Abstract
Purpose of Review
Irritant contact dermatitis is the most common form of contact dermatitis and the most common occupational skin disease. This review provides a comprehensive summary of the endogenous and exogenous factors that play a role in the pathogenesis of irritant contact dermatitis.
Recent Findings
In conjunction with avoidance of irritants, barrier protection, and regular application of moisturizers, management now emphasizes the importance of primary prevention through educational initiatives in high-risk workplaces.
Summary
The diagnosis of irritant contact dermatitis is often difficult, as there is no confirmatory test, and it is often a default diagnosis after allergic contact dermatitis has been excluded. Early recognition, prevention, and treatment are vital in management, especially in the occupational setting.
Similar content being viewed by others
Introduction
Irritant contact dermatitis (ICD) is an inflammatory cutaneous condition caused by skin barrier disruption, in combination with the activation of innate immune responses. ICD results from skin barrier damage from external agents or environmental factors. ICD commonly affects the hands and can affect people of all ages and sex. ICD is more prevalent than allergic contact dermatitis (ACD) [1] and is the most common presentation of occupational skin disease (OSD). At a tertiary referral occupational dermatology clinic in Australia, ICD was diagnosed in 71% of patients with OSD [2••]. However, in the general patch testing population, the most common diagnosis is ACD with a default diagnosis of an endogenous dermatitis, so ICD is less commonly diagnosed [3]. High-risk occupations for ICD include healthcare workers, food service workers, metal workers, hairdressers, and construction workers [4].
The purpose of this review is to provide a comprehensive update with regard to the pathogenesis, risk factors, clinical features, and management of ICD.
Contributing Factors in the Development of ICD
Both exogenous and endogenous factors play a vital role in the pathogenesis of ICD [5].
Exogenous
Skin Irritants
The most common skin irritant is wet work [6] followed by soaps, detergents, solvents, and oils [7]. Wet work has been defined as:
-
1.
Exposure of skin to liquid for > 2 h per day
-
2.
Use of occlusive gloves for > 2 h per day or change of gloves > 20 times per day
-
3.
Frequent hand washing > 20 times per day or use of hand disinfectants > 20 times per day [8]
Exposure to wet work can occur both at work as well as at home. High-risk occupations involving wet work include cleaners, butchers, cooks, beauticians, and health care workers [9•]. The only regulations regarding exposure to wet work are found in Germany, the Technical Standards for Hazardous Substances (TRGS) 401[10], a guideline for employers, providing information on occupational hazardous skin exposure and prevention of OSD.
Since the start of the COVID-19 pandemic, the use of soaps and alcohol-based hand sanitizers (ABHS) and the frequency of handwashing have increased significantly [11] resulting in a higher incidence of ICD [12, 13].
The potency of a chemical irritant and its ability to penetrate the skin are denoted by its properties including molecular size, ionization state, and fat solubility. Different irritants target different structures in the epidermis. For example, sodium lauryl sulfate targets lipid synthesis [14] whereas acetone targets the proliferation of basal keratinocytes [15].
Factors such as concentration, volume, and duration of irritant exposure will all contribute to an irritant’s ability to penetrate the skin. Increasing the volume and duration of exposure will enhance the ability of an irritant to penetrate the skin [16]. Increasing intervals between exposures will usually reduce the chance of irritation. Recurrent irritant exposure may result in an additive effect, with repeated exposures initially inducing an increase, then a reduction in trans-epidermal water loss (TEWL) [17], indicating functional adaptation or skin hardening.
Physical, Mechanical, and Environmental Factors
The role of physical, mechanical, and environmental factors in the development of ICD is often ignored. Mechanical factors include occlusion, friction, pressure, and vibration. The use of gloves or clothing can create an occlusive, humid environment, which increases irritation caused by heat and sweating.
Environmental factors include heat, cold, humidity, and UV irradiation. Exposure of skin to heat often results in sweating. The retention of sweat can contribute to skin irritation, as sweat is more irritating than water [18]. Heat, especially in combination with occlusion, can precipitate ICD.
Exposure to low ambient humidity and cold temperatures are contributing factors to reduced water content in the stratum corneum (SC) and as a result increase the permeability of irritants in the skin. Cold temperatures have also been associated with reducing the plasticity of the horny layer with subsequent cracking of the SC; however, a study has shown that exposure to cold averted the development of ICD [19].
