Abstract
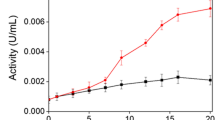
Prompted by multiple applications of laccases, the fungi yielding enzyme with wider geographical applicability and other novel features was explored from Jaisalmer desert (India). One of the isolates TTF6 yielded laccase exhibiting novel many-fold amplification in activity in the presence of lead (Pb) and tolerance to Na. The peroxide-free enzyme showed apparent molecular mass 40 kDa, activity from 10 to 80 °C, temperature optima at 60 °C and pH optima at 5.0. It was highly thermostable losing only 22 and 44% of its maximal activity in 24 and 48 h, respectively, with a half-life of 60 h at 60 °C. It was found resistant to studied metals including Cu and Zn and showed tolerance to many organic solvents. The enzyme was produced best in Olga medium and induced by ethanol and copper sulfate but not by guaiacol, though guaiacol could be utilized as substrate. The partially purified enzyme decolorized selected synthetic dyes at 20–40 °C, the decolorization rate, however, was not affected by Pb. The fungus was identified on the basis of morphological and ITS sequence characteristics as A. nidulans. This is the first report of a Pb-activated laccase and the sixth phenol oxidase from A. nidulans reported so far.




Similar content being viewed by others
References
Hildén K, Hakala TK, Lundell T (2009) Thermotolerant and thermostable laccases. Biotechnol Lett 31:1117–1128
Claus H, Strong PJ (2010) Laccase. In: Flickinger MC (ed) Encyclopedia of industrial biotechnology: bioprocess, bioseparation, and cell technology. Wiley, Hoboken, pp 1–19
Baldrian P (2006) Fungal laccases-occurrence and properties. FEMS Microbiol Rev 30:215–242
Baldrian P, Gabriel L, Nerud F (1996) Effect of cadmium on the ligninolytic activity of Stereum hirsutum and Phanerochaete chrysosporium. Folia Microbiol 41:363–367
Liu H, Du ChengY, B, Tong C, Liang S, Han S, Zheng S, Lin Y (2015) Overexpression of a novel thermostable and chloride-tolerant laccase from Thermus thermophilus SG 0.5JP17-16 in Pichia pastoris and its application in synthetic dye decolorization. PLoSONE 10:e0119833
Osma JF, Toca-Herrera J, Rodríguez-Couto S (2010) Uses of laccases in the food industry. Enzym Res 2010:1–8
Sahay S, Hamid B, Singh P, Ranjan K, Chouhan D, Rana RS, Chaurse VK (2013) Evaluation of pectinolytic activities for oenological uses from psychrotrophic yeasts. Lett Appl Microbiol 57:115–121
Arst HN Jr, Scazzocchio C (1985) Formal genetics and molecular biology of the control of gene expression in Aspergillus nidulans. In: Bennet JW, Lasure LL (eds) Gene manipulations in fungi. Academic Press, New York, pp 309–343
Sahay S (1999) Phenylalanine transport in Aspergillus nidulans: demonstration of role of phenylalanine binding proteins. Indian J Exp Biol 37:152–156
Osmani SA, Hynes M, Oakley BR (2008) Advances in gene manipulations using Aspergillus nidulans. In: Goldman GH, Osmani SA (eds) The Aspergilli: genomics, medical aspects, biotechnology, and research methods. Taylor & Francis, New York, pp 93–512
Kiiskinen LL, Saloheimo M (2004) Molecular cloning and expression in Saccharomyces cerevisiae of a laccase gene from the Ascomycete Melanocarpus albomyces. Appl Environ Microbiol 70:137–144
Coll PM, Abalos JMF, Villanueva JR, Santamaria R, Perez P (1993) Purification and characterization Phenoloxidase (Laccase) from the Lignin-Degrading Basidiomycete PM1 (CECT 2971). Appl Environ Microbiol 59:2607–2613
Slomczynski D, Nakas JP, Tanenbaum SW (1995) Production and characterization of Laccase from Botrytis cinerea 61-34. Appl Environ Microbiol 61:907–912
Munoz C, Guillen F, Martinez AT, Martinez MJ (1997) Laccase isoenzymes of Pleurotus eryngii: characterization, catalytic properties, and participation in activation of molecular oxygen and Mn2+ oxidation. Appl Environ Microbiol 63:2166–2174
Olga VK, Elena VS, Valeria PG, Olga VM, Natalia VL, Aida ND, Alexander IJ, Alexander M (1998) Purification and characterization of the constitutive form of laccase from basidiomycete Coriolus hirsutus and effect of inducers on laccase synthesis. Biotechnol Appl Biochem 28:47–54
Laemmli UK (1970) Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 227:680–685
Machado KMG, Matheus DR (2006) Potential of a ligninolytic enzymatic complex produced by Pleurotus ostreatus during growth on solid substrate for the biodegradation of organic pollutants. Braz J Microbiol 37:468–473
D’Souza-Ticlo D, Sharma D, Raghukumar C (2009) A thermostable metal-tolerant laccase with bioremediation potential from a marine-derived fungus D. Mar Biotechnol 11:725–737
