Abstract
Cervical spondylotic radiculopathy (CSR) is one of the most common degenerative diseases of the spine that is commonly treated with surgery. The primary goal of surgery is to relieve symptoms through decompression or relieving pressure on compressed cervical nerves. Nevertheless, cutaneous pain distribution is not always predictable, making accurate diagnosis challenging and increasing the likelihood of inadequate surgical outcomes. With the widespread application of minimally invasive surgical techniques, the requirement for precise preoperative localization of the affected segments has become critical, especially when treating patients with multi-segmental CSR. Recently, the preoperative use of a selective nerve root block (SNRB) to localize the specific nerve roots involved in CSR has increased. However, few reviews discuss the currently used block approaches, risk factors, and other aspects of concern voiced by surgeons carrying out SNRB. This review summarized the main cervical SNRB approaches currently used clinically and the relevant technical details. Methods that can be used to decrease risk during cervical SNRB procedures, including choice of steroids, vessel avoidance, guidance with radiographs or ultra-sound, contrast agent usage, and other concerns, also are discussed. We concluded that a comprehensive understanding of the current techniques used for cervical SNRB would allow surgeons to perform cervical SNRB more safely.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
This review classified the current approaches used for cervical nerve root blocks for diagnosis or treatment and ways to decrease the risk of complications. |
The approaches involved in the reviewed articles were divided into four categories according to the respective needle trajectory: (1) the anterolateral approach; (2) the lateral approach; (3) the posterolateral approach; and (4) the dorsal approach. Each approach presented specific anatomic considerations for certain conditions and respective risks for injury. |
Specifically, (1) use of small-particle steroids might reduce spinal cord or brain infarction; (2) increased familiarity with the anatomy of regional vessels results in fewer injections into critical arteries; (3) additional procedures such as the use of a lower needle trajectory in the anterolateral approach, the intraoperative lateral view of the cervical spine, and use of contrast agents might help improve the success of cervical nerve root blocks. |
It is beneficial for surgeons to be proficient in multiple techniques, which allows successful management of a range of conditions that arise during selective nerve root blocks. Furthermore, the information provided in this review might allow surgeons to perform cervical nerve root blocks more safely. |
Introduction
Cervical spondylotic radiculopathy (CSR) is a common degenerative disease that is usually treated with a nerve root block or surgery. Especially for minimally invasive cervical surgery, the accurate preoperative assessment of the affected nerve roots is strongly linked with positive postoperative outcomes. It has been reported that approximately 26% of patients tend to report persistent, significant pain during postoperative follow-up assessments [1]. Studies [2, 3] have revealed that atypical cutaneous pain distribution is commonly observed in many cases (24–50% of cases), which, in turn, may decrease the reliability of diagnosis using MRI and neurological examination.
Selective nerve root block (SNRB) is a treatment for CSR to relieve pain in the neck, arms, and shoulders resulting from compressed nerve roots within the cervical spine. Initially, SNRB was primarily used by anesthesiologists or radiologists. Due to its low risk of pharyngeal and esophageal structural damage, SNRB has also been increasingly used as a preoperative diagnostic method to determine affected nerve roots [4]. To our knowledge, few reviews have discussed cervical SNRB. Therefore, this narrative review summarized the brief history and technical details of the various cervical SNRB approaches that are in use currently and factors that could prevent complications, including choice of steroids, vessel avoidance, the guidance method used, use of contrast agents, and other concerns. The author searched PubMed, MEDLINE, and Google Scholar (1950–2021), using the terms “cervical” AND “radiculopathy” AND “nerve root block*”. Related supporting literature from the articles that were retrieved also were examined.
The first cervical spine epidural injection was performed by Dogliotti in 1933 [5]. The block was performed using an intervertebral approach to carry out surgical anesthesia. However, due to the narrow space between the ligamentum flavum and dura (1.5–2 mm in C7) [6], this approach has not been performed routinely for decades. Approximately 20 years later, Robechi and Livre reported using epidural cortisone injections to treat lower back pain [7, 8]. The advent of radiography significantly improved the accuracy and safety of spinal nerve root blocks. Since the 1980s, the clinical application of cervical epidural injections has increased gradually and has been used for CSR, neck pain, cervicogenic headache, and invalid sympathetic block complex regional pain syndromes [9,10,11].
Based on existing literature, we divided the predominant block approaches into an anterolateral approach, lateral approach, posterolateral approach, and dorsal approach. The classification criteria were based on the angle between the penetration trajectory and the horizontal plane of the patient when in the supine position. Specifically, 0° represents a lateral approach, a positive angle represents an anterolateral approach, and a negative angle represents a posterior lateral approach. The dorsal approach is relatively new. This article is based on previously conducted studies and does not contain any new studies with human participants or animals performed by any of the authors.
Anterolateral Approach
In 1988, Morvan et al. [12] described an anterolateral approach that was modified from cervical discography. With this approach, the contrasting bed is placed vertically, and the patient sits on a stool that can rotate, and the head is supported. Using radiography for guidance, the patient's body position is slightly tilted. The symptomatic side is positioned forward to obtain a complete image of the cervical intervertebral foramen, which is achieved using an oblique cervical view. The radiography projecting direction is adjusted so it is parallel to the target segment of the lamina. Then, the trachea, esophagus, carotid artery, and jugular vein are manually pushed medially (Fig. 1), and the needle is gently advanced to the intervertebral foramen under radiographic guidance. Next, the needle tip is placed at the posterior aspect of the intervertebral foramen against the anterior edge of the superior articular process or the posterior aspect of the uncinate process. The author [10] reported a successful completion rate for the procedure of only 14% using this approach. One of the 73 patients reported a headache, which disappeared within several hours. In another patient, who suffered from dizziness, the symptoms disappeared within 24 h. No other serious complications were reported.
