. ACE2 reduces cardiac remodelling, prevents myocardial fibrosis and left ventricular hypertrophy. Most importantly, ACE2 prevents heart failure by pressure overload, and thus improves functioning of heart [36]. Therefore, ACEi and ARBs are recommended first line medications for Cardiovascular diseases (CVD) in diabetes patients by regulating blood pressure and reducing damages in target organs [15, 36].
3.1.3 Nephropathy
It is a microvascular complication of diabetes and is associated with CVD risk. In kidney, the expression of ACE2 predominantly takes place at the proximal tubule, more precisely at its luminal brush border. In case of nephropathy or other chronic kidney diseases, expression of ACE2 is downregulated which causes tubular injury and albuminuria. Therefore, further inhibition of ACE2 increases renal injury [35]. Harris et al. [37], reported an increase in ACE/ACE2 ratio in both tubulointerstitium and glomerulus of nephropathy patients, mainly due to reduced expression of ACE2.
3.1.4 Retinopathy
It is the most prevalent microvascular complication of diabetes in which RAS abnormality plays an essential role [38]. However, ACE2/Ang-(1–7) overexpression may be beneficial in retinopathy [38]. Some animal studies have shown that ACE2 can decrease intra-ocular pressure by counter-regulating Ang-II [38]. Interestingly, treatment including recombinant ACE2 could prevent the loss of ganglionic cells of retina and decrease inflammatory process. These studies suggest that increasing the bioavailability of ACE2 can be critical in the treatment of diabetes and its complications [39].
3.2 Treatment Strategies for Diabetes (Targeting RAS)
3.2.1 ACE2 Therapies
Several approaches are under preclinical stage of investigation to increase the activity of ACE2 in different disorders including diabetes. Among them the most widely used is the gene therapy in animal models [30, 40]. Many groups have investigated the beneficial role of gene therapy in diabetes and other disorders [30, 40, 41]. This therapy showing promising results uses either lentivirus or adenovirus, which carries ACE2 encoding gene, thus increasing ACE2 activity.
In subjects with diabetes, endogenous ACE2 activators such as diminazene and xanthenone, have been beneficial in controlling hyperglycemic condition and different complications of diabetes like endothelial dysfunction and hypertension [42]. This suggests ACE2 activators to be a novel approach in controlling diabetes and its related complications as they increase the activity of ACE2.
Another approach which boosts the ACE2 activity is human recombinant ACE2 (hrACE2) therapy, which has shown to control T2DM and its complications in animal models [43]. The hrACE2 therapy reduces Ang-II mediated activity of NADPH oxidase and promotes Ang-(1–7) signalling in Akita mouse model [43]. These results encourage to investigate the role of rhACE2 in controlling hyperglycemia and secretion of insulin from β-cells as Ang-II and NADPH oxidases act negatively in the progression of T2DM.
3.2.2 Role of ARBs and ACEi in Hyperglycemia
RAS contributes to reduced insulin secretion [44]. In fact, in-vitro and in-vivo studies done on rodent models have shown RAS induced oxidative stress, islet fibrosis, inflammation, and most importantly impaired insulin secretion. Both Angiotensin-II receptor blockers (ARBs) and ACE inhibitors (ACEi) control glucose levels by blocking overactive RAS. Moreover, ARB or ACEi were found to improve the morphology and function of islets and thereby increase the glucose tolerance [45, 46].
In spite of large data from studies done on animal models, facts on humans are not clear. An in-vitro study done on the isolated human pancreatic islets showed that when they were treated with ACEi, they neutralized some of the adverse effects of high-glucose exposure including oxidative stress and impaired glucose secretion [47]. In-vivo studies have shown mixed results. One study demonstrated that treatment with ACEi ramipril (≈ 15mg daily) [48] or ARB valsartan (80 mg twice a day) [49] for 3 years and 6 weeks respectively did not help in improving insulin secretion. Another study showed that early phase secretion of insulin can be increased in cases with impaired glucose tolerance and hypertension when they are treated with ACEi captopril (≈ 81 ± 24 mg daily) or ARB candesartan (≈ 8 mg daily) for 4 and 3 months respectively [50, 51]. These findings indicate that RAS in pancreatic islets may be a causative factor for impaired insulin secretion. Furthermore, RAS blockade can be used as a good approach to increase insulin secretion in subjects who are at higher risk of developing T2DM.