Endogenous
Age
Susceptibility of the skin to irritants decreases with age. It is postulated that differences in microcirculatory efficiency, percutaneous penetration, SC turnover time, and loss of corneocyte cohesion [20] associated with increasing age cause a slower and less intense response to irritants. This is reflected in the decrease of TEWL in aged skin [21].
Sex
ICD is more common in women [22], likely as a result of greater exposure to irritants rather than biological factors. A study from Denmark showed women had 78% higher odds of engaging in occupations involving wet work [9•]. It is not clear whether women have a greater susceptibility to irritants, given that experimental studies show no difference of irritant reactivity between sexes [21]. However, one study produced differing results [23] demonstrating an increased skin reactivity in males compared to females. Further studies are required to determine the difference in risk of developing ICD between sexes.
Race
There is a paucity of evidence to demonstrate a significant difference between black or Asian and Caucasian skin. Studies using bioengineering techniques showed no difference in irritant susceptibility between black and Caucasian skin [24] despite conflicting results from previous studies that relied on visual scoring [25]. A study showed Asian skin was significantly more susceptible to ICD when compared to Caucasian skin both in subjective and objective skin measurements [26].
Atopy
Atopy is a recognized risk factor for ICD involving the hands [27]. Mucosal atopy is less predictive of irritant reactivity, compared to prior atopic dermatitis (AD) [28]. Individuals with a history of AD are thought to have a higher risk for the development of ICD, relating to impairment of the epidermal barrier, higher TEWL, and increased skin permeability to allergens and irritants, compared to those with clinically normal skin [29].
Genetic Factors
Filaggrin is a protein which plays a role in skin barrier function and SC hydration. Filaggrin loss-of-function mutations are associated with increased expression of interleukin (IL)-1, a cytokine which is key to the initiation of the inflammatory response in ICD [30]. A study has shown an increased risk of the development of ICD in individuals with filaggrin loss-of-function mutations; however, it was observed that the association was dependent on the presence of atopy [31]. Atopy, in combination with filaggrin loss-of-function mutation carrier status, has shown to contribute to the severity of ICD affecting the hands [32].
Single nucleotide polymorphisms (SNPs) within the major histocompatibility complex (MHC) class I, II, and III are significantly associated with skin response to irritants [33, 34]. MHC SNPs have been demonstrated to contribute to chemical irritancy thresholds, with different MHC variants being associated with different chemical irritants. At present, the exact mechanism is unknown.
A link has been recognized between a tumor necrosis factor (TNF)-α gene polymorphism and susceptibility to ICD. At the position P308 within the promoter region of the TNF-α gene, a G to A transition polymorphism has been detected. A study of genotypes showed an association between the A allele and a low irritant threshold [35], which has also been significantly linked to contact sensitization to allergens such as p-phenylenediamine (PPD) [36] and chromium [37]. Another G to A transition polymorphism at the position of P238 has been identified. A study found a potential protective effect of the A allele with carriers being less susceptible to developing ICD [38]. This has been speculated to result from increased transcriptional repression of TNF α [39, 40].
These advancements represent a novel potential approach in the detection of ICD susceptibility.
Pathogenesis
Previously, the pathogenesis of ICD was thought to be a non-immunological reaction; however, it is now recognized that the immune system plays a vital role in causing ICD [41].
Irritation occurs by either damaging epidermal cells [42], disruption of the epidermal barrier, or a combination of both [43]. Disruption of the epithelial barrier allows for increased permeability of irritants [44]. Keratinocytes act as “signal transducers” responsible for instigating cutaneous inflammation via the conversion of exogenous stimuli into the secretion of cytokines, adhesion molecules, and chemotactic factors [45]. Upon damage to the keratinocyte, upregulation of primary cytokines IL-1α, IL-1β, and TNF-α occurs [46] which triggers the proliferation of keratinocytes and formation of lipids [47] playing a role in restoring the epidermal barrier. Additional cytokines IL-6, IL-8, and GM-SCF are subsequently secreted, activating Langerhans cells, dermal dendritic cells, and endothelial cells [44] which further recruits inflammatory cells to the site of chemical trauma [48].
ICAM1, an adhesion molecule, is upregulated on endothelial cells and fibroblasts in the skin and results in the secretion of further chemokines including CXCL 8, CCL 20, and IFN gamma [49]. CCL21, a chemokine which enables naïve T lymphocyte migration, is upregulated in ICD [46, 50]. T lymphocytes recruited to irritated skin often express the CLA antigen [51], which plays an important role in transendothelial T lymphocyte migration.