Subsequently, the anterolateral approach has been used widely in cervical SNRB. Schellhas et al. [13] reported on 4612 patients that received cervical vertebral SNRB using the anterolateral approach (diagnostic + therapeutic) in 13 years without incidence of cerebral infarction, spinal cord injury, or other severe complications. In one patient, seizures were reported, but the patient recovered within 30 min. The author speculated that this might have been due to injection into an artery. No serious complications have been reported in other studies [14, 15].
Lateral Approach
In 1996, Bush et al. [9] reported a lateral approach for cervical SNRB in 68 patients. The authors discussed two approaches, in addition to the anterolateral approach described by Morvan; the author also used the lateral approach described by Moore et al. and Zenz et al. [16]. Briefly, patients were placed in a supine position. The needle was gently advanced towards the target segment until a bony docking site was attained (such as the C7 transverse process). Then the needle was slowly withdrawn; if no blood or cerebrospinal fluid were observed, 1 ml of 1% lidocaine and 1 ml 40 mg triamcinolone acetonide were injected. Later, Slipman et al. [17] also used the lateral approach SNRB for 20 cases. They obtained a satisfactory rate of block of 60% and reported no complications (Table 1).
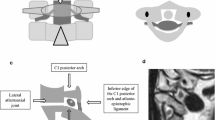
Valle et al. [18] reported several shortcomings associated with the anterolateral approach, including (1) risk of damage to vital structures (trachea, esophagus, jugular artery, and vein), which could be avoided by pushing these structures towards the midline; (2) risk of damage to the vertebral artery, which cannot be avoided by manual manipulation; (3) risk of spinal cord injury caused by deep penetration of the needle tip into the spinal canal, also which cannot be avoided. The author determined that the lateral approach under radiographic guidance was safer when the patient was in a seated position. Performance using this approach will not damage vital structures such as the carotid artery without pushing tissues below the level of C3. However, this approach can provide a safe bony docking site before the needle is finally positioned (Fig. 2A, needle tip against the anterior edge of the superior articular process) [19]. It is noted that the patient’s shoulder will probably be naturally lower when in a seated position, which facilitates manipulation. Thus, a contrast agent is not needed with cervical anteroposterior and oblique radiographs.
However, the lateral approach has been used rarely due to the inconvenient manipulation that is required. In 2013, Miller et al. proposed a modified lateral approach with the shortest penetration trajectory (Fig. 3). With this approach, the patient is placed in a lateral position. Then, under CT guidance, a needle is inserted in a near-vertical orientation. The author reported no severe complications in 116 cases that used this procedure. The author suggested that this modification increased safety and decreased the intraoperative radiation time [20]. This method could expand the clinical applicability of the lateral approach.
Posterolateral Approach
The posterolateral approach was first described by Kumar et al. in 2008. When using this approach, the patient was placed in a lateral position with the affected side up. A pillow was placed beneath the neck to avoid lateral flexion, the neck was slightly stretched, and the patient’s shoulders were pressed down to facilitate obtaining an entire intraoperative cervical radiograph. The C-arm was centered on the target segment, tilted 20°-30°, and the cervical oblique film was used to observe the foramen fully. The entry point was the intersection of the tangent line of the superior edge of the target segment foramen in parallel with the disc plane and the tangent line of the posterior edge of the two lateral masses of the target segment and the adjacent segment. The needle was inserted using imaging guidance until the needle tip was located on the anterolateral side of the posterior edge of the intervertebral foramen. After the needle entered the nerve root canal, it was withdrawn to ensure no blood vessels were penetrated. Subsequently, 0.2–0.5 ml of contrast agent was injected, and the diffusion along the nerve root was confirmed. Then, 1 ml of a long-acting steroid hormone (40 mg triamcinolone acetonide) and 1.0–1.5 ml of a long-acting local anesthetic were injected (0.25% bupivacaine) [21]. The authors observed that 28 of 30 patients exhibited significant improvements in their VAS and NDI scores. No severe complications were reported.
In the study [21], the author described a “safe zone” that was defined as the posterior-superior quadrant of the foramen (Fig. 4). It should not extend beyond the line connecting the midpoints of the articular processes in case of dural penetration [22]. Placement within this area can minimize the possibility of dura or blood vessel damage. Likewise, the SNRB guidelines suggest placing the needle tip on the dorsal side of the foramen, which is based on the assumption that the vertebral artery travels in front of the exiting nerve root [23]. However, Huntoon et al. [24] claimed that even if the needle was placed at the posterior area, there was still a 1% possibility of damage to the anterior spinal artery and suggested that a safe region did not exist.
Dorsal Approaches: Direct and Indirect
Previous studies have reported that severe complications related to cervical SNRB include cerebral edema, cerebral infarction, cervical spinal cord infarction, ventriculomegaly, subarachnoid hemorrhage, and hydrocephalus [25,26,27]. Even though these complications exhibit a relatively low incidence (less than 1%), if they do occur, they could be life-threatening [28]. In 2007, Drape et al. proposed a posterior facet joint block for patients with CSR that presented good outcomes. The injection to facet joints could indirectly affect the nerve root, which might be a safer approach. However, the sample size in this study was small (17 cases).