4 ACE2: Role in COVID-19
Coronavirus (CoV) comprises of 4 structural proteins, notably envelope, membrane, nucleocapsid and most important in this context: the spike protein [52]. The angiotensin-converting-enzyme 2 (ACE2), was found to be one of the main receptors of both SARS-CoV-2 [53] and SARS-CoV [54], which is expressed in many tissues [11]. The host when exposed to the virus, CoVs by the help of spike protein bind to the cells expressing specific ACE2 receptors. After this the spike protein is cleaved off by the host-cell protease, which facilitates viral entry and replication inside the cell [53].
As the expression of ACE2 is down-regulated with the onset of infection, its role in developing lung injury in patients with SARS-CoV infection was disputable [55]. However, it was assumed that, ACE2 has two main roles; one as a receptor helping the virus to enter, and further, protecting from lung injury by it’s down-regulation [55].
Two primary theories exist suggesting the role of RAS in COVID-19 infection. First, few experimental data demonstrate that severe lung injury and fibrosis in SARS-CoV animal models is mainly mediated by Ang-II inflammation [56, 57]. The decrease in ACE2 expression prior to SARS-CoV-2 infection increases local concentration of Ang-II in the lungs and causes severe lung injury. A higher concentration of circulating Ang-II was reported in COVID-19 patients and it was found to correlate with the viral load and symptoms of lung injury [58]. Earlier SARS-CoV epidemic data also suggested that ACE2 dependent SARS-CoV-2 infection may decrease ACE2 expression in heart, promoting severe heart injury [59]. It is important to note that we lack sufficient clinical data to support above mentioned pathophysiological processes.
Second, ARBs and ACEi could increase the expression of ACE2 in lungs, which increases the risk of COVID-19 infection [60]. There is experimental data which demonstrate the increase of ACE2 expression by ARBs and ACEi in heart [61], but there are no evidence showing increase of cardiac ACE2 expression and activity by these agents in lungs of animals or humans. Similarly, there is no such study done so far that proves that these agents increase virulence or viral infectivity by their downstream effects.
Majority of studies actually support the idea that ARBs and ACEi may reduce Ang-II-mediated fibrosis and severe lung injury by limiting the action of Ang-II [62]. These agents as such promise some potent therapies for the treatment of COVID-19 [56, 63]. The limited available data suggest to continue the use of these agents if needed until there is any evidence against them [64, 65].
4.1 Treatment Strategies for COVID-19
Still there is no specific vaccine or medication available against COVID-19. In order to fight against COVID-19, clinicians are using two classes of medications; one which acts directly on coronavirus (CoV) and the other which targets human immune system [13].
4.1.1 Targeting Interaction of Virus and Host Cell Receptor
The most effective therapeutics could be targeting the interaction of host-cell receptor and the virus itself, which will ultimately terminate the entry of the virus into the host cell. As already been discussed, the virus enters the host-cell via ACE2 receptor [66], therefore, by any means stopping the binding of virus to the receptor could stop COVID-19 infection. Chloroquine, which was earlier very effective in the treatment of malaria, HIV, amoebiasis, and autoimmune diseases, has proved to be very potent in treating COVID-19 patients [67]. It prevents the ACE2 glycosylation which reduces the affinity of spike protein and ACE2.
As already discussed, decreased activity of ACE2 increases Ang-II level, which then increases inflammation and oxidative stress in islets leading to onset of diabetes and related complications [38]. ACE2/Ang-(1–7) pathway modulates glucose level by maintaining pancreatic Ang-(1–7) and/or Ang-II levels and thus regulates the secretion of insulin [30, 33]. Therefore, targeting ACE2/Ang-(1–7) pathway could be an effective strategy for treating patients with T2DM.
4.1.2 Targeting Human Immune System
COVID-19 patients showed elevated levels of CXCL10, CCL2 and TNF-α: ‘cytokine storm’, whereas IFN-γ, IL-1, IP-10 levels increased with COVID-19 severity. COVID-19 severity was affected by abnormal host immune response along with lymphocytopenia and cytokine storm. Therefore, modulating immune response might be crucial for COVID-19 treatment [68].
The other effective approach could be development of passive immunization by providing antibodies [69]. The immunotherapy by using monoclonal antibody medication may prove to be effective because of its higher specificity, safety, purity, and less risk of contamination. Khan et al. [70] conducted a study on ARDS patients, and demonstrated that using rhACE2 (GSK2586881) resulted in a decrease in Ang-II and IL-6 plasma levels and an increase in Ang-(1–7). This suggests that such an approach could be effective for the treatment of severe COVID-19.
4.1.3 Development of vaccines for COVID-19
-
1.
