Abstract
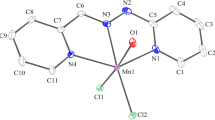
Lithium-7 NMR spectroscopy was used to investigate the stoichiometry and stability of a Li+ complex with N1,N2-bis(pyridin-2-ylmethylene)ethane-1,2-diamine (L1), N1,N3-bis(pyridin-2-ylmethylene)propane-1,3-diamine (L2) and N1,N4-bis(pyridin-2-ylmethylene) butane-1,4-diamine (L3) in acetonitrile (AN) and nitromethane (NM) solutions. A competitive 7Li NMR method was also employed to probe the complexation of Mn2+, Cd2+ and Zn2+ ions with L1, L2 and L3 in the same solvents. The formation constants of the resulting complexes were evaluated from computer fitting of the mole ratio data to an equation that relates the observed chemical shifts to the formation constant. In both solvents, the stability of the resulting 1:1 complexes were found to vary in the order Zn2+>Cd2+>Mn2+>Li+. In addition, the stability of M–L complexes of M2+ with the Schiff base ligands found to vary in the order M2+–L1 > M2+–L2 > M2+–L3.









Similar content being viewed by others
References
Q. Shi, L. Xu, J. Ji, Y. Li, R. Wang, Z. Zhou, R. Cao, M. Hong, A.S.C. Chan, Inorg. Chem. Commun. 7, 1254 (2004)
Z.-L. You, H.-L. Zhu, W.-S. Liu, Z. Anorg, Allg. Chem. 630, 1617 (2004)
A. Golcu, M. Tumer, H. Demirelli, R.A. Wheatley, Inorg. Chim. Acta 358, 1785 (2005)
R. Ziessel, Coord. Chem. Rev. 216, 195 (2001)
M. Albrecht, Chem. Rev. 101, 3457 (2001)
B.B. Tewari, J. Chromatogr. A 718, 454 (1995)
A.K. Pandey, S.D. Pandey, V. Misra, Ecotoxicol. Environ. Saf. 47, 195 (2000)
G. Ibrahim, G.M. Bouet, I.H. Hall, M.A. Khan, J. Inorg. Biochem. 81, 29 (2000)
S.V. Pakhomova, M.A. Proskurnin, V.V. Chernysh, M.Y. Kononets, E.K. Ivanova, J. Anal. Chem. 56, 910 (2001)
S. Hulanicki, A. Glab, G. Ackermann, Pure Appl. Chem. 55, 1137 (1983)
J. Zhou, Z.H. Vin, J.X. Qu, H.Y. Shum, Talanta 43, 1863 (1996)
B.J. Colston, V.J. Robinson, Analyst 122, 1451 (1997)
E.R. Garrett, D.J. Weber, J. Pharm. Sci. 59, 1383 (2006)
H. Doe, K. Wakamiya, T. Kitagawa, B. Chem, Soc. Jpn. 60, 2231 (1987)
A. Safavi, L. Fotouhi, Microchem. J. 59, 351 (1998)
A. Afkhami, T. Madrakian, H. Keypour, S. Soltanbeygi, F. Khajavi, M. Rezaeivala, J. Mol. Struct. 985, 86 (2011)
A. Afkhami, H. Keypour, F. Khajavi, M. Rezaeivala, J. Chem. Eng. Data 55, 4725 (2010)
M. Shamsipur, A.I. Popov, Inorg. Chim. Acta 43, 1243 (1980)
H. Keypour, A.A. Dehghani-Firouzabadi, H.R. Khavasi, Polyhedron 28, 1546 (2009)
E.T. Roach, P.R. Handy, A.I. Popov, Inorg. Nucl. Chem. Lett. 9, 359 (1973)
M. Shamsipur, L. Shafiee Dastjerdi, N. Alizadeh, H.R. Bijanzadeh, Spectrochim. Acta A 69, 1265 (2008)