In 2009, Wolter et al. [29] first reported on the dorsal approach (Fig. 5A). A year later, they used the same approach to perform dorsal cervical SNRB in 53 patients [30]. In this approach, the patient is placed in a prone position. The needle is inserted at an angle of 10–45° to the sagittal plane to avoid cervical vessel penetration. Ideal needle insertion should occur against the lateral edge of the posterior wall of the intervertebral foramen. The facet joint capsule can serve as a guide to positioning the needle. In some cases, the patient may complain of induced analogous radicular pain when the tip of the needle is positioned correctly. When a satisfactory needle tip position is obtained, 0.5 ml of iopamidol and 0.75% bupivacaine (1:1 mixture) are injected. The author suggested that this approach can minimize the risk of accidental damage to the radicular artery (Table 2).
On the other hand, Sutter et al. suggested that the dorsal approach described by Wolter et al. is still a direct penetration technique that utilizes a dorsal approach to reduce the risk of inadvertent injection of a blood vessel. However, the needle tip placement is still within the intervertebral foramen area, which can potentially damage the nearby blood vessels. Sutter et al. established a new approach called an “indirect nerve root block” [31], during which the needle tip trajectory was far removed from the nerve roots and blood vessels to avoid related complications (Fig. 5B). With this approach, the patient is placed in a prone position. After the target segment is localized via a lateral cervical radiograph, the needle is inserted vertically into the dorsal skin, directly on the lateral surface of the facet joint, which is also adjacent to the target intervertebral foramen. After sterilization, the needle is advanced under imaging guidance until it reaches the bone at the facet joint, after which 0.5 ml of contrast agent is injected to verify whether the needle tip insertion is correct.
A contrast agent is used to visualize the distribution of drugs diffused around the dorsal and lateral sides of the facet joints and ventral to the facet joints, as well as the direct contact of the nerve roots. After contrast agent injection, 4 mg (1 ml) of a non-particulate corticosteroid (dexamethasone) and 1 ml of 0.2% bupivacaine are slowly injected. Bensler and Sutter et al. [32] examined a dorsal indirect approach in 112 patients and concluded that this approach is safer than the direct dorsal approach.
Procedures to Decrease Complications
Use Non-particulate Steroids
Cervical SNRB has great value as a non-surgical treatment and a diagnostic method. However, the rare but catastrophic complications are an unavoidable dilemma that surgeons must face. In 2007, Scanlon et al. [33] published a cross-sectional study describing 78 critical neurological complications caused by cervical foramen blocks. In 2013, Hodler et al. [34] reported on two cases of tetraplegia after cervical SNRB. The surgeon in this study had performed more than 1000 cases of SNRB. The procedure was performed under the guidance of CT, and a contrast agent was used before drug injection.
Studies have revealed that critical complications after cervical SNRB, including quadriplegia, cerebral infarction, and cortical blindness, might be related to inadvertent arterial injection of a specific steroid. Some steroids contain large or small particles that may form larger aggregates and, consequently, create emboli. When an inadvertent arterial injection occurs, the spinal cord or brain may be infarcted, depending on which terminal blood vessels are blocked [27, 33, 35]. Scanlon and colleagues revealed a strong association between particulate corticosteroid usage and cerebral and spinal cord infarction [36]. A significant mortality rate (death of 4/4 animals) was observed in a porcine model in which animals received an injection of particulate steroids (methylprednisolone) into the vertebral artery. In contrast, animals injected with non-particulate steroids (dexamethasone, prednisolone) survived without MR or histological abnormality.
Get Familiar with the Vulnerable Vessels’ Anatomy
The position of the carotid sheath is often a primary factor that affects the final choice of approach. When the carotid sheath is lower on a cross section, surgeons tend to choose a smaller angle with the horizontal plane to avoid the carotid sheath (e.g., lateral approach or posterolateral approach). Fitzgerald et al. [37] reported that the smaller the needle angle, the easier the needle tip trajectory intersects the vertebral artery. Therefore, it is essential to confirm the location of the vertebral arteries preoperatively.
Incorrect injection into the radicular artery and not the vertebral artery has been recognized as the cause of observed complications. The radicular artery (Fig. 6) originates from the vertebral artery and the ascending carotid artery. It accompanies the spinal nerve into the nerve root canal and connects with the anterior spinal artery and the medullary vascular network. Surgeons need to have a detailed understanding of the precise local anatomy for cervical SNRB. Still, only a few studies have investigated the precise local anatomy related to cervical SNRB, especially the relevant vascular anatomy. There also is confusion caused by the inconsistent naming of arteries. As mentioned above, the radicular artery has been called the “radicular artery,” or “radiculomedullary artery,” or “spinal branch” in different studies [38]. The artery is considered a high-risk vessel for incorrect injection and the cause of fatal complications during cervical SNRB (Fig. 6). A recent study of the optimal needle insertion angle for cervical SNRB using an anterolateral cervical approach (190 cases) concluded that the optimal needle insertion angle was between 33° and 68°, and the average was slightly less than 50° (Fig. 7). The authors concluded that maintaining the needle along the posterior wall of the foramen could minimize or even avoid accidental injury to the nerve root and vertebral artery. Still, it could not avoid entering the root canal or radicular artery. Thus, the risk of mistaken injection still exists.
