Abstract
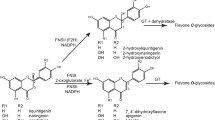
Flavonoids are ubiquitous secondary metabolites in plants and possess a wide range of biological activities such as anti-oxidant, anti-microbial, and anti-cancer activities. As a food ingredient, flavonoids are also thought to have health-promoting and disease-preventing properties. Structural modifications of flavonoids can improve their biological activities. Methylation is a common flavonoid modification that is catalyzed by O-methyltransferases (OMTs). We isolated four putative OMTs from the miniature tomato cultivar Micro-Tom. One OMT (SlOMT3) was successfully expressed in E. coli and purified. Recombinant SlOMT3 was found to be a regiospecific flavonoid 3′/5′-O-methyltransferase. Biochemical parameters show that SlOMT3 has higher binding affinity and catalytic efficiency for quercetin and luteolin than for eriodictyol. This suggests that flavonols and flavones are preferable substrates for SlOMT3. Biotransformation is considered a promising method to modify flavonoid structures. Transgenic E. coli expressing SlOMT3 was used to modify flavonoid substrates and was found to efficiently convert quercetin, luteolin, and eriodictyol to isorhamnetin, chrysoeriol and homoeriodictyol, respectively.
Similar content being viewed by others
References
Aida Y, Tamogami S, Kodama O, and Tsukiboshi T (1996) Synthesis of 7-methoxyapigeninidin and its fungicidal activity against Gloeocercospora sorghi. Biosci Biotech Biochem 60, 1495–1496.
Allister EM, Borradaile NM, Edwards JY, and Huff MW (2005) Inhibition of microsomal triglyceride transfer protein expression and apolipoprotein B100 secretion by the citrus flavonoid naringenin and by insulin involves activation of the mitogen-activated protein kinase pathway in hepatocytes. Diabetes 54, 1676–1683.
Amor IL, Hehn A, Guedone E, Ghedira K, Engasser JM, Chekir-Ghedrira L et al. (2010) Biotransformation of naringenin to eriodictyol by Saccaromyces cerevisiea functionally expressing flavonoid 3 hydroxylase. Nat Prod Commun 5, 1893–1898.
Aoki K, Yano K, Suzuki A, Kawamura S, Sakurai N, Suda K et al. (2010) Large-scale analysis of full-length cDNAs from the tomato (Solanum lycopersicum) cultivar Micro-Tom, a reference system for the Solanaceae genomics. BMC Genomics 11, 210.
Azerad R (2001) Chemical biotechnology: Better enzymes for green chemistry. Curr Opin Biotechnol 12, 533–534.
Buer CS, Imin N, and Djordjevic MA (2010) Flavonoids: new roles for old molecules. J Integr Plant Biol 52, 98–111.
Cacace S, Schröder G, Wehinger E, Strack D, Schmidt J, and Schröder J (2003) A flavonol O-methyltransferase from Cathranthus roseus performing two sequential methylations. Phytochemistry 62, 127–137.
Doostdar H, Burke MD, and Mayer RT (2000) Bioflavonoids: selective substrates and inhibitors for cytochrome P450 CYP1A and CYP1B1. Toxicology 144, 31–38.
Fowler ZL and Koffas MAG (2009) Biosynthesis and biotechnological production of flavanones: current state and perspectives. Appl Microbiol Biotechnol 83, 799–808.
Garo E, Maillard M, Antus S, Mavi S, and Hostettmann K (1996) Five flavans from Mariscus psilostachys. Phytochemistry 43, 1265–1269.
Greenwald P (2004) Clinical trials in cancer prevention: current results and prospectives for the future. J Nutr 134, 3507S–3512S.
Hou DX, Fujii M, Terahara N, and Yoshimoto M (2004) Molecular mechanisms behind the chemopreventive effects of anthocyanidins. J Biomed Biotechnol 2004, 321–325.
Ibrahim RK, Bruneau A, and Bantignies B (1998) Plant O-methyltransferases: molecular analysis, common signature and classification. Plant Mol Biol 36, 1–10.
