Abstract
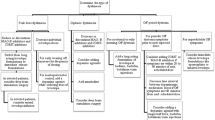
Dyskinesias encompass a variety of different hyperkinetic phenomenologies, particularly chorea, dystonia, stereotypies, and akathisia. The main types of drug-induced dyskinesias include levodopa-induced dyskinesia (LID) in patients with Parkinson’s disease and tardive syndrome (TS), typically present in patients with psychiatric or gastrointenstinal disorders treated with dopamine receptor blocking drugs, also referred to as neuroleptics. Besides preventive measures (i.e., avoiding the use of the offending drugs), general treatment strategies include slow taper of the offending agent and use of dopamine-depleting agents like tetrabenazine. Botulinum toxin may be helpful for wearing off focal dystonia and some forms of tardive dystonia. Deep brain stimulation is usually reserved for patients with disabling motor fluctuations, LID, and for severe TS that cannot be managed medically.
Similar content being viewed by others
References
Loonen AJMAJ. New insights into the mechanism of drug-induced dyskinesia. CNS spectrums. 2013;18(1):15.
Walters AS, McHale D, Sage JI, Hening WA, Bergen M. A blinded study of the suppressibility of involuntary movements in Huntington’s chorea, tardive dyskinesia, and L-dopa-induced chorea. Clin Neuropharmacol. 1990;13(3):236–40.
Waln O, Jankovic J. An update on tardive dyskinesia: from phenomenology to treatment. Tremor Other Hyperkinet Mov. 2013;12(3):03–161.
Reich SG. Pearls: hyperkinetic movement disorders. Semin Neurol. 2010;30(1):15–22.
Cloud LJ, Zutshi D, Factor SA. Tardive dyskinesia: therapeutic options for an increasingly common disorder. Neurotherapeutics. 2014;11(1):166–76.
Patel N, Jankovic J, Hallett M. Sensory aspects of movement disorders. Lancet Neurol. 2014;13(1):100–12.
Anand VS, Dewan MJ. Withdrawal-emergent dyskinesia in a patient on risperidone undergoing dosage reduction. Ann Clin Psychiatry. 1996;8(3):179–82.
American Psychiatric Association. Medication-induced movement disorders and other adverse effects of medication. Diagnostic and statistical manual of mental disorders, Fifth edn: American Psychiatric Association; 2013.
Mejia NI, Jankovic J. Tardive dyskinesia and withdrawal emergent syndrome in children. Expert Rev Neurother. 2010;10(6):893–901.
Karama S, Lal S. Tardive dyskinesia following brief exposure to risperidone–a case study. Eur Psychiatry. 2004;19(6):391–2.
O’Brien A. Comparing the risk of tardive dyskinesia in older adults with first-generation and second-generation antipsychotics: a systematic review and meta-analysis. Int J Geriatr Psychiatry. 2015. doi:10.1002/gps.4399
Novick D, Haro JM, Bertsch J, Haddad PM. Incidence of extrapyramidal symptoms and tardive dyskinesia in schizophrenia: thirty-six-month results from the European schizophrenia outpatient health outcomes study. J Clin Psychopharmacol. 2010;30(5):531–40.
Aquino CC, Lang AE. Tardive dyskinesia syndromes: current concepts. Parkinsonism Relat Disord. 2014;20(Suppl 1):S113–7.
Kiriakakis V, Bhatia KP, Quinn NP, Marsden CD. The natural history of tardive dystonia. A long-term follow-up study of 107 cases. Brain J Neurol. 1998;121(Pt 11):2053–66.
Correll CU, Schenk EM. Tardive dyskinesia and new antipsychotics. Curr Opin Psychiatry. 2008;21(2):151–6.
Jankovic J. Bradley’s Neurology in Clinical Practice 2012.
Lehosit JBP, Fermina, Dhanya V, Leslie C. Tardive Dyskinesia. Wiley: Chichester. 2015.
Correll CUCU. Tardive dyskinesia and new antipsychotics. Curr Opin Psychiatry. 2008;21(2):151–6.
Correll CU, Leucht S, Kane JM. Lower risk for tardive dyskinesia associated with second-generation antipsychotics: a systematic review of 1-year studies. Am J Psychiatry. 2004;161(3):414–25.