Developing a vaccine is a long game. Ever since the genetic sequence of SARS-CoV-2 is known, several candidate vaccines are in clinical trials (https://clinicaltrials.gov/) (Table 1). Vaccine manufacturers are racing to develop vaccines for COVID-19 pandemic, but challenges still remain. Below is the table highlighting difference vaccines under clinical trials.
5 RAS Inhibitor Drugs: Role in COVID-19
As mentioned earlier, ACE2 being the functional receptor for corona virus, helps in the entry of both SARS-CoV [52] and SARS-CoV-2 [58]. The importance of this finding has now increased as the medications used currently are considered to increase ACE2 levels. Earlier Italian Society of Hypertension (SIIA) and other International Societies recommended to continue ACEi and/or ARBs in CVDs and hypertensive patients [71]. Later on, clinical trials [72] investigated the impact of continuing versus suspending ACEi and ARBs in COVID-19 patients. BRACE CORONA Trial (NCT04364893) in Brazil was one of those randomized clinical trials [73]. The primary objective of BRACE was to investigate whether continuous versus discontinuous therapy of ACEi and/or ARBs affected days alive and out from hospital in 30 days [73]. Moreover, several studies have proposed both benefits as well as harms of treatment including ACEi and ARBs during COVID-19 infection. In addition, there are few studies which have shown neutral data too.
5.1 Harmful Effect of RAS Inhibitors
At first, the use of RAS modulators was not only thought to increase the risk of COVID-19 infection, but increase the severity of infection and mortality as well. An early study considered the use of RAS inhibitor medications to increase lethal COVID-19 infection [74].
Fang et al. [2] hypothesised that patients with diabetes and hypertension, who are treated with RAS modulators are at increased risk of COVID-19 as it elevates the ACE2 concentration. They raised concerns over the use of ibuprofen and thiazolidinediones as they also increase ACE2. Furthermore, they also suggested the use of calcium-channel blockers as an alternative for the treatment of hypertension as no data supported the increase in ACE2 concentration by the use of these drugs. Sommerstein et al. [75] also proposed similar hypothesis and had raised concerns over the use of ACEi/ARBs as they might increase the risk and severity of COVID-19.
There are several points which raises some doubts over these studies. The studies mentioned above reported severity of COVID-19 in patients with diabetes, hypertension, and coronary artery disease (CAD). But the studies have not shown the outcomes separately in patients treated and untreated with ACEi/ARBs. Moreover, to conclude that these medications are directly related with severe outcomes, the studies should be done on the patients having these medications, and not to emphasise on the disease for which these medications are used.
5.2 Beneficial Effect of RAS Inhibitors
Recently, studies have shown the beneficial role of RAS inhibitors (Table 2) and elevated ACE2 levels in COVID-19 patients. As discussed previously, binding of ACE2 and SARS-CoV-2 spike protein downregulates ACE2 and thus increase Ang-II concentration. This causes pulmonary congestion and difficulty in breathing [56, 76]. Therefore, certain medication which increase ACE2 concentration can be helpful in reducing the severity of COVID-19 [12]. Several retrospective studies done on patients with hypertension and COVID-19, showed decreased mortality rate in patients receiving RAS inhibitors when compared with patients receiving other hypertensive medications [77, 78]. Lam et al [79] investigated continued versus discontinued ACEi or ARBs use in hypertensive COVID-19 patients and found better clinical outcomes and suggested to continue their use.
Meng et al. in their case control study divided patients into two groups; RAS inhibitor (17 patients) and non-inhibitor (25 patients) group. They concluded that groups receiving RAS inhibitors showed less disease severity, increased blood CD-8 and CD-3 T-cells, decreased serum interleukin-6 (IL-6) and decreased viral load when compared to group receiving on-RAS inhibitors. Thus, improved clinical outcomes in hypertension patients with COVID-19 was found related with medications including RAS inhibitors [80].
5.3 Neutral Effect of RAS Inhibitors
Apart from studies showing harmful effects and benefits of using RAS inhibitors, there are several studies which have demonstrated no association of RAS inhibitors with the severity of COVID-19 (Table 3). A study done on patients in Italy, included 6,272 cases compared with 30,759 controls and found no association of RAS antagonist risk and severity of COVID-19 infection [81]. Another study done by Reynolds et al. [82] included approximately 17.0% severe COVID-19 patients of around 12,500 patients tested for COVID-19. The authors concluded that there was no relation between RAS antagonist medication and COVID-19 infection or its severity. In addition, they also studied calcium channel blockers, beta-adrenoceptor blockers and thiazide diuretics and demonstrated no association of these medications with increased risk of COVID-19 infection.
