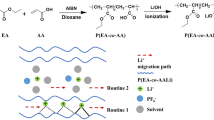
Abstract
Li metal possesses a high theoretical specific capacity, high electronic conductivity, and a low electrochemical potential, making it a promising anode material for building next-generation rechargeable metal batteries. In case conventional liquid electrolytes were used, and the anode using Li metal has been hindered by unstable(electro)chemistry at Li/electrolyte interface and the accompanied dendrite issue. Specifically, for the Li-Se batteries, the dissolution and shuttle of polyselenide intermediates lead to the deposition of poorly-conductive species on the anode, which further aggravates the chemical environment at the anode. In this work, we proposed to stabilize the Li-Se electrochemistry by constructing a gel polymer electrolyte via in situ gelations of conventional ether-based electrolytes at room temperature. The results demonstrate that the in situ gelated electrolyte helps to build electrochemically stable electrode/electrolyte interfaces and promote the efficient transfer of charge carriers across the interface. Compared with the liquid electrolytes, the gelated electrolyte shows improved chemical compatibility with the Li metal anode, which effectively alleviates the unfavorable side reactions and dendrite formation at the anode/electrolyte interface, and the polyselenide shuttle from the cathode to the anode. As a result, the Li-Se battery shows a higher Coulombic efficiency and improved cycling performance.
Similar content being viewed by others
References
Evarts E. C., Nature, 2015, 526, S93
Wu F., Yushin G., Energy Environ. Sci., 2017, 10, 435
Wang H., Tang Y., Chem. Res. Chinese Universities, 2020, 36(3), 402
Xu G. L., Liu J., Amine R., Chen Z., Amine K., ACS Energy Lett., 2017, 2, 605
Zhang W., Yu H. C., Wu L., Liu H., Abdellahi A., Qiu B., Bai J., Orvananos B., Strobridge F. C., Zhou X., Liu Z., Ceder G., Zhu Y., Thornton K., Grey C. P., Wang F., Sci. Adv. 2018, 4, aao2608
Ding J. F., Xu R., Yan C., Xiao Y., Liang Y. R., Yuan H., Huang J. Q., Chin. Chem. Lett., 2020, 31, 2339
Xi X. T., Li W. H., Hou B. H., Yang Y., Gu Z. Y., Wu X. L., ACS Appl. Energy Mater., 2019, 2, 201
Pu K. C., Zhang X., Qu X. L. Hu J. J., Li H. W., Gao M. X., Pan H. G., Liu Y. F., Rare Met., 2020, 39, 616
Xi X. T., Feng X., Nie X. J., Hou B. H., Li W. H., Yang X., Yang A. B., Sun W. D., Wu X. L., Chem. Commun., 2019, 55, 8406
Liu Y., He P., Zhou H., Adv. Energy Mater., 2018, 8, 201701602
Abouimrane A., Dambournet D., Chapman K. W., Chupas P. J., Weng W., Amine K., J. Am. Chem. Soc., 2012, 10, 4505
Wang Y. H., Li X. T., Wang W. P., Yan H. J., Xin S., Guo Y. G., Sci. China Chem., 2020, 63, 1402
Zheng J. M., Engelhard M. H., Mei D. H., Jiao S. H., Polzin B. J., Zhang J. G., Xu W., Nat. Energy, 2017, 2, 17012
Pan H., Han K. S., Engelhard M. H., Cao R., Chen J., Zhang J. G., Mueller K. T., Shao Y., Liu J., Adv. Funct. Mater., 2018, 28, 1870275
Yuan H., Liu J., Lu Y., Zhao C. Z., Cheng X. B., Nan H. X., Liu Q. B., Huang J. Q., Zhang Q., Chem. Res. Chinese Universities, 2020, 36(3), 377
Chen Y., Zhang W., Zhou D., Tian H., Su D., Wang C., Stockdale D., Kang F., Li B., Wang G., ACS Nano, 2019, 13, 4731