Kinon BJ, Kollack-Walker S, Jeste D, Gupta S, Chen L, Case M, et al. Incidence of tardive dyskinesia in older adult patients treated with olanzapine or conventional antipsychotics. J Geriatr Psychiatry Neurol. 2015;28(1):67–79.
Lee PE, Sykora K, Gill SS, Mamdani M, Marras C, Anderson G, et al. Antipsychotic medications and drug-induced movement disorders other than parkinsonism: a population-based cohort study in older adults. J Am Geriatr Soc. 2005;53(8):1374–9.
Rosenheck RA. Evaluating the cost-effectiveness of reduced tardive dyskinesia with second-generation antipsychotics. Br J Psychiatry J Mental Sci. 2007;191:238–45.
Rosenheck R, Cramer J, Xu W, Thomas J, Henderson W, Frisman L, et al. A comparison of clozapine and haloperidol in hospitalized patients with refractory schizophrenia. Department of Veterans Affairs Cooperative Study Group on Clozapine in Refractory Schizophrenia. N Engl J Med. 1997;337(12):809–15.
Rosenheck R, Perlick D, Bingham S, Liu-Mares W, Collins J, Warren S, et al. Effectiveness and cost of olanzapine and haloperidol in the treatment of schizophrenia: a randomized controlled trial. JAMA. 2003;290(20):2693–702.
Obeso JA. The Movement Disorders journal 2016 and onward. Mov Disord. 2016;31(1):1–2 (Epub 2016/01/11).
Adrianzen C, Arango-Davila C, Araujo DM, Ruiz I, Walton RJ, Dossenbach M, et al. Relative association of treatment-emergent adverse events with quality of life of patients with schizophrenia: post hoc analysis from a 3-year observational study. Hum Psychopharmacol. 2010;25(6):439–47.
Farah A. Atypicality of atypical antipsychotics. Prim Care Companion J Clin Psychiatry. 2005;7(6):268–74.
Kapur S, Seeman P. Does fast dissociation from the dopamine d(2) receptor explain the action of atypical antipsychotics?: A new hypothesis. Am J Psychiatry. 2001;158(3):360–9.
Kapur S, Zipursky R, Jones C, Shammi CS, Remington G, Seeman P. A positron emission tomography study of quetiapine in schizophrenia: a preliminary finding of an antipsychotic effect with only transiently high dopamine D2 receptor occupancy. Arch Gen Psychiatry. 2000;57(6):553–9.
Standaert DG, Galanter JM. Principles of Pharmacology: The Pathophysiologic Basis of Drug Therapy: Lippincott Williams & Wilkins, 2008.
Pena MS, Yaltho TC, Jankovic J. Tardive dyskinesia and other movement disorders secondary to aripiprazole. Mov Disord. 2011;26(1):147–52.
Casey DE. Tardive dyskinesia: pathophysiology and animal models. J Clin Psychiatry. 2000;4:5–9.
Corson PW, Nopoulos P, Miller DD, Arndt S, Andreasen NC. Change in basal ganglia volume over 2 years in patients with schizophrenia: typical versus atypical neuroleptics. Am J Psychiatry. 1999;156(8):1200–4.
Ogawa T, Nagao T, Kashiwabara K, Fujiwara Y, Harada T, Otsuki S. Tardive dyskinesia and neurotransmitters: effects of sodium valproate, cyproheptadine, oxypertine, hydroxyzine pamoate and Ca-hopantenate on monoamine metabolites, cyclic nucleotides and gamma-aminobutyric acid in human cerebrospinal fluid. Clin Ther. 1984:1–17.
Mizuno Y. Parkinson’s Disease: CRC Press, 2012.
Aia PG, Revuelta GJ, Cloud LJ, Factor SA. Tardive dyskinesia. Curr Treat Options Neurol. 2011;13(3):231–41.
Glazer WM, Morgenstern H, Schooler N, Berkman CS, Moore DC. Predictors of improvement in tardive dyskinesia following discontinuation of neuroleptic medication. Br J Psychiatry. 1990;157:585–92.
Modestin J, Wehrli MV, Stephan PL, Agarwalla P. Evolution of neuroleptic-induced extrapyramidal syndromes under long-term neuroleptic treatment. Schizophr Res. 2008;100(1–3):97–107.
Fernandez HH, Krupp B, Friedman JH. The course of tardive dyskinesia and parkinsonism in psychiatric inpatients: 14-year follow-up. Neurology. 2001;56(6):805–7.
