Abstract
Purpose of Review
To understand the role of neprilysin inhibition in the management of chronic heart failure with reduced ejection fraction (HFrEF) and review effects of neprilysin inhibition on concentrations of natriuretic peptides and other biomarkers.
Recent Findings
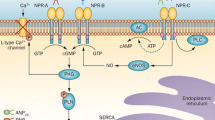
Neprilysin inhibition improves cardiovascular outcomes in patients with chronic HFrEF. As bioactive natriuretic peptides are degraded by neprilysin, treatment with sacubitril/valsartan results in an increase in concentrations of atrial natriuretic peptide (ANP), B-type natriuretic peptide (BNP), and C-type natriuretic peptide (CNP). In contrast, neprilysin inhibition led to reduction in concentrations of N-terminal pro-B-type natriuretic peptide (NT-proBNP). Reduction in NT-proBNP to ≤ 1000 pg/mL with neprilysin inhibition improved cardiovascular outcomes in a recent analysis. Other biomarkers may be affected by neprilysin inhibition.
Summary
Neprilysin inhibition results in an increase in ANP, BNP, and CNP with corresponding reduction in NT-proBNP concentrations. Other biomarkers may be similarly affected. Given widespread clinical measurement, more data are needed to better understand potential impact on neprilysin inhibition on ability to interpret BNP.

Similar content being viewed by others
References
Papers of particular interest, published recently, have been highlighted as: • Of importance •• Of major importance
• McMurray JJV, Packer M, Desai AS, et al. Angiotensin–neprilysin inhibition versus enalapril in heart failure. New England Journal of Medicine. 2014;371(11):993–1004. The largest and first trial in a decade to give us a medication that improved mortality in patients with heart failure with reduced ejection fraction- sacubitril/valsartan. https://doi.org/10.1056/NEJMoa1409077.
• Yancy CW, Jessup M, Bozkurt B, Butler J, Casey d Jr, Colvin MM, et al. 2016 ACC/AHA/HFSA focused update on new pharmacological therapy for heart failure: an update of the 2013 ACCF/AHA Guideline for the Management of Heart FailureA Report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines and the Heart Failure Society of America. J Am Coll Cardiol. 2016;68(13):1476–88. The guideline that incorporated sacubitril/valsartan into the treatment strategy of patients with heart failure with reduced ejection fraction. https://doi.org/10.1016/j.jacc.2016.05.011.
Yancy CW, Jessup M, Bozkurt B, Butler J, Casey d Jr, Drazner MH, et al. 2013 ACCF/AHA guideline for the management of heart failure: executive summary A report of the American College of Cardiology Foundation/American Heart Association Task Force on Practice Guidelines. J Am Coll Cardiol. 2013;62(16):1495–539. https://doi.org/10.1016/j.jacc.2013.05.020.
Troughton RW, Frampton CM, Brunner-La Rocca H-P, Pfisterer M, Eurlings LWM, Erntell H, et al. Effect of B-type natriuretic peptide-guided treatment of chronic heart failure on total mortality and hospitalization: an individual patient meta-analysis. Eur Heart J. 2014;35(23):1559–67. https://doi.org/10.1093/eurheartj/ehu090.
Savarese G, Trimarco B, Dellegrottaglie S, et al. Natriuretic peptide-guided therapy in chronic heart failure: a meta-analysis of 2,686 patients in 12 randomized trials. PLoS One. 2013;8:e58287.
Langenickel TH, Dole WP. Angiotensin receptor-neprilysin inhibition with LCZ696: a novel approach for the treatment of heart failure. Drug Discovery Today: Therapeutic Strategies. 2012;9:e131–9.
McMurray JJV. Neprilysin inhibition to treat heart failure: a tale of science, serendipity, and second chances. Eur J Heart Fail. 2015;17(3):242–7. https://doi.org/10.1002/ejhf.250.
Vanneste Y, Michel A, Dimaline R, Najdovski T, Deschodt-Lanckman M. Hydrolysis of alpha-human atrial natriuretic peptide in vitro by human kidney membranes and purified endopeptidase-24.11. Evidence for a novel cleavage site. Biochem J. 1988;254(2):531–7. https://doi.org/10.1042/bj2540531.
Chen Y, Burnett JC. Biochemistry, therapeutics, and biomarker implications of neprilysin in cardiorenal disease. Clin Chem. 2017;63(1):108–15. https://doi.org/10.1373/clinchem.2016.262907.
Vardeny O, Miller R, Solomon SD. Combined neprilysin and renin-angiotensin system inhibition for the treatment of heart failure. JACC: Heart Failure. 2014;2:663–70.
Ibrahim NE, Januzzi JL. Beyond natriuretic peptides for diagnosis and management of heart failure. Clin Chem. 2017;63(1):211–22. https://doi.org/10.1373/clinchem.2016.259564.
de Bold AJ, de Bold MLK. Determinants of natriuretic peptide production by the heart: basic and clinical implications. J Investig Med: Off Publ Am Fed Clin Res. 2005;53(7):371–7. https://doi.org/10.2310/6650.2005.53710.