Lowry OM, Rosebrough NJ, Farr A, Randall RJ (1951) Protein measurement with Folin Phenol reagent. J Biol Chem 193:265–270
Sahay S (1999) The use of psyllium as alternative gelling agent for microbial culture media. World J Microbiol Biotechnol 15:733–735
Alcalde M, Bulter T, Arnold FH (2002) Colorimetric assays for biodegradation of polycyclic aromatic hydrocarbons by fungal laccases. J Biomol Screen 7:547–553
Esteve-Zarzoso B, Belloch C, Uruburu F, Querol A (2009) Identification of yeasts by RFLP analysis of the 5.8 s rRNA gene and the two ribosomal internal transcribed spacers. Int J Syst Bacteriol 49:329–335
Tamura K, Stecher G, Peterson D, Filipski A, Kumar S (2013) Mega 6: molecular evolutionary genetics analysis version 6.0. Mol Biol Evol 30:2725–2729
Viswanath B, Chandra MS, Pallavi H, Reddy BR (2008) Screening and assessment of laccase producing fungi isolated from different environmental samples. Afr J Biotechnol 7:1129–1133
Galhaup C, Goller S, Peterbauer CK, Strauss J, Haltrich D (2002) Characterization of the major laccase isoenzyme from Trametes pubescens and regulation of its synthesis by metal ions. Microbiology 148:2159–2169
Chen S, Ma D, Ge W, Buswell JA (2006) Induction of laccase activity in the edible straw mushroom, Volvariella volvace. FEMS Microbiol Lett 218:143–148
Alves AMCR, Record E, Lomascolo A, Scholtmeijer K, Asther M, Wessels JGH (2004) Highly efficient production of laccase by the basidiomycete Pycnoporus cinnabarinus. Appl Environ Microbiol 70:116379–116384
Scherer M, Fischer R (2001) Molecular characterization of a blue-copper laccase, TILA, of Aspergillus nidulans. FEMS Microbiol Lett 199:207–213
Kurtz MB, Champe SP (1982) Purification and characterization of the conidial laccase of Aspergillus nidulans. J Bact 151:1338–1345
Wang ZX, Cai YJ, Liao XR, Tao GJ, Li YY, Zhang F (2010) Purification and characterization of two thermostable laccases with high cold adapted characteristics from Pycnoporus sp. SYBC-L1. Proc Biochem 45:1720–1729
Lončar N, Božić N, Vujči Z (2016) Expression and characterization of thermostable organic-solvent-tolerant laccase from Bacillus licheniformis ATCC9945a. J Mol Catal B 134:390–395
Stoilova I, Krastanov A, Stanchev V (2010) Properties of crude laccase from Trametes versicolor produced by solid-substrate fermentation. Adv Biosci Biotechnol 1:208–215
Kittl R, Mueangtoom K, Gonaus C, Khazaneh ST, Sygmund C, Haltrich D (2012) A chloride tolerant laccase from the plant pathogen ascomycete Botrytis aclada expressed at high levels in Pichia pastoris. J Biotechnol 157:304–314
Mate DM, Alcalde M (2016) Laccase: a multi-purpose biocatalyst at the forefront of biotechnology. Microbial Biotechnol 10:1457–1467
Sahay S (2018) Wine enzymes: potential and practices. In: Kuddus M (ed) Enzymes in food biotechnology: Production, application and future prospects. Academic press, New York, pp 73–92
Piontek K, Antorini M, Choinowski T (2002) Crystal structure of a laccase from the fungus Trametes versicolor at 1.90-A resolution containing a full complement of coppers. J Biol Chem 277:37663–37669
Sahay S, Chouhan D (2018) Study on the potential of cold-active lipases from psychrotrophic fungi for detergent formulation. J Genet Biotechnol. https://doi.org/10.1016/j.geb.2018.04.006
Acknowledgements
A fruitful discussion during screening of laccase producing fungus with Dr. Harald Claus, Institut fúr Mikrobiologie und Weinforschung, Johannes Gutenberg-Universität Mainz, Mainz, Germany, is gratefully acknowledged herewith. The work was supported by Madhya Pradesh Council of Science and Technology (Grant Number 3902/CST/R&D/2011) to the corresponding author.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that there is no conflict of interest to publish this manuscript.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Significance Statement
TTF6 laccases with tolerance to various inhibitory conditions, residual activity at wider temperature range and novel Pb activation for smaller substrates (ABTS) not larger substrates (dyes) are opening new vistas in laccases’ science and applications.
Electronic Supplementary Material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Sahay, S., Chaurse, V. & Chauhan, D. Laccase from Aspergillus nidulans TTF6 Showing Pb Activation for Smaller Substrates and Dyes Remediation in All Climates. Proc. Natl. Acad. Sci., India, Sect. B Biol. Sci. 90, 143–150 (2020). https://doi.org/10.1007/s40011-019-01092-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40011-019-01092-y