Viguera AC, Baldessarini RJ, Hegarty JD, van Kammen DP, Tohen M. Clinical risk following abrupt and gradual withdrawal of maintenance neuroleptic treatment. Arch Gen Psychiatry. 1997;54(1):49–55.
Bhidayasiri R, Fahn S, Weiner WJ, Gronseth GS, Sullivan KL, Zesiewicz TA. Evidence-based guideline: treatment of tardive syndromes: report of the Guideline Development Subcommittee of the American Academy of Neurology. Neurology. 2013;81(5):463–9.
Hazari N, Kate N, Grover S. Clozapine and tardive movement disorders: a review. Asian J Psychiatr. 2013;6(6):439–51.
Jankovic J, Clarence-Smith K. Tetrabenazine for the treatment of chorea and other hyperkinetic movement disorders. Expert Rev Neurother. 2011;11(11):1509–23.
Ondo WG, Hanna PA, Jankovic J. Tetrabenazine treatment for tardive dyskinesia: assessment by randomized videotape protocol. Am J Psychiatry. 1999;156(8):1279–81.
Kazamatsuri H, Chien CP, Cole JO. Long-term treatment of tardive dyskinesia with haloperidol and tetrabenazine. Am J Psychiatry. 1973;130(4):479–83.
Chen JJ, Ondo WG, Dashtipour K, Swope DM. Tetrabenazine for the treatment of hyperkinetic movement disorders: a review of the literature. Clin Ther. 2012;34(7):1487–504.
Jankovic J, Orman J. Tetrabenazine therapy of dystonia, chorea, tics, and other dyskinesias. Neurology. 1988;38(3):391–4.
Shen V, Clarence-Smith K, Hunter C, Jankovic J. Safety and efficacy of tetrabenazine and use of concomitant medications during long-term, open-label treatment of chorea associated with huntington’s and other diseases. Tremor Other Hyperkinet Mov. 2013;22(3):03–191.
Kenney C, Hunter C, Mejia N, Jankovic J. Is history of depression a contraindication to treatment with tetrabenazine? Clin Neuropharmacol. 2006;29(5):259–64 (Epub 2006/09/09).
Stamler D, Bradbury M, Brown F, editors. The pharmacokinetics and safety of deuterated-tetrabenazine. 65th AAN Annual Meeting; 2013; San Diego: Neurology.
Muller T. Valbenazine granted breakthrough drug status for treating tardive dyskinesia. Expert opinion on investigational drugs. 2015;24(6):737–42 (Epub 2015/03/27).
O’Brien CF, Jimenez R, Hauser RA, Factor SA, Burke J, Mandri D, et al. NBI-98854, a selective monoamine transport inhibitor for the treatment of tardive dyskinesia: A randomized, double-blind, placebo-controlled study. Mov Disord. 2015;30(12):1681–7.
Thaker GK, Nguyen JA, Strauss ME, Jacobson R, Kaup BA, Tamminga CA. Clonazepam treatment of tardive dyskinesia: a practical GABAmimetic strategy. Am J Psychiatry. 1990;147(4):445–51.
Woods SW, Saksa JR, Baker CB, Cohen SJ, Tek C. Effects of levetiracetam on tardive dyskinesia: a randomized, double-blind, placebo-controlled study. J Clin Psychiatry. 2008;69(4):546–54.
Libov I, Miodownik C, Bersudsky Y, Dwolatzky T, Lerner V. Efficacy of piracetam in the treatment of tardive dyskinesia in schizophrenic patients: a randomized, double-blind, placebo-controlled crossover study. J Clin Psychiatry. 2007;68(7):1031–7.
Zhang WF, Tan YL, Zhang XY, Chan RC, Wu HR, Zhou DF. Extract of Ginkgo biloba treatment for tardive dyskinesia in schizophrenia: a randomized, double-blind, placebo-controlled trial. J Clin Psychiatry. 2011;72(5):615–21.
Schrodt GR Jr, Wright JH, Simpson R, Moore DP, Chase S. Treatment of tardive dyskinesia with propranolol. J Clin Psychiatry. 1982;43(8):328–31.
Waln OO. Zolpidem improves tardive dyskinesia and akathisia. Mov Disord. 2013;28(12):1748–9.
Yasui-Furukori N, Kikuchi A, Katagai H, Kaneko S. The effects of electroconvulsive therapy on tardive dystonia or dyskinesia induced by psychotropic medication: a retrospective study. Neuropsychiatr Dis Treat. 2014;10:1209–12.