Considerable anatomic variation of blood vessels around the cervical intervertebral foramen is another problem. A previous study revealed that unpredictable vascular anatomical variation is one of the main reasons for incorrect intravascular injections [39]. Huntoon et al. [24, 40] reported that critical structures, such as the ascending carotid artery and deep carotid artery pass through the posterior part of the intervertebral foramen, which is the needle tip placement area for conventional cervical SNRB. There are as many as 29% of vertebral artery variants that pass less than 2 mm to the posterior wall of the foramen. Recent research related to the distribution of vulnerable blood vessels that intersect the needle trajectory during cervical SNRB (C3-C7) with ultrasound (104 cases) [41] showed seven blood vessels (8.33%) at the level of C3, 14 blood vessels (13.86%) at the level of C4, 17 blood vessels (16.35%) at the C5 level, 27 blood vessels (25.96%) at the C6 level, and 31 blood vessels (29.81%) at the level of C7. The author reported [42] that 37.5% of 104 patients exhibited at least one vulnerable blood vessel, and approximately 14.89% of the 497 foramina had vulnerable blood vessels in the posterior half of the foramen. The author also reported that the risk of vascular injury in the lower cervical spine was greater than that of the upper region, which indicates the need for additional caution. This conclusion was supported by a recent study [43].
Apart from the localized anatomical considerations, the risk of cervical SNRB is also influenced by pathological changes due to the associated degeneration. A previous study showed that the severity of degenerative foraminal stenosis was positively correlated with the vertebral artery’s position. The osteophytes of the Luschka joint tend to push the vertebral artery towards the posterolateral foramen, and the osteophytes of the facet joints tend to push the posterior edge of the foramen towards the vertebral artery, thus causing the foramen to be covered by the vertebral artery. As a result, the vertebral artery may be extremely close to the conventional needle tip location during cervical SNRB in patients with cervical degeneration. Nearly all patients with CSR who need to receive SNRB may suffer from some degree of intervertebral degeneration. To design the safest needle trajectory and needle placement point, surgeons need to be aware of the possibility of a change in the position of the vertebral artery, especially for patients with Luschka joints or facet joint osteophytes.
Consider Alternative Guidance Way: Ultrasound
Recently, the clinical application of cervical SNRB combined with ultrasound has increased. In 2004, Galiano et al. [44] used cadavers to examine ultrasound-guided transforaminal injections on the lumbar spine. The CT results confirmed that the needle tip could be accurately positioned using ultrasound guidance. In 2009, Narouze et al. reported that ultrasound-guided cervical SNRB was a safe approach that could allow the needle tip to stay within 5 mm of the target, and the risk of blood vessel damage was minimized in the target area. This was later confirmed by Nakagawa et al. [45]. Both authors emphasized the advantage that was provided when the transverse process with nerve roots and vessels could be recognized under ultrasound guidance, which enables safer SNRB. The cadaver study revealed that a solution injected in SNRB under ultrasound spread in the extraforaminal direction, which would not cause an epidural block [46].
Other more recent studies have reported on the safety of cervical SNRB under ultrasound guidance [47,48,49]. Jee et al. [50] compared the clinical outcomes of cervical SNRB under ultrasound and radiographic guidance (120 cases). Five cases of intravascular injection in the radiographic guidance group were reported with no significant difference observed between the groups. Furthermore, Park et al. [51] reported on three cases of intravascular injection in a group that used fluoroscopy guidance in their cohort study. Moreover, a retrospective comparative study reported that ultrasound-guided SNRB might be safer and have similar pain relief compared with SNRB under fluoroscopy (seven cases of intravascular contrast injection were observed) [52]. Wakeling and colleagues [53] reported on the application of cervical SNRB under combined fluoroscopy and ultrasound guidance to avoid vessel damage when using only ultrasound and to avoid mistaken vascular injection when using only fluoroscopy. The author believed that this approach could reduce complications and irradiation exposure [41]. More recently, a retrospective comparative study compared interlaminar SNRB under ultrasound guidance with transforaminal SNRB under fluoroscopy guidance. The authors of this study concluded that SNRB guided by ultrasound had the lowest intravascular injection rate and similar pain relief in the lower cervical spine [54]. Moreover, the logistic regression analysis revealed that the guidance method, gender, age, and pain duration were not independent predictors of the block success [54].
Current evidence has not revealed any difference between cervical SNRB under ultrasound guidance and SNRB under fluoroscopy guidance. Thus, the unique advantage of real-time vascular visualization may help reduce intravascular injection.
Value the Intraoperative Lateral Cervical View
The International Spinal Injection Association and Windsoret et al. [55] recommended that the needle tip position should be confirmed with the aid of cervical anteroposterior and oblique radiographs. Consequently, Ma et al. [56] proposed lateral cervical spine division (Fig. 8). The author emphasized the importance of the lateral cervical view, which has been ignored in cervical SNRB. To avoid vertebral artery damage as much as possible, the authors suggested that the needle tip should be closely attached to the anterior surface of the lateral mass and that position can be determined only with a standard lateral cervical view. Even if the position of the needle tip on the oblique cervical radiograph appears to be appropriate on the lateral radiograph, the needle tip might be placed too anteriorly. According to their study of 1036 cases of cervical injection, the complication rate of injection in zone C was significantly higher than in zones A and B (complication rate of 6.06, 1.51, and 1.81%, respectively).