Irritants can also trigger pattern recognition receptors such as toll-like receptors (TLRs) and NOD-like receptors, which result in activation of the innate immune response via inflammasome and NFĸB pathways [44, 52].
There is growing evidence that oxidative stress with the formation of reactive oxygen species plays a role in the pathogenesis of ICD [53]. It has been postulated that targeting oxidative stress could be beneficial in the treatment of ICD, with clinical studies demonstrating the therapeutic benefits of antioxidants [54].
Clinical Types
Acute Irritant Contact Dermatitis
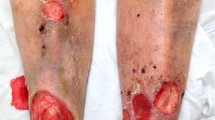
Acute ICD occurs when the skin is exposed to a potent irritant often in a single exposure, such as concentrated acids, strong alkalis, and solvents such acrylonitrile. In severe cases, this may present as burns, such as from kneeling in wet concrete.
The irritant reaction quickly reaches a peak and then begins to heal, usually described as a “decrescendo phenomenon” [55]. Symptoms reported include burning, stinging, and soreness of affected skin. The clinical signs of ICD are variable and include erythema, edema, bullae, and necrosis [56]. While ACD may present with similar clinical signs, it is characterized by a “crescendo phenomenon” where signs worsen, despite removal of the offending allergen [55].
Complete healing can take weeks in acute ICD, generally with a good prognosis [57].
Delayed Acute Irritant Contact Dermatitis
Delayed acute ICD represents the most common form of ICD. Chemicals such as benzalkonium chloride, anthralin, calcipotriol, and tretinoin may cause a delayed inflammatory response, not visible until 8–24 h after primary exposure [58]. This may lead to misdiagnosis as ACD. Clinical symptoms are similar to acute ICD with the skin becoming sensitive to touch and water [59].
Irritant Reaction
Irritant reaction is a subclinical type of ICD that typically affects individuals exposed to wet work. Clinical features are monomorphic, characterized by erythema, scaling, vesicles, pustules, and erosions [59]. It often begins on fingers under occlusive jewelry such as rings and spreads to involve the hands and forearms.
This condition may spontaneously resolve with cessation of exposure, or can also progress to cumulative ICD [60].
Subjective/Sensorial Irritation
Subjective or sensorial irritation is characterized as sensory discomfort in the form of stinging, burning, or a sensation of itch, in the absence of clinical or histological evidence of skin irritation. Neural pathways are believed to be responsible [61]. Irritants identified include propylene glycol, hydroxy acids, ethanol, lactic acid, azelaic acids, benzoic acid, benzoyl peroxide, mequinol, and tretinoin.
Non-Erythematous Irritation
Non-erythematous ICD refers to early skin irritation without clinical signs of inflammation. Associated SC barrier changes can be demonstrated using assays that are usually available only in research settings [62]. Symptoms are comparable to subjective irritation. Prognosis is variable.
Cumulative (Chronic) ICD
Cumulative ICD is a result of multiple subthreshold insults to the skin, if the interval between skin exposures is too short to allow complete recovery of the skin barrier function [63]. It develops slowly and is linked to exposures to weak irritants [64].
Clinical features include erythema, vesicles, and dryness with progression to lichenification, hyperkeratosis, and chapping. Clinical features develop when the damage goes beyond the elicitation threshold. The threshold is dependent on the individual and may decrease as the disease progresses.
Given that exposure to weak irritants often occurs both at home and in the workplace, cumulative ICD may result from exposure to multiple irritants, rather than a single agent. The impact of combined multiple irritants produces an increased skin inflammatory response; however, the extent of interactive effect is unpredictable [65].
Frictional Dermatitis
Frictional dermatitis results from repeated frictional trauma, specifically shearing forces acting horizontally to the skin surface [66]. It is characterized by hyperkeratosis, acanthosis, and lichenification. Frictional ICD is recognized to contribute to ACD by enhancing percutaneous penetration of allergens [66].
Traumatic ICD
Traumatic ICD occurs after acute trauma to the skin such as burns, lacerations, or exposure to a potent irritant. The hands are the most commonly involved; however, there are reported cases of nail involvement [67]. Usually, there is delayed healing of the original trauma followed by the development of eczematous lesions [64]. It is usually a chronic condition and may be resistant to treatment.