Vodovar N, Séronde M-F, Laribi S, Gayat E, Lassus J, Boukef R, et al. Post-translational modifications enhance NT-proBNP and BNP production in acute decompensated heart failure. Eur Heart J. 2014;35(48):3434–41. https://doi.org/10.1093/eurheartj/ehu314.
Ibrahim N, Januzzi JL. The potential role of natriuretic peptides and other biomarkers in heart failure diagnosis, prognosis and management. Expert Rev Cardiovasc Ther. 2015:1–14.
Ichiki T, Huntley BK, Sangaralingham SJ, Burnett JC. Pro-atrial natriuretic peptide: a novel guanylyl cyclase-A receptor activator which goes beyond atrial and B-type natriuretic peptides JACC heart failure 2015;3:715–723.
D'Elia E, Iacovoni A, Vaduganathan M, Lorini FL, Perlini S, Senni M. Neprilysin inhibition in heart failure: mechanisms and substrates beyond modulating natriuretic peptides. European Journal of Heart Failure 2017:n/a-n/a.
Hornig B, Kohler C, Drexler H. Role of bradykinin in mediating vascular effects of angiotensin-converting enzyme inhibitors in humans. Circulation. 1997;95(5):1115–8. https://doi.org/10.1161/01.CIR.95.5.1115.
Craig TJ, Bernstein JA, Farkas H, Bouillet L, Boccon-Gibod I. Diagnosis and treatment of bradykinin-mediated angioedema: outcomes from an angioedema expert consensus meeting. Int Arch Allergy Immunol. 2014;165(2):119–27. https://doi.org/10.1159/000368404.
O'Connor TM, O'Connell J, O'Brien DI, Goode T, Bredin CP, Shanahan F. The role of substance P in inflammatory disease. J Cell Physiol. 2004;201(2):167–80. https://doi.org/10.1002/jcp.20061.
Kato J, Tsuruda T, Kita T, Kitamura K, Eto T. Adrenomedullin. A protective factor for blood vessels 2005;25:2480–2487.
Taubman MB. Angiotensin II. A vasoactive hormone with ever-increasing biological roles 2003;92:9–11.
Januzzi JL Jr, Ibrahim NE. Renin-angiotensin system blockade in heart failure: more to the picture than meets the eye∗. J Am Coll Cardiol. 2017;69(7):820–2. https://doi.org/10.1016/j.jacc.2016.10.083.
van Kimmenade RRJ, Januzzi JL. Emerging biomarkers in heart failure. Clin Chem. 2012;58(1):127–38. https://doi.org/10.1373/clinchem.2011.165720.
Böhm F, Pernow J. The importance of endothelin-1 for vascular dysfunction in cardiovascular disease. Cardiovasc Res. 2007;76(1):8–18. https://doi.org/10.1016/j.cardiores.2007.06.004.
Bayes-Genis A, Barallat J, de Antonio M, Domingo M, Zamora E, Vila J, et al. Bloodstream amyloid-beta (1-40) peptide, cognition, and outcomes in heart failure. Revista Española de Cardiología (English Edition). 2017;70(11):924–32. https://doi.org/10.1016/j.rec.2017.02.021.
Langenickel TH, Tsubouchi C, Ayalasomayajula S, et al. The effect of LCZ696 (sacubitril/valsartan) on amyloid-β concentrations in cerebrospinal fluid in healthy subjects. Br J Clin Pharmacol. 2016;81:878–90.
Northridge D, Alabaster C, Connell JC, et al. Effects of UK 69 578: a novel atriopeptidase inhibitor. Lancet. 1989;334:591–3.
Packer M, Califf RM, Konstam MA, et al. Comparison of omapatrilat and enalapril in patients with chronic heart failure. The Omapatrilat Versus Enalapril Randomized Trial of Utility in Reducing Events (OVERTURE). 2002;106:920–6.
Kostis JB, Packer M, Black HR, Schmieder R, Henry D, Levy E. Omapatrilat and enalapril in patients with hypertension: the Omapatrilat Cardiovascular Treatment vs. Enalapril (OCTAVE) trial*. Am J Hypertens. 2004;17(2):103–11. https://doi.org/10.1016/j.amjhyper.2003.09.014.
Campbell DJ, Krum H, Esler MD. Losartan increases bradykinin levels in hypertensive humans. Circulation. 2005;111(3):315–20. https://doi.org/10.1161/01.CIR.0000153269.07762.3B.
Claggett B, Packer M, McMurray JJV, et al. Estimating the long-term treatment benefits of sacubitril–valsartan. N Engl J Med. 2015;373:2289–90.
Solomon SD, Zile M, Pieske B, et al. The angiotensin receptor neprilysin inhibitor LCZ696 in heart failure with preserved ejection fraction: a phase 2 double-blind randomised controlled trial. Lancet. 2012;380:1387–95.