V.A. Nicely, J.L. Dye, J. Chem. Educ. 48, 443 (1971)
R.K. Harris, B.E. Mann, NMR and the periodic Table (Academic Press, New York, 1978)
E. Karkhaneei, J. Zolgharnein, A. Afkhami, M. Shamsipur, J. Coord. Chem. 46, 1 (1998)
M. Shamsipur, E. Karkhaneei, A. Afkhami, Polyhedron 17, 3809 (1998)
M. Shamsipur, E. Karkhaneei, A. Afkhami, J. Coord. Chem. 44, 23 (1998)
R.M. Izatt, J.S. Bradshaw, S.A. Nielson, J.D. Lamb, J.J. Christensen, D. Sen, Chem. Rev. 85, 271 (1985)
R.M. Izatt, K. Pawlak, J.S. Bradshaw, R.L. Bruening, B.J. Tarbet, Chem. Rev. 92, 1261 (1992)
B. Tüemmler, G. Maass, F. Vögtle, H. Sieger, U. Heimann, E. Weber, J. Am. Chem. Soc. 99, 4683 (1977)
B. Tüemmler, G. Maass, F. Vögtle, H. Sieger, U. Heimann, E. Weber, J. Am. Chem. Soc. 101, 2588 (1979)
H.-J. Buschmann, Inorg. Chim. Acta 102, 95 (1985)
J.H. Jung, I. Yoon, K.J. Park, S.S. Lee, K.S. Choi, S.B. Park, Microchem. 63, 100 (1999)
R.D. Shannon, Acta Crystallogr. Sect. A 32, 751 (1976)
R.G. Pearson, J. Am. Chem. Soc. 84, 3533 (1963)
R. Barbucci, P. Paoletti, A. Vacca, Inorg. Chem. 14, 302 (1975)
H. Keypour, M. Dehdari, S. Salehzadeh, K.P. Wainwright, Trans. Met. Chem. 28, 425 (2003)
M. Shamsipur, Z. Talebpour, N. Alizadeh, J. Solution Chem. 32, 227 (2003)
R.M. Izatt, K. Pawlak, J.S. Bradshaw, R.L. Bruening, Chem. Rev. 91, 1721 (1991)
M. Shamsipur, E. Karkhanei, A. Afkhami, J. Coord. Chem. 44, 23 (1998)
E. Karkhanei, J. Zolgharnein, A. Afkhami, M. Shamsipur, J. Coord. Chem. 46, 1 (1998)
E. Karkhanei, M.H. Zebarjadian, M. Shamsipur, J. Chin. Chem. Soc. 48, 727 (2001)
E. Karkhanei, M.H. Zebarjadian, M. Shamsipur, J. Incl. Phenom. 54, 309 (2006)
M. Shamsipur, N. Alizadeh, M.K. Rofouei, K. Alizadeh, Pol. J. Chem. 81, 1743 (2007)
M. Shamsipur, M. Irandoust, J. Solution Chem. 37, 657 (2008)
M. Irnadoust, M. Shamsipur, H. Darabi, J. Incl. Phenom. 66, 365 (2010)
E. Karkhaneei, M.H. Zebarjadian, M. Shamsipur, J. Solution Chem. 30, 323 (2001)
V. Gutmann, The Donor-Acceptor Approach to Molecular Interactions (Plenum Press, New York, 1978)
Acknowledgments
The financial support Faculty of Research Council of Bu-Ali Sina University gratefully acknowledged.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Keypour, H., Zebarjadian, M.H., Rezaeivala, M. et al. Complexation studies of Mn2+, Zn2+ and Cd2+ ions with a series of tetradentate (N4) Schiff base ligands containing pyridine moiety in acetonitrile and nitromethane solutions by a competitive NMR technique using 7Li nucleus as a probe. J IRAN CHEM SOC 10, 1137–1143 (2013). https://doi.org/10.1007/s13738-013-0248-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13738-013-0248-y