Pustular and Acneiform Dermatitis
Exposure to oils, naphthalene, chlorinated aromatic hydrocarbons, and fluorinated compounds may result in pustular and acneiform dermatitis. Patients most affected are those with seborrheic dermatitis, prior acne vulgaris, and atopy. The prognosis is variable.
Asteatotic Irritant Dermatitis
Asteatotic irritant dermatitis, also referred to as “exsiccation eczematid,” “winter eczema,” and “eczema craquelé,” is seen primarily during winter in cool climates. This variant is common amongst the elderly with dry skin on the lower legs, especially in low-humidity environments. Individuals report intense pruritus, dry skin, and scaling. Reduction in natural moisturizing factors and lipids in the SC is thought to contribute. Intense pruritus is usually relieved by the use of moisturizing creams, and topical corticosteroid ointments are helpful [68].
Airborne Dermatitis
Airborne ICD results from exposure to irritants such as fibers, floating dusts, solvents, and sprays [69] dispersed and carried in the air before coming into contact with exposed skin [64]. Most cases are reported in occupational settings [70].
Diagnosis
ICD is a diagnosis of exclusion, as there is no routine diagnostic test. The diagnosis is established from the history, clinical examination, and by excluding ACD with negative patch testing. A thorough history regarding occupational and domestic exposures is crucial, and relevant information includes the frequency, intensity, and duration of exposure to skin irritants, as well as the affected skin area(s) [44].
The pattern and distribution of dermatitis plays a key role in the diagnosis. ICD always starts at the site of skin contact with the irritant and generally does not spread. Common sites include the hands, face, and perineal areas, particularly in infants and adults experiencing incontinence.
Patch testing is recommended to exclude the differential diagnosis of ACD, which may be clinically indistinguishable from ICD. Even if the results are negative, patch testing has been shown to have a positive impact on quality of life [71].
Bioengineering techniques, such as the measurement of TEWL, provide a non-invasive method to measure skin irritation in ICD; however, these are not always available in a clinical setting.
The role of skin biopsy remains limited in the diagnosis of ICD, given that histopathological changes vary between irritants and relates to their mode of action and concentration [72]. Histological findings in chronic ICD are generally similar to those of ACD. Reflectance-mode confocal microscopy (RCM) is a non-invasive novel technique which can be used to differentiate between acute ICD and ACD in experimental settings, with superficial epidermal changes (SC disruption, parakeratosis, and separation of individual corneocytes) being more prominent in ICD compared to ACD [73, 74]. RCM is postulated to be more sensitive and specific than clinical examination during patch testing and could potentially play a role in distinguishing between doubtful-positive and negative reactions [75]. RCM allows repeated observation of the affected area in real time, which is non-invasive compared to conventional histology [76].
Prevention
Approaches to preventing exposure to irritants in the workplace involve standard occupational hygiene principles including elimination and/or substitution of the irritant, isolation, and engineering controls including changing the way a job is performed, administrative controls, and finally use of personal protective equipment (PPE) [77].
Recent studies have highlighted the importance of pre-employment screening tools in the workplace in early identification of hand dermatitis [78].
The use of PPE is recommended in the prevention of ICD. The choice of PPE is guided by the nature of the irritants, the areas of skin exposed, the chemical and physical properties of PPE, and its functionality in relation to the occupation [79].
Gloves are a frequently used form of PPE, given that ICD usually affects the hands. Selection of appropriate gloves for the management of ICD may be challenging, given that the protective capabilities of gloves are dependent on many variables.
Gloves should be selected based on the specific task and associated chemical exposures. The nature of the irritant as well as glove permeation time will determine the necessary type of glove material [80]. For example, neoprene gloves have been shown to be the most protective against acrylate monomers compared with polythene polymer gloves [81]. Occasionally, components of protective gloves can cause sensitization and ACD; common culprits include thiurams and carbamates found in both latex and nitrile gloves, but are absent from those made of polyvinyl chloride [82].
Glove thickness plays a major role in chemical permeation. However, thin gloves provide greater user comfort and dexterity than thicker gloves made from the same material [83].
Even when a suitable glove has been chosen, if the user does not don or remove the glove appropriately, skin contamination can occur. Incorrect glove sizing can increase the rate of glove perforation [84]. It is important that gloves are changed regularly, as sweating may exacerbate existing dermatitis [85]. Additionally, there is evidence that occlusion from gloves may impair the function of the skin barrier [86].
