Abstract
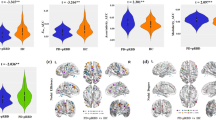
Search for Parkinson’s disease (PD) progression biomarkers has led to the identification of both motor and non-motor symptoms relevant of prodromal PD that could be eye-opening to the spreading underlying Lewy body pathogenesis. One most robust predictor of PD is the REM sleep behavior disorder (RBD), and one most common early non-motor symptom of PD is depression. With RBD, frequently coexisting with clinical depression and both predicting dopamine transmission dysfunction, we aimed to survey structural associates of depressive symptoms in early PD patients with comorbid RBD. Through diffusion MRI connectometry, we tracked fiber differences comparing DWI images obtained from 14 patients with depressive symptoms and 18 without depression from a group with comorbid RBD and PD. DWI images and patients were recruited from the Parkinson’s Progression Markers Initiative database. PD-RBD patients with depressive symptoms showed pathways with significantly reduced connectivity in the right cingulum, left and right fornix, left inferior longitudinal fasciculus, right corticospinal tract, left middle cerebellar peduncle and genu of corpus callosum (FDR = 0.0228). Diffusivity alteration of the mentioned fibers in depressed, early PD patients with RBD might reflect underlying PD pathology and serve as common structural DWI signatures for early PD diagnosis.

Similar content being viewed by others
References
Devi L, Raghavendran V, Prabhu BM, Avadhani NG, Anandatheerthavarada HK (2008) Mitochondrial import and accumulation of α-synuclein impair complex I in human dopaminergic neuronal cultures and Parkinson disease brain. J Biol Chem 283(14):9089–9100
Braak H, Ghebremedhin E, Rüb U, Bratzke H, Del Tredici K (2004) Stages in the development of Parkinson’s disease-related pathology. Cell Tissue Res 318(1):121–134
Munhoz RP, Moro A, Silveira-Moriyama L, Teive HA (2015) Non-motor signs in Parkinson’s disease: a review. Arq Neuropsiquiatr 73(5):454–462
Zhang JR, Chen J, Yang ZJ, Zhang HJ, Fu YT, Shen Y, He PC, Mao CJ, Liu CF (2016) Rapid eye movement sleep behavior disorder symptoms correlate with domains of cognitive impairment in Parkinson’s disease. Chin Med J 129(4):379–385. https://doi.org/10.4103/0366-6999.176077
Lim JS, Shin SA, Lee JY, Nam H, Lee JY, Kim YK (2016) Neural substrates of rapid eye movement sleep behavior disorder in Parkinson’s disease. Parkinsonism Relat Disord 23:31–36. https://doi.org/10.1016/j.parkreldis.2015.11.027
Zoccolella S, Savarese M, Lamberti P, Manni R, Pacchetti C, Logroscino G (2011) Sleep disorders and the natural history of Parkinson’s disease: the contribution of epidemiological studies. Sleep Med Rev 15(1):41–50
Fujishiro H, Okuda M, Iwamoto K, Miyata S, Otake H, Noda A, Iritani S, Ozaki N (2016) REM sleep without atonia in middle-aged and older psychiatric patients and Lewy body disease: a case series. Int J Geriatr Psychiatry. https://doi.org/10.1002/gps.4477
Howell MJ, Schenck CH (2015) Rapid eye movement sleep behavior disorder and neurodegenerative disease. JAMA Neurol 72(6):707–712. https://doi.org/10.1001/jamaneurol.2014.4563
Heller J, Brcina N, Dogan I, Holtbernd F, Romanzetti S, Schulz JB, Schiefer J, Reetz K (2016) Brain imaging findings in idiopathic REM sleep behavior disorder (RBD)—A systematic review on potential biomarkers for neurodegeneration. Sleep Med Rev. https://doi.org/10.1016/j.smrv.2016.06.006
Schrag A, Barone P, Brown RG, Leentjens AF, McDonald WM, Starkstein S, Weintraub D, Poewe W, Rascol O, Sampaio C, Stebbins GT, Goetz CG (2007) Depression rating scales in Parkinson’s disease: critique and recommendations. Mov Disord Off J Mov Disord Soc 22(8):1077–1092. https://doi.org/10.1002/mds.21333