Use Contrast Agents
Initially [19], it was believed that contrast agents increase the infection risk, prolong the procedure time, and induce radicular pain due to locally increased pressure caused by the contrast agents. However, all subsequent studies have supported the use of contrast agents. First, Bush et al. [9] suggested that the reason for the poor outcomes (satisfaction rate of only 14%) in the study reported by Morvan et al. was because they did not use contrast agents. As a result, they could not be sure that the drugs were injected around the responsible nerve root appropriately or if they were flushed away by cerebrospinal fluid or blood flow. Also, the use of contrast agents can minimize inappropriate artery injections. If no contrast agent is used during surgery, inappropriate vessel injection is primarily determined by the appearance of blood in the syringe after withdrawal. However, it has been reported that the artery may be penetrated even if no blood is observed. When assessing if a vessel was incorrectly injected based on the presence of blood in the withdrawn syringe, the specificity is 97%, and the sensitivity is only 45.9% [57, 58]. Moreover, the contrast agent used in SNRB has been proved to be safe. A prospective study (504 cases of cervical SNRB) reported that in approximately 19% of the cases, contrast agents were observed to be injected into the bloodstream, but no severe complications were reported [57]. Also, the application of a contrast agent did not influence the effect of the nerve root block [59].
In conclusion, even if the needle is ideally placed and no blood is observed when the syringe is withdrawn, the possibility of inappropriate artery injection cannot be excluded. Furthermore, fast clearance of the contrast agent indicates the possibility of a mistaken artery injection, while slow clearance indicates the possibility of a mistaken vein injection. Rathmell et al. [60] reported on a case where, after injection of a contrast agent, a curved filament was observed medial to the nerve root. With digital subtraction angiography, a clear outline of the radicular artery could be identified. The author stopped further manipulation immediately, and the patient showed no adverse reaction. Therefore, the application of a contrast agent in cervical SNRB probably is advantageous.
Some Other Controversial Points
-
a)
Although multiple “direct approach” studies have reported that homologous radicular pain reappearance sometimes occurs when the needle is placed in the correct position, this “reappearance” does not influence the block effect [59].
-
b)
There has been controversy related to blunt needle application in SNRB, and no evidence has proven that use of a blunt needle provides better safety [24, 61].
-
c)
Ryan et al. [62] claimed that CT guidance might not be sufficient to identify the intravascular injection due to the following reasons: (1) once injected, the contrast agents are washed away before the CT is performed; (2) the vessels enter the cord at a different level, and therefore, are not properly imaged.
-
d)
It has been proposed that a narrow intervertebral foramen might impact the effectiveness of SNRB. A recent retrospective study [63] reported an unexpected conclusion that patients with severe foramen narrowness respond well to SNRB. On the other hand, the response of a patient to SNRB with mild to moderate foramen narrowness is unpredictable.
-
e)
The medical compounds used with SNRB vary among surgeons. In general, local anesthetics are required for diagnostic purposes, and non-steroidal hormones are required for therapeutic procedures. In clinical reports, various drug mixtures have been used for cervical SNRB. The common compound is a steroid and lidocaine mixture 1.0–1.5 ml (mixing ratio 1:1–1:1.5) [24, 61]. Based on a study spanning 13 years and involving 4612 patients, it has been recommended to use a 1.0–1.6 ml mixture (steroid and lidocaine of 1: 2–1: 3) for the initial diagnostic block and a 1.3–1.6 ml mixture (steroid and lidocaine of 1: 1 or 1: 2) for repeated blocks (therapeutic protocols) [13].
-
f)
Does drug volume affect the spread of the solution? According to a prospective study [64], there was no intraforaminal epidural spread in patients given a 1 ml injection, but it did occur in 13 cases (24.5%) treated with a 4-ml injection. While the spreading pattern did not seem to influence pain relief, it still needs to be investigated in studies with larger sample sizes.
Conclusions
SNRB has been an effective non-surgical treatment for the neurological symptoms caused by CSR. Currently, the widespread, minimally invasive surgery emphasizes SNRB due to the requirement for accurate preoperative localization of the affected nerve roots. This requires the surgeons performing cervical SNRB to be familiar with the various possible approaches and the risk factors for complications to be able to perform cervical SNRB more safely. In summary, based on the studies discussed in this review, a good understanding of the relative location of the carotid and vertebral arteries during preoperative MR are essential.
-
a)
When the carotid sheath is positioned anteriorly, the anterolateral approach is recommended. In this instance, the risk of vertebral artery damage is the lowest.
-
b)
When the carotid sheath is positioned posteriorly during the lateral or posterolateral approach, the needle trajectory is likely to intersect the vertebral artery, especially in cases with Luschka joints or facet joint osteophytes. In this instance, it is suggested to estimate the relative location of the vertebral artery to avoid accidental injury.
-
c)
When the carotid sheath is positioned posteriorly and accompanied by a deviation of the vertebral artery or the posterior wall of the intervertebral foramen, which increases the risk associated with the lateral and posterolateral approaches, the dorsal approach may be a better choice. Thus, it is beneficial for surgeons to be proficient in multiple techniques to manage various conditions that could be encountered.