Training in glove choice and use has been shown to reduce user error and allergen exposure [87, 88]; however, studies have shown that workplace glove education remains limited [89, 90].
Given that half of the cases of occupational contact dermatitis (OCD) have been observed to appear within the first 2 years of employment [91], there is a role for educational strategies to promote awareness of potential irritants, appropriate use of PPE, and recognition of the early signs of OCD, specifically ICD. Interdigital dermatitis, also referred to as the “sentinel” sign, is regarded as an early stage of hand ICD in occupations involving wet work [92]. The impact of work-related educational programs has been shown to be effective in Danish healthcare workers [93], student nurses [94], and hairdressers [95].
A soap-free cleanser is preferable, as synthetic detergents are less irritating to the skin because of the neutral or slightly acidic pH [96]. The relatively high-free fatty acid content of soap free cleansers also provides a moisturizing effect, preventing hand irritation and dryness. The use of ABHS including moisturizers avoiding common allergens is recommended as an alternative to detergents [97].
Barrier creams (BCs) are designed to prevent penetration of irritants into the skin. They are thought to play a role in the prevention of ICD but are only recommended for low grade irritants [98]. A recent Cochrane review found that use of barrier creams alone may have a slight protective effect, but the evidence was deemed low quality and not clinically significant [99••]. Application methods can impact the efficacy of barrier creams, with studies highlighting that BCs are often poorly applied during real-world use, especially on the dorsa of the hands and at significantly lower doses than required to prevent irritation [100]. Inappropriate application has been shown to exacerbate some skin conditions [101]. Nevertheless, BC can raise awareness of the possibility of skin problems in the workplace.
Inflammasome-targeted therapies such as topical disulfiram have been shown to be effective in inhibiting ICD in a recent study on human subjects [102]. This is thought to be a result of the reduction in the inflammatory cytokine IL-18. This advancement provides a novel approach in the prevention of ICD.
Management
The primary treatment in ICD is avoidance, skin protection from the offending irritant(s) both in the workplace and at home [103], and use of topical therapy, particularly moisturizing creams.
Moisturizers are commonly used to improve dry skin symptoms and maintain healthy skin [104]. There is increasing evidence of their role in the treatment of ICD by preventing the absorption of exogenous substances and improving skin barrier recovery [105]. It is thought that using moisturizers increases skin hydration and that their lipid components modify endogenous epidermal lipids, with high lipid content moisturizers significantly preventing ICD when compared to formulations with lower lipid content [104]. Patients should be advised to apply moisturizers frequently, particularly before and after shifts, and after handwashing [106]. Evidence has also shown the protective role that moisturizers play in the long and short term, in the primary prevention of occupational ICD [99••, 107].
Strontium salts have been shown to be effective in treating sensory irritation and are thought to act by selectively blocking the activation of cutaneous type C nociceptors [108]. However, this treatment is not commonly used worldwide.
Cool compresses are a primary treatment of acute ICD by providing an environment which reduces the inflammation and surface temperature changes associated with acute ICD [109, 110].
Complications of ICD include bacterial superinfections. These are usually treated aggressively with antibiotics to prevent the development of cellulitis.
Although histamine is not involved in the mechanism of ICD, in clinical practice, oral antihistamines are often prescribed for symptomatic relief. Studies on mice have shown the potential role of topical antihistamines in ICD by reducing inflammation and enhancing barrier function [111]. However, to date, there have been no randomized clinical trials demonstrating the efficacy of antihistamines in ICD [111].
Despite the frequent use of topical corticosteroids in ICD, their use remains controversial. In humans, studies of clinical efficacy are inconsistent [112] with some suggesting that they may reduce barrier function associated with inhibition of lipid synthesis in the epidermis [113]. There may be a role for topical corticosteroids in chronic hyperkeratotic irritant dermatitis [56], but prolonged use can result in epidermal atrophy and therefore increase irritant sensitivity. However, systemic corticosteroids may be required during severe acute phases of ICD [114].