There are several other studies which have reported no association between RAS inhibitor medication and increased severity and mortality of COVID-19 patients [83,84,85]. All these studies had certain limitations but proposed same conclusion, that there is no association between RAS inhibitors and COVID-19 infection.
6 Future Perspectives
There can be several strategies that can be developed which might show positive results in dealing with ACE2 mediated COVID-19 infection (Fig. 2):
-
1.
As mentioned earlier, SARS-CoV and SARS-CoV-2 enters the host-cell by the help of a protein termed as SPIKE (S-protein), expressed on the viral coat. These S-proteins have receptor binding regions which binds to ACE2’s extra cellular domain [62]. Therefore, developing a vaccine based on S-protein may be helpful as it will hamper the spike protein-ACE2 receptor interaction (Fig. 2). Furthermore, these interaction sites could be blocked with antibodies and/or some other small peptide molecules (Fig. 2).
-
2.
SARS-CoV-2 entry into the host cell also depends on the priming between S-protein and TMPRSS2 (a transmembrane protease serine 2) [86]. It is also important to note that camostat mesylate, a serine protease inhibitor, has been proved to block the activity of TMPRSS2 [87, 88], and in Japan they have been approved for human use. Therefore, camostat mesylate can be used to hamper the priming of S-protein and TMPRSS2, thus preventing the SARS-CoV-2 infection (Fig. 2).
-
3.
If high levels of ACE2 delivered it may competitively bind to S-protein of SARS-CoV-2 and thus neutralize the virus and rescue cellular activity of ACE2 (Fig. 2).
7 Conclusion
At present time, no data supports the hypothesis that use of ARBs/ACEi increases patients’ susceptibility to COVID-19 infection, its severity and mortality. The studies done so far on the use of RAS medications in patients suffering from COVID-19 have shown conflicting data. In the joint statement, American Heart Association (AHA), American College of Cardiology (ACC) and Heart Failure Society of America (HFSA) have recommended physicians not to begin or discontinue the usual RAS related medications (ARBs/ACEi) in COVID-19 patients. Any decision taken in this regard should be based on patient’s hemodynamic status and its clinical presentation [89, 90].
Therefore, after reviewing the available clinical data till date, recommendations and opinions from different experts in different scientific societies, we can conclude that ARBs/ACEi can be continued by the physicians to treat patients that are in need. Furthermore, research done on RAS expression, especially ACE2/Ang-(1–7) pathway in COVID-19 cases can fill the knowledge gaps and may significantly affect the treatment guidelines of the patients on ACEi/ARBs medications.
References
Guan W, Ni Z, Hu Y, et al. Clinical characteristics of coronavirus disease 2019 in China. N Engl J Med. 2020;382:1708–20.
Fang L, Karakiulakis G, Roth M. Are patients with hypertension and diabetes mellitus at increased risk for COVID-19 infection? Lancet Respir Med. 2020;8(4):e21.
Huang C, Wang Y, Li X, et al. Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. Lancet. 2020;395:497–506.
Deng SQ, Peng HJ. Characteristics of and public health responses to the coronavirus disease 2019 outbreak in China. J Clin Med. 2020;9(2):575.
Wu Z, McGoogan JM. Characteristics of and important lessons from the coronavirus disease 2019 (COVID-19) outbreak in China: summary of a report of 72,314 cases from the Chinese Center for disease control and prevention. JAMA. 2020;323(13):1239–42.
Onder G, Rezza G, Brusaferro S. Case-fatality rate and characteristics of patients dying in relation to COVID-19 in Italy. JAMA. 2020;323(18):1775–6.
Mancusi C, Grassi G, Borghi C, et al. Determinants of healing among patients with COVID-19: the results of the SARS-RAS study of the Italian Society of Hypertension. J Hypertens. 2020. https://doi.org/10.1097/HJH.0000000000002666.
Gebhard C, Regitz-Zagrosek V, Neuhauser HK, et al. Impact of sex and gender on COVID-19 outcomes in Europe. Biol Sex Differ. 2020;11:29.
Sex, gender and COVID-19. Global Health 5050. 2020. https://globalhealth5050.org/covid19/.
Mosleh W, Chen K, Pfau SE, et al. Endothelial dysfunction in patients with COVID-19: its role in thrombosis and adverse outcomes. J Clin Med. 2020;9:1862.