Karlström P, Alehagen U, Boman K, Dahlström U, on behalf of the U-sg. Brain natriuretic peptide-guided treatment does not improve morbidity and mortality in extensively treated patients with chronic heart failure: responders to treatment have a significantly better outcome. Eur J Heart Fail. 2011;13(10):1096–103. https://doi.org/10.1093/eurjhf/hfr078.
Januzzi JL, van Kimmenade R, Lainchbury J, et al. NT-proBNP testing for diagnosis and short-term prognosis in acute destabilized heart failure: an international pooled analysis of 1256 patients. The International Collaborative of NT-proBNP Study. Eur Heart J. 2006;27:330–7.
Salah K, Kok WE, Eurlings LW, Bettencourt P, Pimenta JM, Metra M, et al. A novel discharge risk model for patients hospitalised for acute decompensated heart failure incorporating N-terminal pro-B-type natriuretic peptide levels: a European coLlaboration on Acute decompeNsated Heart Failure: ÉLAN-HF Score. Heart. 2014;100(2):115–25. https://doi.org/10.1136/heartjnl-2013-303632.
Masson S, Latini R, Anand IS, et al. Direct comparison of B-type natriuretic peptide (BNP) and amino-terminal proBNP in a large population of patients with chronic and symptomatic heart failure: the Valsartan Heart Failure (Val-HeFT) data. Clin Chem. 2006;52:1528–38.
•• Zile MR, Claggett BL, Prescott MF, McMurray JJV, Packer M, Rouleau JL, et al. Prognostic implications of changes in N-terminal pro-B-type natriuretic peptide in patients with heart failure. Journal of the American College of Cardiology. 2016;68(22):2425–36. This study demonstrated that a reduction in NT-proBNP concentrations during treatment with sacubitril/valsartan was associated with better outcomes. https://doi.org/10.1016/j.jacc.2016.09.931.
Norman JA, Little D, Bolgar M, Di Donato G. Degradation of brain natriuretic peptide by neutral endopeptidase: species specific sites of proteolysis determined by mass spectrometry. Biochem Biophys Res Commun. 1991;175(1):22–30. https://doi.org/10.1016/S0006-291X(05)81194-5.
Januzzi JL. B-type natriuretic peptide testing in the era of neprilysin inhibition: are the winds of change blowing? Clin Chem. 2016;62(5):663–5. https://doi.org/10.1373/clinchem.2016.255455.
Semenov AG, Katrukha AG. Different susceptibility of B-type natriuretic peptide (BNP) and BNP precursor (proBNP) to cleavage by neprilysin: the N-terminal part does matter. Clin Chem. 2016;62(4):617–22. https://doi.org/10.1373/clinchem.2016.254524.
Jaffe AS. Unwinding the interaction of natriuretic peptides and neprilysin∗. J Am Coll Cardiol. 2015;65(7):666–7. https://doi.org/10.1016/j.jacc.2014.12.011.
Ky B, French B, McCloskey K et al. High-sensitivity ST2 for prediction of adverse outcomes in chronic heart failure. Circulation: Heart Failure 2011;4:180–187.
O’Meara E, Prescott MF, Rouleau JL, et al. Association between sST2 levels and cardiovascular outcomes and effect of sacubitril/valsartan on sST2 levels: results from the PARADIGM-HF trial. J Card Fail. 2016;22(8):S29–30. https://doi.org/10.1016/j.cardfail.2016.06.094.
Pascual-Figal DA, Casas T, Ordonez-Llanos J, et al. Highly sensitive troponin T for risk stratification of acutely destabilized heart failure. Am Heart J. 2012;163:1002–10.
Packer M, McMurray JJV, Desai AS, et al. Angiotensin receptor neprilysin inhibition compared with enalapril on the risk of clinical progression in surviving patients with heart failure. Circulation. 2014;
Funding Sources
Dr. Ibrahim is supported in part by the Dennis and Marilyn Barry Fellowship in Cardiology. Dr. Januzzi is supported in part by the Hutter Family Professorship in Cardiology.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
Dr. Januzzi has received grant support from Siemens, Singulex and Prevencio, consulting income from Roche Diagnostics, Critical Diagnostics, Philips, and Novartis, and participates in clinical endpoint committees/data safety monitoring boards for Amgen, Janssen, Abbvie, Pfizer, and Boehringer Ingelheim. Dr. Ibrahim has nothing to disclose.
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Additional information
This article is part of the Topical Collection on Heart Failure
Rights and permissions
About this article
Cite this article
Ibrahim, N.E., Januzzi, J.L. Monitoring Biomarkers in Patients Receiving Neprilysin Inhibitors. Curr Emerg Hosp Med Rep 6, 8–16 (2018). https://doi.org/10.1007/s40138-018-0149-6
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40138-018-0149-6