Zhou G. M., Pei S. F., Li L., Wang D. W., Wang S. G., Huang K., Yin L. C., Li F., Cheng H. M., Adv. Mater., 2014, 26, 625
Liu J., Zhang L., Li H., Zhao P., Ren P., Shi W., Cheng P., Sci. China Chem., 2019, 62, 602
Dong Y., Ben T., Chem. Res. Chinese Universities, 2019, 35(4), 654
Rahul J., Islam M. M., Nanoscale, 2020, 12, 14087
Yang C. P., Yin Y. X., Guo Y. G., J. Phys. Chem. Lett., 2015, 6, 256
Xin S., Chang Z., Zhang X., Guo Y. G., Natl. Sci. Rev., 2017, 4, 54
Liu Y. Y., Lin D. C., Yuen P. Y., Liu K., Xie J., Dauskardt R. H., Cui Y., Adv. Mater., 2017, 29, 1605531
Ye H., Xin S., Yin Y. X., Li J. Y., Guo Y. G., Wan L. J., J. Am. Chem. Soc., 2017, 139, 5916
Peng H. J., Huang J. Q., Zhang Q., Chem. Soc. Rev., 2017, 46, 5237
Goodenough J. B., Gao H. A., Sci. China Chem., 2019, 62, 1555
Ma L., Kim M. S., Archer L. A., Chem. Mater., 2017, 29, 4181
Hu L. B., Xu K., Proc. Natl. Acad. Sci. USA, 2014, 111, 3205
Liu F. Q., Wang W. P., Yin Y. X., Zhang S. F., Shi J. L., Wang L., Zhang X. D., Zheng Y., Zhou J. J., Li L., Guo Y. G., Sci. Adv., 2018, 4, aat5383
Gao S., Wang K. L., Wang R. X., Jiang M., Han J., Gu T. T., Cheng S. J., Jiang K., J. Mater. Chem. A, 2017, 5, 17889
Xu K., Chem. Rev., 2004, 104, 4303
Zhang Q., Wang Y., She Z. W., Fu Z., Zhang R., Cui Y., Nano Lett., 2015, 15, 3780
Chai J. C., Liu Z. H., Ma J., Wang J., Liu X. C., Liu H. S., Zhang J. J., Cui G. L., Chen L. Q., Adv. Sci., 2017, 4, 1600377
Liu M., Zhou D., He Y. B., Fu Y., Qin X., Miao C., Du H., Li B., Yang Q. H., Lin Z., Zhao T. S., Kang F., Nano Energy, 2016, 22, 278
Ali R., Farah A., Binkhathlan Z., Saudi Pharm. J., 2017, 25, 258
Wang K., Wu Y., Liu K., Wu H., Chem. Res. Chinese Universities, 2020, 36(3), 351
Cai D., Liu B., Zhu D., Chen D., Lu M., Cao J., Wang Y., Huang W., Shao Y., Tu H., Han W., Adv. Energy Mater., 2020, 10, 1904273
Zhou Y., Li Z., Lu Y. C., Nano Energy, 2017, 39, 554
Xu G., Kushima A., Yuan J., Dou H., Xue W., Zhang, Yan X., Li J., Energy Environ. Sci., 2017, 10, 2544
Zhao Z., Qin D., Wang S., Chen G., Li Z., Electrochim. Acta, 2014, 127, 123
Guo J., Yang Z., Yu Y., Abruna H. D., Archer L. A., J. Am. Chem. Soc., 2013, 135, 763
Zhang S., Ueno K., Dokko K., Watanabe M., Adv. Energy Mater., 2015, 5, 201500117
Zhang S. S., Tran D. T., Electrochimica Acta, 2013, 114, 296
Yang C. P., Yin Y. X., Guo Y. G., Wan L. J., J. Am. Chem. Soc., 2015, 137, 2215
Wang L. N., Liu J. Y., Yuan S. Y., Wang Y. G., Xia Y. Y., Energy Environ. Sci., 2016, 9, 224
Acknowledgements
This work was supported by the National Key R&D Program of China(No. 2016YFA0202500), the National Natural Science Foundation of China(Nos. 21975266, 21805062), and the Beijing National Laboratory for Molecular Sciences, China(No.BNLMS-CXXM-201906).
Author information
Authors and Affiliations
Corresponding authors
Additional information
Conflicts of Interest
The authors declare no conflicts of interest.
Rights and permissions
About this article
Cite this article
Wang, WP., Zhang, J., Li, XT. et al. Stabilizing the Electrochemistry of Lithium-Selenium Battery via In situ Gelated Polymer Electrolyte: A Look from Anode. Chem. Res. Chin. Univ. 37, 298–303 (2021). https://doi.org/10.1007/s40242-021-0448-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40242-021-0448-4