Ibrahim RK, De Luka V, Khouri H, Latchinian L, Brisson L, and Charest PM (1987) Enzymology and compartmentation of polymethylated flavonol glucosides in Chrysosplenium americanum. Phytochemistry 26, 1237–1245.
Kim BG, Lee Y, Hur HG, Lim Y, and Ahn JH (2006) Flavonoid 3-O-methyltransferase from rice: cDNA cloning, characterization and functional expression. Phytochemistry 67, 387–394.
Kim DH, Kim BG, Lee Y, Ryu JY, Lim Y, Hur HG et al. (2005) Regiospecific methylation of naringenin to ponciretin by soybean O-methyltransferase expressed in Escherichia coli. J Biotechnol 119, 155–162.
Lee J, Jung E, Lee J, Kim S, Huh S, Kim Y et al. (2009) Isorhamnetin represses adipogenesis in 3T3-L1 cells. Obesity 17, 226–232.
Lee YJ, Kim BG, Chong Y, Lim Y, and Ahn JH (2008) Cation dependent O-methyltransferases from rice. Planta 227, 641–647.
Ma G, Yang C, Qu Y, Wei H, Zhang T, and Zhang N (2007) The flavonoid component isorhamnetin in vitro inhibits proliferation and induces apoptosis in Eca-109 cells. Chem-Biol Interact 167, 153–160.
Muzac I, Wang J, Anzellotti D, Zhang H, and Ibrahim RK (2000) Functional expression of an Arabidopsis cDNA clone encoding a flavonol 3-O-methyltransferase and characterization of the gen product. Arch Biochem Biophys 375, 385–388.
Patel RN (2001) Biocatalytic synthesis of intermediates for the synthesis of chiral drug substances. Curr Opin Biotechnol 12, 587–604.
Popiolkiewicz J, Polkowski K, Skierski JS, and Mazurek AP (2005) In vitro toxicity evaluation in the development of new anticancer drugs-genistein glycosides. Cancer Lett 229, 67–75.
Schmidt A, Li C, Shi F, Jones AD, and Pichersky E (2011) Polymethylated myricetin in trichomes of the wild tomato species Solanum habrochaites and characterization of trichome-specific 3/5- and 7/4-myricetin O-methyltransferases. Plant Physiol 155, 1999–2009.
Suomela JP, Ahotupa M, Yang B, Vasankari T, and Kallio H (2006) Absorption of flavonols derived from sea buckthorn (Hippophaë rhamnoides L.) and their effect on emerging risk factors for cardiovascular disease in humans. J Agric Food Chem 54, 7364–7369.
Tamura K, Peterson D, Peterson N, Stecher G, Nei M, and Kumar S (2011) MEGA5: Molecular evolutionary genetics analysis using maximum likelihood, evolutionary distance, and maximum parsimony methods. Mol Biol Evol 28, 2731–2739.
Williams CA and Grayer RJ (2004) Anthocyanins and other flavonoids. Nat Prod Rep 21, 539–573.
Winkel-Shirley B (2002) Biosynthesis of flavonoids and effects of stress. Curr Opin Plant Biol 5, 18–223.
Woo Y, Chin SY, Hyun J, Lee SD, Lee YH, and Lim Y (2012) Flavonones inhibit the clonogenicity of HCT116 cololectal cancer cells. Int J Mol Med 29, 403–408.
Zhang L, Kong Y, Wu D, Zhang H, Wu J, Chen J et al. (2008) Three flavonoids targeting the â-hydroxyacyl-acyl carrier protein dehydratase from Helicobacter pylori: Crystal structure characterization with enzymatic inhibition assay. Protein Sci 17, 1971–1978.
Author information
Authors and Affiliations
Corresponding authors
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Cho, MH., Park, H.L., Park, JH. et al. Characterization of regiospecific flavonoid 3′/5′-O-methyltransferase from tomato and its application in flavonoid biotransformation. J Korean Soc Appl Biol Chem 55, 749–755 (2012). https://doi.org/10.1007/s13765-012-2193-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13765-012-2193-3