Postuma RB, Berg D (2016) Advances in markers of prodromal Parkinson disease. Nat Rev Neurol 12(11):622–634. https://doi.org/10.1038/nrneurol.2016.152
Rahmani F, Ansari M, Pooyan A, Mirbagheri MM, Aarabi MH (2016) Differences in white matter microstructure between Parkinson’s disease patients with and without REM sleep behavior disorder. In: Engineering in Medicine and Biology Society (EMBC), 2016 IEEE 38th annual international conference of the, 2016. IEEE, pp 1124–1126
Ansari M, Rahmani F, Dolatshahi M, Pooyan A, Aarabi MH (2017) Brain pathway differences between Parkinson's disease patients with and without REM sleep behavior disorder. Sleep breath 21(1):155–161. https://doi.org/10.1007/s11325-016-1435-8
Jooyandeh SM, Baghai T, Aarabi M, Dolatshahi M, Langguth B (2017) Brain pathway differences between Parkinson’s disease patients with and without depressive symptoms. Eur Psychiatry 41:S349
Wen M-C, Heng HS, Ng SY, Tan LC, Chan LL, Tan EK (2016) White matter microstructural characteristics in newly diagnosed Parkinson’s disease: an unbiased whole-brain study. Sci Rep 6:35601
Marek K, Jennings D, Lasch S, Siderowf A, Tanner C, Simuni T, Coffey C, Kieburtz K, Flagg E, Chowdhury S (2011) The parkinson progression marker initiative (PPMI). Prog Neurobiol 95(4):629–635
Gagnon JF, Bedard MA, Fantini ML, Petit D, Panisset M, Rompre S, Carrier J, Montplaisir J (2002) REM sleep behavior disorder and REM sleep without atonia in Parkinson’s disease. Neurology 59(4):585–589
Mahajan A, Rosenthal LS, Gamaldo C, Salas RE, Pontone GM, McCoy A, Umeh C, Mari Z (2014) REM sleep behavior and motor findings in Parkinson’s disease: a cross-sectional analysis. Tremor Other Hyperkinet Mov (New York, NY) 4:245. https://doi.org/10.7916/d84b2zdf
Stiasny-Kolster K, Mayer G, Schafer S, Moller JC, Heinzel-Gutenbrunner M, Oertel WH (2007) The REM sleep behavior disorder screening questionnaire—a new diagnostic instrument. Mov Disord Off J Mov Disord Soc 22(16):2386–2393. https://doi.org/10.1002/mds.21740
Bouwmans AEP, Weber WEJ (2012) Neurologists’ diagnostic accuracy of depression and cognitive problems in patients with parkinsonism. BMC Neurol 12:37–37. https://doi.org/10.1186/1471-2377-12-37
Leemans A, Jeurissen B, Sijbers J, Jones D (2009) ExploreDTI: a graphical toolbox for processing, analyzing, and visualizing diffusion MR data. In: 17th annual meeting of intl soc mag reson med, 2009, p 3537
Yeh FC, Badre D, Verstynen T (2015) Connectometry: a statistical approach harnessing the analytical potential of the local connectome. NeuroImage. https://doi.org/10.1016/j.neuroimage.2015.10.053
Yeh FC, Tseng WY (2011) NTU-90: a high angular resolution brain atlas constructed by q-space diffeomorphic reconstruction. Neuroimage 58(1):91–99. https://doi.org/10.1016/j.neuroimage.2011.06.021
Yeh F-C, Vettel JM, Singh A, Poczos B, Grafton ST, Erickson KI, Tseng W-YI, Verstynen TD (2016) Quantifying differences and similarities in whole-brain white matter architecture using local connectome fingerprints. PLoS Comput Biol 12(11):e1005203. https://doi.org/10.1371/journal.pcbi.1005203
Yeh FC, Badre D, Verstynen T (2016) Connectometry: a statistical approach harnessing the analytical potential of the local connectome. Neuroimage 125:162–171. https://doi.org/10.1016/j.neuroimage.2015.10.053
Yeh F-C, Panesar S, Fernandes D, Meola A, Yoshino M, Fernandez-Miranda JC, Vettel J, Verstynen T (2017) A population-based atlas of the macroscale structural connectome in the human. Brain. bioRxiv. https://doi.org/10.1101/136473
Scherfler C, Frauscher B, Schocke M, Iranzo A, Gschliesser V, Seppi K, Santamaria J, Tolosa E, Hogl B, Poewe W (2011) White and gray matter abnormalities in idiopathic rapid eye movement sleep behavior disorder: a diffusion-tensor imaging and voxel-based morphometry study. Ann Neurol 69(2):400–407. https://doi.org/10.1002/ana.22245