Future Perspectives
As a beneficial method for CSR therapy and responsible segment localization, SNRB has been adopted increasingly in recent years. Based on this narrative review, there are still some controversial questions in the SNRB procedure. For instance, (1) if the steroid types, anesthetic types, and their mixture ratio will affect the outcome? (2) if it exists a preferable approach for more or even every condition? (3) if there is a better option in US, X-ray and CT guidance? This review was hoping to encourage more research with high quality and a larger sample size focusing on the issue in the future. Many technical details within the procedure need further investigation in order to establish eventually a safe and effective standard operating procedure.
References
Sampath P, Bendebba M, Davis JD, Ducker T. Outcome in patients with cervical radiculopathy. Prospective, multicenter study with independent clinical review. Spine (Phila Pa 1976). 1999;24(6):591–7.
Siivola SM, Levoska S, Tervonen O, Ilkko E, Vanharanta H, Keinanen-Kiukaanniemi S. MRI changes of cervical spine in asymptomatic and symptomatic young adults. Eur Spine J. 2002;11(4):358–63.
Anderberg L, Annertz M, Rydholm U, Brandt L, Saveland H. Selective diagnostic nerve root block for the evaluation of radicular pain in the multilevel degenerated cervical spine. Eur Spine J. 2006;15(6):794–801.
Lee SH, Kim KT, Jeong BO, et al. The safety zone of percutaneous cervical approach: a dynamic computed tomographic study. Spine (Phila Pa 1976). 2007;32(20):E569–74.
Dogliotti AM. Segmental peridural spinal anesthesia. Am J Surg. 1933;20(1):107–18.
Han KR, Kim C, Park SK, Kim JS. Distance to the adult cervical epidural space. Reg Anesth Pain Med. 2003;28(2):95–7.
Robecchi A, Capra R. Hydrocortisone (compound F); first clinical experiments in the field of rheumatology. Minerva Med. 1952;43(98):1259–63.
Lievre JA, Bloch-Michel H, Attali P. Trans-sacral injection; clinical and radiological study. Bull Mem Soc Med Hop Paris. 1957;73(33–34):1110–8.
Bush K, Hillier S. Outcome of cervical radiculopathy treated with periradicular/epidural corticosteroid injections: a prospective study with independent clinical review. Eur Spine J. 1996;5(5):319–25.
Castagnera L, Maurette P, Pointillart V, Vital JM, Erny P, Senegas J. Long-term results of cervical epidural steroid injection with and without morphine in chronic cervical radicular pain. Pain. 1994;58(2):239–43.
Cicala RS, Jones JW, Westbrook LL. Causalgic pain responding to epidural but not to sympathetic nerve blockade. Anesth Analg. 1990;70(2):218–9.
Morvan G, Monpoint D, Bard M, Levi-Valensin G. Direct intra-foraminal injection of corticosteroids in the treatment of cervico-brachial pain. In: Interventional radiology in bone and joint. Berlin: Springer; 1988. p. 253–7.
Schellhas KP, Pollei SR, Johnson BA, Golden MJ, Eklund JA, Pobiel RS. Selective cervical nerve root blockade: experience with a safe and reliable technique using an anterolateral approach for needle placement. AJNR Am J Neuroradiol. 2007;28(10):1909–14.
Desai A, Saha S, Sharma N, Huckerby L, Houghton R. The short- and medium-term effectiveness of CT-guided selective cervical nerve root injection for pain and disability. Skelet Radiol. 2014;43(7):973–8.
Ito K, Yukawa Y, Machino M, et al. Treatment outcomes of intradiscal steroid injection/selective nerve root block for 161 patients with cervical radiculopathy. Nagoya J Med Sci. 2015;77(1–2):213–9.
Moore DC. Regional block: a handbook for use in the clinical practice of medicine and surgery. Charles C Thomas Pub Limited;1965.
Slipman CW, Lipetz JS, Jackson HB, Rogers DP, Vresilovic EJ. Therapeutic selective nerve root block in the nonsurgical treatment of atraumatic cervical spondylotic radicular pain: a retrospective analysis with independent clinical review. Arch Phys Med Rehabil. 2000;81(6):741–6.
Vallée JN, Feydy A, Carlier RY, Mutschler C, Mompoint D, Vallée CA. Chronic cervical radiculopathy: lateral-approach periradicular corticosteroid injection. Radiology. 2001;218(3):886–92.
Vallee JN, Feydy A, Carlier RY, Mutschler C, Mompoint D, Vallee CA. Chronic cervical radiculopathy: lateral-approach periradicular corticosteroid injection. Radiology. 2001;218(3):886–92.
Miller TS, Fruauff K, Farinhas J, et al. Lateral decubitus positioning for cervical nerve root block using CT image guidance minimizes effective radiation dose and procedural time. AJNR Am J Neuroradiol. 2013;34(1):23–8.
Kumar N, Gowda V. Cervical foraminal selective nerve root block: a “two-needle technique” with results. Eur Spine J. 2008;17(4):576–84.
Rathmell JP, Aprill C, Bogduk N, Weiskopf RB. Cervical transforaminal injection of steroids. J Am Soc Anesthesiol. 2004;100(6):1595–600.
Baker R, Dreyfuss P, Mercer S, Bogduk N. Cervical transforaminal injection of corticosteroids into a radicular artery: a possible mechanism for spinal cord injury. Pain. 2003;103(1–2):211–5.
Huntoon MA. Anatomy of the cervical intervertebral foramina: vulnerable arteries and ischemic neurologic injuries after transforaminal epidural injections. Pain. 2005;117(1–2):104–11.