Bernardi S, Michelli A, Zuolo G, et al. Update on RAAS modulation for the treatment of diabetic cardiovascular disease. J Diabetes Res. 2016;2016:1–17.
Gurwitz D. Angiotensin receptor blockers as tentative SARS-CoV-2 therapeutics. Drug Dev Res. 2020;81(5):537–40.
Li G, Clercq ED. Therapeutic options for the 2019 novel coronavirus (2019-nCoV), vol. 19. Berlin: Nature Publishing Group; 2020.
Honardoost M, Aghili R, Khamsch ME. Coronavirus 2019 infection in people with associated comorbidities: case fatality and ACE2 inhibitors treatment concerns. Preprints 2020.
Song Z, Xu Y, Bao L, et al. From SARS to MERS, thrusting coronaviruses into the spotlight. Viruses. 2019;11(1):59.
Gembardt F, Sterner-Kock A, Imboden H, et al. Organ-specific distribution of ACE2 mRNA and correlating peptidase activity in rodents. Peptides. 2005;26:1270–7.
Paizis G, Tikellis C, Cooper ME, et al. Chronic liver injury in rats and humans upregulates the novel enzyme angiotensin converting enzyme 2. Gut. 2005;54:1790–6.
Santos RAS, Silva ACS, Maric C, et al. Angiotensin-(1–7) is an endogenous ligand for the G protein-coupled receptor Mas. Proc Natl Acad Sci. 2003;100(14):8258–63.
Chhabra KH, Xia H, Pedersen KB, et al. Pancreatic angiotensin-converting enzyme 2 (ACE2) improves glycemia in angiotensin II-infused mice. Am J Physiol Endocrinol Metab. 2013;304:874–84.
Hitomi H, Kiyomoto H, Nishiyama A. Angiotensin II and oxidative stress. Curr Opin Cardiol. 2007;22:311–5.
Yoshitaka K, Hideaki K. Role of oxidative stress in pancreatic β -cell dysfunction. Ann NY Acad Sci. 2004;1011:168–76.
Hayden MR, Karuparthi PR, Habibi J, et al. Ultrastructural Islet study of early fibrosis in the Ren2 Rat model of hypertension emerging role of the Islet pancreatic pericyte-stellate cell. JOP. 2007;8(6):725–38.
Donath MY, Böni-Schnetzler M, Ellingsgaard H, et al. Islet inflammation impairs the pancreatic β-cell in type 2 diabetes. Am Physiol Soc. 2009;24:325–31.
Carlsson PO, Andersson A, Jansson L. Pancreatic islet blood flow in normal and obese-hyperglycemic (ob/ob) mice. Am J Physiol Endocrinol Metab. 1996;271:990–5.
Flamment M, Hajduch E, Ferre P, et al. New insights into ER stress-induced insulin resistance. Trends Endocrinol Metab. 2012;23:381–90.
Young CN, Cao X, Guruju MR, Pierce JP, et al. ER stress in the brain subfornical organ mediates angiotensin-dependent hypertension. J Clin Invest. 2012;122(11):3960–4.
Chhabra KH, Pedersen KB, Xia H, et al. Pancreatic ACE2 shedding is associated with impaired glycemia in high fat diet-fed mice. FASEB J. 2013;27:1154.
Negi S, Park SH, Jetha A, et al. Evidence of endoplasmic reticulum stress mediating cell death in transplanted human islets. Cell Transplant. 2012;21:889–900.
Tikellis C, Johnston CI, Forbes JM, et al. Characterization of renal angiotensin-converting enzyme 2 in diabetic nephropathy. Hypertension. 2003;41:392–7.
Bindom SM, Hans CP, Xia H, et al. Angiotensin-I converting enzyme type 2 (ACE2) gene therapy improves glycemic control in diabetic mice. Diabetes. 2010;59:2540–8.
Cardellini M, Menghini R, Martelli E, et al. TIMP3 is reduced in atherosclerotic plaques from subjects with type 2 diabetes and increased by SirT1. Diabetes. 2009;58(10):2396–401.
Lambert DW, Yarski M, Warner FJ, et al. Tumor necrosis factor-alpha convertase (ADAM17) mediates regulated ectodomain shedding of the severe-acute respiratory syndrome-coronavirus (SARS-CoV) receptor, angiotensin-converting enzyme-2 (ACE2). J Biol Chem. 2005;280:30113–9.
Chhabra KH, Chodavarapu H, Lazartigues E. Angiotensin converting enzyme 2: a new important player in the regulation of glycemia. IUBMB Life. 2013;65:731–8.