Wing YK, Li SX, Mok V, Lam SP, Tsoh J, Chan A, Yu MWM, Lau CY, Zhang J, Ho CKW (2012) Prospective outcome of rapid eye movement sleep behaviour disorder: psychiatric disorders as a potential early marker of Parkinson’s disease. J Neurol Neurosurg Psychiatry 83(4):470–472
Fereshtehnejad S-M, Romenets SR, Anang JB, Latreille V, Gagnon J-F, Postuma RB (2015) New clinical subtypes of Parkinson disease and their longitudinal progression: a prospective cohort comparison with other phenotypes. JAMA Neurol 72(8):863–873
Borek LL, Kohn R, Friedman JH (2006) Mood and sleep in Parkinson’s disease. J Clin Psychiatry 67(6):958–963
Wing YK, Lam SP, Zhang J, Leung E, Ho CL, Chen S, Cheung MK, Li SX, Chan JW, Mok V, Tsoh J, Chan A, Ho CK (2015) Reduced striatal dopamine transmission in REM sleep behavior disorder comorbid with depression. Neurology 84(5):516–522. https://doi.org/10.1212/wnl.0000000000001215
Ford AH, Duncan GW, Firbank MJ, Yarnall AJ, Khoo TK, Burn DJ, O’Brien JT (2013) Rapid eye movement sleep behavior disorder in Parkinson’s disease: magnetic resonance imaging study. Mov Disord 28(6):832–836
Boucetta S, Salimi A, Dadar M, Jones BE, Collins DL, Dang-Vu TT (2016) Structural brain alterations associated with rapid eye movement sleep behavior disorder in Parkinson’s disease. Sci Rep 6:26782
Aarabi MH, Kamalian A, Mohajer B, Shandiz MS, Eqlimi E, Shojaei A, Safabakhsh H (2015) A statistical approach in human brain connectome of Parkinson Disease in elderly people using Network Based Statistics. In: conference proceedings: annual international conference of the IEEE Engineering in Medicine and Biology Society IEEE Engineering in Medicine and Biology Society Annual Conference 2015: 4310–4313. https://doi.org/10.1109/embc.2015.7319348
Jooyandeh SM, Aarabi MH, Rahmani F (2017) Microstructural changes in patients with Parkinson’s diseases and REM sleep behavior disorder: depressive symptoms versus non-depressed. Eur Psychiatry 41(Supplement):S287. https://doi.org/10.1016/j.eurpsy.2017.02.147
Luo C, Song W, Chen Q, Yang J, Gong Q, Shang H-F (2016) Cortical thinning in drug-naive Parkinson’s disease patients with depression. J Neurol 263(10):2114–2119
Ebert D, Ebmeier KP (1996) The role of the cingulate gyrus in depression: from functional anatomy to neurochemistry. Biol Psychiatry 39(12):1044–1050
Wen MC, Chan L, Tan L, Tan E (2016) Depression, anxiety, and apathy in Parkinson’s disease: insights from neuroimaging studies. Eur J Neurol 23(6):1001–1019
Zgaljardic DJ, Borod JC, Foldi NS, Mattis P (2003) A review of the cognitive and behavioral sequelae of Parkinson’s disease: relationship to frontostriatal circuitry. Cognit Behav Neurol 16(4):193–210
Tymofiyeva O, Connolly CG, Ho TC, Sacchet MD, Blom EH, LeWinn KZ, Xu D, Yang TT (2017) DTI-based connectome analysis of adolescents with major depressive disorder reveals hypoconnectivity of the right caudate. J Affect Disord 207:18–25
Matsui H, Nishinaka K, Oda M, Niikawa H, Komatsu K, Kubori T, Udaka F (2007) Depression in Parkinson’s disease. J Neurol 254(9):1170–1173
Sobhani S, Rahmani F, Aarabi MH, Sadr AV (2017) Exploring white matter microstructure and olfaction dysfunction in early parkinson disease: diffusion MRI reveals new insight. Brain Imaging Behav. https://doi.org/10.1007/s11682-017-9781-0
Rahmani F, Aarabi MH (2017) Does apolipoprotein A1 predict microstructural changes in subgenual cingulum in early Parkinson? J Neurol. https://doi.org/10.1007/s00415-017-8403-5
Haghshomar M, Rahmani F, Aarabi MH, Shahjouei S, Sobhani S, Rahmani M (2017) White matter changes correlates of peripheral neuroinflammation in patients with Parkinson's disease. Neuroscience. https://doi.org/10.1016/j.neuroscience.2017.10.050