Muro K, O’Shaughnessy B, Ganju A. Infarction of the cervical spinal cord following multilevel transforaminal epidural steroid injection: case report and review of the literature. J Spinal Cord Med. 2007;30(4):385–8.
Beckman WA, Mendez RJ, Paine GF, Mazzilli MA. Cerebellar herniation after cervical transforaminal epidural injection. Reg Anesth Pain Med. 2006;31(3):282–5.
Wallace MA, Fukui MB, Williams RL, Ku A, Baghai P. Complications of cervical selective nerve root blocks performed with fluoroscopic guidance. AJR Am J Roentgenol. 2007;188(5):1218–21.
Carragee EJ, Hurwitz EL, Cheng I, et al. Treatment of neck pain: injections and surgical interventions: results of the Bone and Joint Decade 2000–2010 Task Force on Neck Pain and Its Associated Disorders. Spine (Phila Pa 1976). 2008;33(4 Suppl):S153–69.
Wolter T, Mohadjer M, Berlis A, Knoeller S. Cervical CT-guided, selective nerve root blocks: improved safety by dorsal approach. AJNR Am J Neuroradiol. 2009;30(2):336–7.
Wolter T, Knoeller S, Berlis A, Hader C. CT-guided cervical selective nerve root block with a dorsal approach. AJNR Am J Neuroradiol. 2010;31(10):1831–6.
Sutter R, Pfirrmann CW, Zanetti M, Hodler J, Peterson CK. CT-guided cervical nerve root injections: comparing the immediate post-injection anesthetic-related effects of the transforaminal injection with a new indirect technique. Skelet Radiol. 2011;40(12):1603–8.
Bensler S, Sutter R, Pfirrmann CW, Peterson CK. Long term outcomes from CT-guided indirect cervical nerve root blocks and their relationship to the MRI findings–a prospective Study. Eur Radiol. 2015;25(11):3405–13.
Scanlon GC, Moeller-Bertram T, Romanowsky SM, Wallace MS. Cervical transforaminal epidural steroid injections: more dangerous than we think? Spine (Phila Pa 1976). 2007;32(11):1249–56.
Hodler J, Boos N, Schubert M. Must we discontinue selective cervical nerve root blocks? Report of two cases and review of the literature. Eur Spine J. 2013;22(Suppl 3):S466–70.
Dreyfuss P, Baker R, Bogduk N. Comparative effectiveness of cervical transforaminal injections with particulate and nonparticulate corticosteroid preparations for cervical radicular pain. Pain Med. 2006;7(3):237–42.
Benedetti EM, Siriwetchadarak R, Stanec J, Rosenquist RW. Epidural steroid injections: complications and management. Tech Reg Anesth Pain Manag. 2009;13(4):236–50.
Fitzgerald RT, Bartynski WS, Collins HR. Vertebral artery position in the setting of cervical degenerative disease: implications for selective cervical transforaminal epidural injections. Interv Neuroradiol. 2013;19(4):425–31.
Amato ACM, Stolf NAG. Anatomy of spinal blood supply. J Vasc Bras. 2015;14(3):248–52.
Suzuki A, Daubs MD, Inoue H, et al. Prevalence and motion characteristics of degenerative cervical spondylolisthesis in the symptomatic adult. Spine (Phila Pa 1976). 2013;38(17):E1115–20.
Huston CW. Cervical epidural steroid injections in the management of cervical radiculitis: interlaminar versus transforaminal. A review. Curr Rev Musculoskelet Med. 2009;2(1):30–42.
Lee HH, Park D, Oh Y, Ryu JS. Ultrasonography evaluation of vulnerable vessels around cervical nerve roots during selective cervical nerve root block. Ann Rehabil Med. 2017;41(1):66–71.
Park D, Ryu JS. Distribution patterns of vasculature around cervical nerve roots. Pm r. 2019;11(8):815–20.
Murata S, Iwasaki H, Natsumi Y, Minagawa H, Yamada H. Vascular evaluation around the cervical nerve roots during ultrasound-guided cervical nerve root block. Spine Surg Relat Res. 2020;4(1):18–22.
Galiano K, Obwegeser AA, Bodner G, et al. Real-time sonographic imaging for periradicular injections in the lumbar spine: a sonographic anatomic study of a new technique. J Ultrasound Med. 2005;24(1):33–8.
Nakagawa M, Shinbori H, Ohseto K. Ultrasound-guided and fluoroscopy-assisted selective cervical nerve root blocks. Masui. 2009;58(12):1506–11.
Yamauchi M, Suzuki D, Niiya T, et al. Ultrasound-guided cervical nerve root block: spread of solution and clinical effect. Pain Med. 2011;12(8):1190–5.
Wu J, Xu Y, Pu S, et al. US-guided transforaminal cervical nerve root block: a novel lateral in-plane approach. Pain Med. 2021;22(9):1940–5.
Takeuchi M, Kamiya M, Wakao N, et al. A simple, 10-minute procedure for transforaminal injection under ultrasonic guidance to effect cervical selective nerve root block. Neurol Med Chir. 2014;54(9):746–51.
Choi DH, Jung HG, Lee JH, Park JH, Choi YS. Effectiveness of doppler image of the vertebral artery as an anatomical landmark for identification of ultrasound-guided target level in cervical spine. Asian Spine J. 2015;9(5):683–8.