Chamsi-Pasha MAR, Shao Z, et al. Angiotensin-converting enzyme 2 as a therapeutic target for heart failure. Curr Heart Fail Rep. 2014;11(1):58–63.
Tikellis C, Thomas MC. Angiotensin-converting enzyme 2 (ACE2) is a key modulator of the renin angiotensin system in health and disease. Int J Pept. 2012;2012:1–8.
Wang J, He W, Guo L, et al. The ACE2-Ang (1–7)-Mas receptor axis attenuates cardiac remodeling and fibrosis in post-myocardial infarction. Mol Med Rep. 2017;16(2):1973–81.
Harris RC. Podocyte ACE2 protects against diabetic nephropathy. Kidney Int. 2012;82(3):255–6.
Foureaux G, Nogueira BS, Coutinho DCO, et al. Activation of endogenous angiotensin converting enzyme 2 prevents early injuries induced by hyperglycemia in rat retina. Braz J Med Biol Res. 2015;48(12):1109–14.
Verma A, Xu K, Du T, et al. Expression of human ACE2 in Lactobacillus and beneficial effects in diabetic retinopathy in mice. Mol Ther Methods Clin Dev. 2019;14:161–70.
Raizada MK, Ferreira AJ. ACE2: a new target for cardiovascular disease therapeutics. J Cardiovasc Pharmacol. 2007;50:112–9.
Bindom SM, Lazartigues E. The sweeter side of ACE2: physiological evidence for a role in diabetes. Mol Cell Endocrinol. 2009;302:193–202.
Murca TM, Moraes PL, Capuruco CAB, et al. Oral administration of an angiotensin-converting enzyme 2 activator ameliorates diabetes-induced cardiac dysfunction. Regul Pept. 2012;177:107–15.
Oudit GY, Liu GC, Zhong J, et al. Human recombinant ACE2 reduces the progression of diabetic nephropathy. Diabetes. 2010;59:529–38.
Tikellis C, Cooper ME, Thomas MC. Role of the renin-angiotensin system in the endocrine pancreas: implications for the development of diabetes. Int J Biochem Cell Biol. 2006;38:737–51.
Ko SH, Kwon HS, Kim SR, et al. Ramipril treatment suppresses islet fibrosis in Otsuka Long-Evans Tokushima fatty rats. Biochem Biophys Res Commun. 2004;316:114–22.
Chu KY, Lau T, Carlsson PO, et al. Angiotensin II type 1 receptor blockade improves beta-cell function and glucose tolerance in a mouse model of type 2 diabetes. Diabetes. 2006;55:367–74.
Lupi R, Del Guerra S, Bugliani M, et al. The direct effects of the angiotensin-converting enzyme inhibitors, zofenoprilat and enalaprilat, on isolated human pancreatic islets. Eur J Endocrinol. 2006;154:355–61.
Hanley AJ, Zinman B, Sheridan P, et al. Effect of Rosiglitazone and Ramipril on -cell function in people with impaired glucose tolerance or impaired fasting glucose: the DREAM trial. Diabetes Care. 2010;33:608–13.
Bokhari S, Israelian Z, Schmidt J, et al. Effects of angiotensin II type 1 receptor blockade on beta-cell function in humans. Diabetes Care. 2007;30:181–181.
Pollare T, Lithell H, Berne C. A comparison of the effects of hydrochlorothiazide and captopril on glucose and lipid metabolism in patients with hypertension. N Engl J Med. 1989;321:868–73.
Suzuki K, Nakagawa O, Aizawa Y. Improved early-phase insulin response after candesartan treatment in hypertensive patients with impaired glucose tolerance. Clin Exp Hypertens. 2008;30:309–14.
Adedeji AO, Severson W, Jonsson C, et al. Novel inhibitors of severe acute respiratory syndrome coronavirus entry that act by three distinct mechanisms. J Virol. 2013;87(14):8017–28.
Letko M, Marzi A, Munster V. Functional assessment of cell entry and receptor usage for SARS-CoV-2 and other lineage B betacoronaviruses. Nat Microbiol. 2020;5:562–9.
Li W, Moore MJ, Vasilieva N, et al. Angiotensin converting enzyme 2 is a functional receptor for the SARS coronavirus. Nature. 2003;426:450–4.
Zhang H, Penninger JM, Li Y, et al. Angiotensin-converting enzyme 2 (ACE2) as a SARS-CoV-2 receptor: molecular mechanisms and potential therapeutic target. Intensive Care Med. 2020;46:586–90.