Fox MD, Buckner RL, White MP, Greicius MD, Pascual-Leone A (2012) Efficacy of transcranial magnetic stimulation targets for depression is related to intrinsic functional connectivity with the subgenual cingulate. Biol Psychiatry 72(7):595–603
Hall JM, Martens KAE, Walton CC, O’Callaghan C, Keller PE, Lewis SJ, Moustafa AA (2016) Diffusion alterations associated with Parkinson’s disease symptomatology: a review of the literature. Parkinsonism Relat Disord 33:12–26. https://doi.org/10.1016/j.parkreldis.2016.09.026
Huang P, Xu X, Gu Q, Xuan M, Yu X, Luo W, Zhang M (2014) Disrupted white matter integrity in depressed versus non-depressed Parkinson’s disease patients: a tract-based spatial statistics study. J Neurol Sci 346(1):145–148
Kamarajah SK (2016) Postural instability and gait disorder (PIGD) in Parkinson’s disease: is there another target for deep brain stimulation? (P6.381). Neurology 86(16 Supplement):P6
Bloom JS, Hynd GW (2005) The role of the corpus callosum in interhemispheric transfer of information: excitation or inhibition? Neuropsychol Rev 15(2):59–71
Won E, Choi S, Kang J, Kim A, Han K, Chang H, Tae W, Son K, Joe S, Lee M (2016) Association between reduced white matter integrity in the corpus callosum and serotonin transporter gene DNA methylation in medication-naive patients with major depressive disorder. Transl Psychiatry 6(8):e866
Stiasny-Kolster K, Sixel-Döring F, Trenkwalder C, Heinzel-Gutenbrunner M, Seppi K, Poewe W, Högl B, Frauscher B (2015) Diagnostic value of the REM sleep behavior disorder screening questionnaire in Parkinson’s disease. Sleep Med 16(1):186–189
Acknowledgements
PPMI is funded by Grants from the Michael J Fox Foundation for Parkinson’s Research, the W Garfield Weston Foundation, and the Alzheimer’s Association, the Canadian Institutes for Health Research, and the Natural Sciences and Engineering Research Council of Canada. We thank Christian Beckmann and Simon Eickhoff for their advice on data analysis. Data used in this article were obtained from the Parkinson’s Progression Markers Initiative (PPMI) database (http://www.ppmi-info.org/data). For up-to-date information on the study, visit http://www.ppmi-info.org. PPMI is sponsored and partially funded by the Michael J Fox Foundation for Parkinson’s Research and funding partners, including AbbVie, Avid Radiopharmaceuticals, Biogen, Bristol-Myers Squibb, Covance, GE Healthcare, Genentech, GlaxoSmithKline (GSK), Eli Lilly and Company, Lundbeck, Merck, Meso Scale Discovery (MSD), Pfizer, Piramal Imaging, Roche, Servier, and UCB (http://www.ppmi-info.org/fundingpartners).
Funding
No funding was received for this research.
Author information
Authors and Affiliations
Contributions
FGS, FR, and MHA contributed to the conception and design of the study; MHA contributed to data collection and analysis; and FGS, FR, and SMJ contributed to writing the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors of this manuscript declare no relationships with any companies, whose products or services may be related to the subject matter of the article.
Ethical approval
All procedures performed here, including human participants, were in accordance with the ethical standards of the institutional research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed consent
Informed consent had been obtained from all individual participants included in the study, by the PPMI.
Rights and permissions
About this article
Cite this article
Ghazi Sherbaf, F., Rahmani, F., Jooyandeh, S.M. et al. Microstructural changes in patients with Parkinson disease and REM sleep behavior disorder: depressive symptoms versus non-depressed. Acta Neurol Belg 118, 415–421 (2018). https://doi.org/10.1007/s13760-018-0896-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13760-018-0896-x