Peng LY, Lee Y, Lin PY. Electroconvulsive therapy for a patient with persistent tardive dyskinesia: a case report and literature review. J Ect. 2013;29(3).
Malek-Ahmadi P, Weddige RL. Tardive Dyskinesia and Electroconvulsive Therapy. Convuls Ther. 1988;4(4):328–31.
Hennings JM, Krause E, Botzel K, Wetter TC. Successful treatment of tardive lingual dystonia with botulinum toxin: case report and review of the literature. Prog Neuropsychopharmacol Biol Psychiatry. 2008;32(5):1167–71.
Beckmann YY, Secil Y, Saka S, Kuserli A, Ciftci Y. Treatment of intractable tardive lingual dyskinesia with botulinum toxin. J Clin Psychopharmacol. 2011;31(2):250–1 (Epub 2011/03/03).
Rapaport A, Sadeh M, Stein D, Levine J, Sirota P, Mosheva T, et al. Botulinum toxin for the treatment of oro-facial-lingual-masticatory tardive dyskinesia. Mov Disord. 2000;15(2):352–5.
Tschopp L, Salazar Z, Micheli F. Botulinum toxin in painful tardive dyskinesia. Clin Neuropharmacol. 2009;32(3):165–6.
van Harten PN, Hovestadt A. Botulinum toxin as a treatment for tardive dyskinesia. Mov Disord. 2006;21(8):1276–7.
Spindler MA, Galifianakis NB, Wilkinson JR, Duda JE. Globus pallidus interna deep brain stimulation for tardive dyskinesia: case report and review of the literature. Parkinsonism Relat Disord. 2013;19(2):141–7.
Kefalopoulou Z, Paschali A, Markaki E, Vassilakos P, Ellul J, Constantoyannis C. A double-blind study on a patient with tardive dyskinesia treated with pallidal deep brain stimulation. Acta Neurol Scand. 2009;119(4):269–73.
Damier P, Thobois S, Witjas T, Cuny E, Derost P, Raoul S, et al. Bilateral deep brain stimulation of the globus pallidus to treat tardive dyskinesia. Arch Gen Psychiatry. 2007;64(2):170–6.
Sun B, Chen S, Zhan S, Le W, Krahl SE. Subthalamic nucleus stimulation for primary dystonia and tardive dystonia. Acta Neurochir Suppl. 2007;97(Pt 2):207–14.
Zhang JG, Zhang K, Wang ZC, Ge M, Ma Y. Deep brain stimulation in the treatment of secondary dystonia. Chin Med J. 2006;119(24):2069–74.
Lorberboym M, Treves TA, Melamed E, Lampl Y, Hellmann M, Djaldetti R. [123I]-FP/CIT SPECT imaging for distinguishing drug-induced parkinsonism from Parkinson’s disease. Mov Disord. 2006;21(4):510–4.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
None.
Conflict of interest
Dr. Jankovic has received grants from Adamas Pharmaceuticals, Inc.; Allergan, Inc.; CHDI Foundation; Civitas/Acorda Therapeutics; Huntington Study Group; Ipsen Limited; Kyowa Haako Kirin Pharma, Inc.; Lundbeck Inc.; Medtronic; Merz Pharmaceuticals; Michael J. Fox Foundation for Parkinson Research; National Institutes of Health; National Parkinson Foundation; Omeros Corporation; Parkinson Study Group; Pfizer; Prothena Biosciences Inc.; Psyadon Pharmaceuticals, Inc.; St. Jude Medical; and Teva Pharmaceutical Industries Ltd. Dr. Jankovic receives consulting fees from Adamas Pharmaceuticals, Inc.; Allergan, Inc.; and Teva Pharmaceutical Industries Ltd. Dr. Jankovic has received royalties from Cambridge; Elsevier; Future Science Group; Hodder Arnold; Lippincott Williams and Wilkins; and Wiley-Blackwell. Dhanya Vijayakumar, MD, has no conflicts of interest to declare.
Rights and permissions
About this article
Cite this article
Vijayakumar, D., Jankovic, J. Drug-Induced Dyskinesia, Part 2: Treatment of Tardive Dyskinesia. Drugs 76, 779–787 (2016). https://doi.org/10.1007/s40265-016-0568-1
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40265-016-0568-1