Kuba K, Imai Y, Rao S, et al. A crucial role of angiotensin converting enzyme 2 (ACE2) in SARS coronavirus-induced lung injury. Nat Med. 2005;11:875–9.
Li X, Molina-Molina M, Abdul-Hafez A, et al. Angiotensin converting enzyme-2 is protective but downregulated in human and experimental lung fibrosis. Am J Physiol Lung Cell Mol Physiol. 2008;295:178–85.
Liu Y, Yang Y, Zhang C, et al. Clinical and biochemical indexes from 2019-nCoV infected patients linked to viral loads and lung injury. Sci China Life Sci. 2020;63:364–74.
Oudit GY, Kassiri Z, Jiang C, et al. SARS-coronavirus modulation of myocardial ACE2 expression and inflammation in patients with SARS. Eur J Clin Invest. 2009;39:618–25.
Watkins J. Preventing a covid-19 pandemic. BMJ. 2020;368:m810.
Ferrario CM, Jessup J, Chappell MC, et al. Effect of angiotensin-converting enzyme inhibition and angiotensin II receptor blockers on cardiac angiotensin-converting enzyme 2. Circulation. 2005;111:2605–10.
South AM, Diz D, Chappell MC. COVID-19, ACE2 and the cardiovascular consequences. Am J Physiol Heart Circ Physiol. 2020;318:1084–90.
Ye R, Liu Z. ACE2 exhibits protective effects against LPS-induced acute lung injury in mice by inhibiting the LPS-TLR4 pathway. Exp Mol Pathol. 2019;113:104350.
Danser AHJ, Epstein M, Batlle D. Renin-angiotensin system blockers and the COVID-19 pandemic: at present there is no evidence to abandon renin-angiotensin system blockers. Hypertension. 2020;75:1382–5.
Vaduganathan M, Vardeny O, Michel T, et al. Renin-angiotensin-aldosterone system inhibitors in patients with COVID-19. N Engl J Med. 2020;382:1653–9.
de Wit E, van Doremalen N, Falzarano D, et al. SARS and MERS: recent insights into emerging coronaviruses. Nat Rev Microbiol. 2016;14(8):523–34.
Cortegiani A, Ingoglia G, Ippolito M, et al. A systematic review on the efficacy and safety of chloroquine for the treatment of COVID-19. J Crit Care. 2020;57:279–83.
Castelli V, Cimini A, Claudio F. Cytokine storm in COVID-19: “When you come out of the storm, you won’t be the same person who walked in.” Front Immunol. 2020;11:2132.
Sui J, Li W, Murakami A, et al. Potent neutralization of severe acute respiratory syndrome (SARS) coronavirus by a human mAb to S1 protein that blocks receptor association. PNAS. 2004;101(8):2536–41.
Khan A, Benthin C, Zeno B, et al. A pilot clinical trial of recombinant human angiotensin-converting enzyme 2 in acute respiratory distress syndrome. Crit Care. 2017;21:234.
Laccarino G, Borghi C, Cicero AFG, et al. Renin-Angiotensin System inhibition in cardiovascular patients at the time of COVID-19: much Ado for nothing? A statement of activity from the directors of the board and the scientific directors of the Italian Society of Hypertension. High Blood Press Cardiovasc Prev. 2020;27:105–8.
Edmonston DL, South AM, Sparks MA, et al. Coronavirus disease 2019 and hypertension: the role of angiotensin-converting enzyme2 and the renin–angiotensin-system. Adv Chronic Kidney Dis. 2020. https://doi.org/10.1053/j.ackd.2020.07.002.
Lopes RD, Macedo AVS, Barros-e-Silva PGM, et al. Continuing versus suspending angiotensin-converting enzyme inhibitors and angiotensin receptor blockers: impact on adverse outcomes in hospitalized patients with severe acute respiratory syndrome coronavirus2 (SARS-CoV-2)-The BRACE CORONA Trial. Am Heart J. 2020;226:49–59.
Zheng YY, Ma YT, Zhang JY, et al. COVID-19 and the cardiovascular system. Nat Rev Cardiol. 2020;17(5):259–60.
Sommerstein R, Kochen MM, Messerli FH, et al. Coronavirus disease 2019 (COVID-19): do angiotensin-converting enzyme inhibitors/angiotensin receptor blockers have a biphasic effect? J Am Heart Assoc. 2020;9(7):e016509.
Imai Y, Kuba K, Rao S, et al. Angiotensin-converting enzyme 2 protects from severe acute lung failure. Nature. 2005;436(7047):112–6.