Jee H, Lee JH, Kim J, Park KD, Lee WY, Park Y. Ultrasound-guided selective nerve root block versus fluoroscopy-guided transforaminal block for the treatment of radicular pain in the lower cervical spine: a randomized, blinded, controlled study. Skelet Radiol. 2013;42(1):69–78.
Park Y, Ahn JK, Sohn Y, et al. Treatment effects of ultrasound guide selective nerve root block for lower cervical radicular pain: a retrospective study of 1-year follow-up. Ann Rehabil Med. 2013;37(5):658–67.
Park KD, Lee WY, Nam SH, Kim M, Park Y. Ultrasound-guided selective nerve root block versus fluoroscopy-guided interlaminar epidural block for the treatment of radicular pain in the lower cervical spine: a retrospective comparative study. J Ultrasound. 2019;22(2):167–77.
Wakeling C, Bateman A, Hatrick A, Chatakondu S. Combined fluoroscopic and ultrasound guided cervical nerve root injections. Int Orthop. 2016;40(12):2547–51.
Jang JH, Lee WY, Kim JW, Cho KR, Nam SH, Park Y. Ultrasound-guided selective nerve root block versus fluoroscopy-guided interlaminar epidural block versus fluoroscopy-guided transforaminal epidural block for the treatment of radicular pain in the lower cervical spine: a retrospective comparative study. Pain Res Manag. 2020;2020:9103421.
Windsor RE, Storm S, Sugar R, Nagula D. Cervical transforaminal injection: review of the literature, complications, and a suggested technique. Pain Physician. 2003;6(4):457–65.
Ma DJ, Gilula LA, Riew KD. Complications of fluoroscopically guided extraforaminal cervical nerve blocks. An analysis of 1036 injections. J Bone Jt Surg Am. 2005;87(5):1025–30.
Furman MB, Giovanniello MT, O’Brien EM. Incidence of intravascular penetration in transforaminal cervical epidural steroid injections. Spine (Phila Pa 1976). 2003;28(1):21–5.
Verrills P, Nowesenitz G, Barnard A. Penetration of a cervical radicular artery during a transforaminal epidural injection. Pain Med. 2010;11(2):229–31.
Mallinson PI, Tapping CR, Bartlett R, Maliakal P. Factors that affect the efficacy of fluoroscopically guided selective spinal nerve root block in the treatment of radicular pain: a prospective cohort study. Can Assoc Radiol J. 2013;64(4):370–5.
Rathmell JP, Aprill C, Bogduk N. Cervical transforaminal injection of steroids. Anesthesiology. 2004;100(6):1595–600.
Heavner JE, Racz GB, Jenigiri B, Lehman T, Day MR. Sharp versus blunt needle: a comparative study of penetration of internal structures and bleeding in dogs. Pain Pract. 2003;3(3):226–31.
Ryan TM, Kavanagh EC, MacMahon PJ. Is there a need for contrast administration prior to CT-guided cervical nerve root block? AJNR Am J Neuroradiol. 2013;34(4):E45.
Ray WZ, Akbari S, Shah LM, Bisson E. Correlation of foraminal area and response to cervical nerve root injections. Cureus. 2015;7(7):e286.
Kang S, Yang SN, Kim SH, Byun CW, Yoon JS. Ultrasound-guided cervical nerve root block: does volume affect the spreading pattern? Pain Med. 2016;17(11):1978–84.
Cyteval C, Thomas E, Decoux E, et al. Cervical radiculopathy: open study on percutaneous periradicular foraminal steroid infiltration performed under CT control in 30 patients. AJNR Am J Neuroradiol. 2004;25(3):441–5.
Wagner AL. CT fluoroscopic-guided cervical nerve root blocks. AJNR Am J Neuroradiol. 2005;26(1):43–4.
Chen B, Rispoli L, Stitik TP, Foye PM, Georgy JS. Optimal needle entry angle for cervical transforaminal epidural injections. Pain Physician. 2014;17(2):139–44.
Acknowledgements
We deeply appreciate Mrs. Gao Yuan for helping us with all the schematic drawings in this review. All named authors meet the International Committee of Medical Journal Editors (ICMJE) criteria for authorship for this article, take responsibility for the integrity of the work as a whole, and have given their approval for this version to be published
Funding
The journal’s Rapid Service Fee was funded by the authors.
Authorship
All named authors meet the International Committee of Medical Journal Editors (ICMJE) criteria for authorship for this article, take responsibility for the integrity of the work as a whole, and have given their approval for this version to be published.
Author Contributions
Conceptualization: [Weibing Xu]; Methodology: [Dongfang Yang]; Formal analysis and investigation: [Yutong Hu, Lichen Xu]; Writing—original draft preparation: [Yutong Hu, Lichen Xu]; Writing—review and editing: [Dongfang Yang]; Resources: [Weibing Xu]; Supervision: [Weibing Xu].
Disclosures
Dongfang Yang, Lichen Xu, Yutong Hu, and Weibing Xu have nothing to disclose.
Compliance with Ethics Guidelines
This article is based on previously conducted studies and does not contain any new studies with human participants or animals performed by any of the authors.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License, which permits any non-commercial use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc/4.0/.
About this article
Cite this article
Yang, D., Xu, L., Hu, Y. et al. Diagnosis and Treatment of Cervical Spondylotic Radiculopathy Using Selective Nerve Root Block (SNRB): Where are We Now?. Pain Ther 11, 341–357 (2022). https://doi.org/10.1007/s40122-022-00357-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40122-022-00357-1