Zhang P, Zhu L, Cai J, et al. Association of inpatient use of angiotensin converting enzyme inhibitors and angiotensin II receptor blockers with mortality among patients with hypertension hospitalized with COVID-19. Circ Res. 2020;126(12):1671–81.
Gao C, Cai Y, Zhang K, et al. Association of hypertension and antihypertensive treatment with COVID-19 mortality: a retrospective observational study. Eur Heart J. 2020;41(22):2058–66.
Lam KW, Chow KW, Vo J, et al. Continued in-hospital angiotensin-converting enzyme inhibitor and angiotensin II receptor blocker use in hypertensive COVID-19 patients is associated with positive clinical outcome. J Infect Dis. 2020;222(8):1256–64.
Meng J, Xiao G, Zhang J, et al. Renin-angiotensin system inhibitors improve the clinical outcomes of COVID-19 patients with hypertension. Emerg Microbes Infect. 2020;9(1):757–60.
Fosbøl EL, Butt JH, Østergaard L, et al. Association of angiotensin-converting enzyme inhibitor or angiotensin receptor blocker use with COVID-19 diagnosis and mortality. JAMA. 2020;324(2):168–77.
Reynolds HR, Adhikari S, Pulgarin C, et al. Renin–angiotensin–aldosterone system inhibitors and risk of covid-19. New Engl J Med. 2020;382(25):2441–8.
Iaccarino G, Grassi G, Borghi C, et al. Age and multimorbidity predict death among COVID-19 patients: results of the SARS-RAS study of the italian society of hypertension. Hypertension. 2020;76(2):366–72.
de Abajo FJ, Rodríguez-Martín S, Lerma V, et al. Use of renin-angiotensin-aldosterone system inhibitors and risk of COVID-19 requiring admission to hospital: a case-population study. Lancet. 2020;395(10238):1705–14.
Jung SY, Choi JC, You SH, et al. Association of renin-angiotensin-aldosterone system inhibitors with COVID-19-related outcomes in Korea: a nationwide population-based cohort study. Clin Infect Dis. 2020. https://doi.org/10.1093/cid/ciaa624.
Hoffmann M, Kleine-Weber H, Krüger N, et al. The novel coronavirus 2019 (2019-nCoV) uses the SARS-coronavirus receptor ACE2 and the cellular protease TMPRSS2 for entry into target cells. bioRxiv. 2020. https://doi.org/10.1101/2020.01.31.929042
Kawase M, Shirato K, van der Hoek L, et al. Simultaneous treatment of human bronchial epithelial cells with serine and cysteine protease inhibitors prevents severe acute respiratory syndrome coronavirus entry. J Virol. 2012;86:6537–45.
Zhou Y, Vedantham P, Lu K, et al. Protease inhibitors targeting coronavirus and filovirus entry. Antiviral Res. 2015;116:76–84.
European Society of Cardiology [Internet]. Sophia Antipolis (France). [cited 2020 Mar 13]. Position statement of the ESC Council on hypertension on ACE-inhibitors and angiotensin receptor blockers. Available from: https://www.escardio.org/Councils/Council-on-Hypertension-%A0(CHT)/News/position-statement-of-the-esc-council-on-hypertension-on-ace-inhibitors-%A0and-ang.
American College of cardiology [Internet]. Washington (DC). [cited 2020 Mar 17]. HFSA/ACC/AHA Statement Addresses Concerns Re: using RAAS Antagonists in COVID-19. Available from: https://www.acc.org/latest-in-cardiology/articles/2020/03/17/08/59/hfsa-acc-aha-statement-addresses-concerns-re-using-raas-antagonists-in-covid-19.
Acknowledgements
The authors would like to acknowledge Centre of Excellence (COE), Higher Education, Government of Uttar Pradesh, Lucknow, India. The authors are also grateful to DST-FIST-PURSE, New Delhi, India.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
This study was not funded by any funding agency.
Conflict of interest
The authors declare no conflict of interest.
Ethics approval
Not applicable.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Availability of data and materials
Not applicable.
Code availability
Not applicable.
Contributors
First author contributed in conceptual writing and author two contributed in conceptual writing, critical reading and reviewing the article.
Rights and permissions
About this article
Cite this article
Shukla, A.K., Banerjee, M. Angiotensin-Converting-Enzyme 2 and Renin-Angiotensin System Inhibitors in COVID-19: An Update. High Blood Press Cardiovasc Prev 28, 129–139 (2021). https://doi.org/10.1007/s40292-021-00439-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40292-021-00439-9