The Osnabrueck tertiary intervention program (TIP) is a multidisciplinary approach intended to treat severe recalcitrant OSD, comprising a 3-week inpatient phase followed by a 3 week no-exposure outpatient phase. Therapy aims to be free of corticosteroids in order to promote long-term stabilization of the epidermal barrier. TIP intervention has been shown to significantly reduce the use of corticosteroids [115, 116, 117••]. Management also includes intensive patient education, health-psychological intervention, and specialized employment consultants [118]. Follow-up studies have shown a significant reduction in hand eczema severity and days of absence from work as well as improvement in quality of life both in short-term [115] and long-term follow up [116, 117••].
Topical calcineurin inhibitors (TCIs) are topical immunomodulators that provide a safe alternative to corticosteroids. An association of topical TCIs with skin cancer has been suggested, but there is no strong evidence [119], while there may be an association with risk of lymphoma [120]. TCIs have been shown to be favorable in the treatment of ICD [121, 122]. Other systemic treatments include alitretinoin, which has been shown to be an effective treatment in study of patients with chronic hand eczema, 43.2% of whom were diagnosed with ICD [123].
Oral immunomodulators may be required in the treatment of chronic ICD if other first- or second-line treatments fail. Cyclosporine is reported to be beneficial in the treatment of chronic hand eczema [114] but is used with caution given its associated side effects.
Limited evidence has highlighted the use of dupilumab, a monoclonal antibody treatment approved for the treatment of atopic dermatitis, to be effective in the treatment of chronic recalcitrant hyperkeratotic ICD [124] and non-atopic hyperkeratotic hand eczema [125]. Further studies are required to determine the use of dupilumab as a potential therapeutic agent in ICD.
Phototherapy has proven to be beneficial in the treatment of cumulative ICD, where repeated low levels of UV exposure upregulate skin barrier function by reducing epidermal proliferation [126]. Grenz–ray therapy is an alternative which may induce an extended response by suppressing Langerhans cells [127, 128].
Prognosis
Severe ICD is associated with a poor prognosis despite advancements in prevention and treatment strategies. A worse prognosis is linked to history of atopy, female sex, and delayed diagnosis [44]. Prognosis can be improved with early detection, patch testing to clarify the diagnosis, and exclude ACD and educational interventions to increase knowledge of ICD in affected individuals [129].
Conclusion
ICD is a complex disease influenced by endogenous and exogenous factors. ICD remains a diagnosis of exclusion, and currently, there is no diagnostic test available. For many clinicians, the diagnosis may be difficult, especially if patch testing is not available. Delays in diagnosing ICD are associated with a poorer prognosis. Advancements into RCM as a diagnostic approach appear to be promising. The mainstay of treatment in ICD remains avoidance of skin irritants and the use of PPE, with moisturizers to maintain skin barrier function. In cases of occupationally acquired ICD, the role of health promotion and preventative strategies has been shown to be beneficial, but are often poorly implemented in workplaces. Further studies are required to more fully evaluate predisposing factors, as well as informing clinicians of therapeutic approaches to better manage ICD.
Abbreviations
- ACD:
-
Allergic contact dermatitis
- ICD:
-
Irritant contact dermatitis
- CLA:
-
Cutaneous lymphocyte association
- OSD:
-
Occupational skin disease
- PPE:
-
Personal protective equipment
- SC:
-
Stratum corneum
- TEWL:
-
Trans epidermal water loss
- TNF:
-
Tumor necrosis factor
References
Papers of particular interest, published recently, have been highlighted as: • Of importance •• Of major importance
Novak-Bilic G, Vucic M, Japundzic I, Mestrovic-Stefekov J, Stanic-Duktaj S, Lugovic-Mihic L. Irritant and allergic contact dermatitis - skin lesion characteristics. Acta Clin Croat. 2018;57(4):713–20. https://doi.org/10.20471/acc.2018.57.04.13.
•• Cahill JL, Williams JD, Matheson MC, Palmer AM, Burgess JA, Dharmage SC et al. Occupational skin disease in Victoria, Australia. Australas J Dermatol. 2016;57(2):108-14. https://doi.org/10.1111/ajd.12375. Large retrospective review evaluating risk factors of patients presenting with occupational skin disease.
DeKoven JG, DeKoven BM, Warshaw EM, Mathias CGT, Taylor JS, Sasseville D et al. Occupational contact dermatitis: retrospective analysis of North American Contact Dermatitis Group Data, 2001 to 2016. J Am Acad Dermatol. 2021. https://doi.org/10.1016/j.jaad.2021.03.042.
Zhai H, Maibach H. Skin occlusion and irritant and allergic contact dermatitis: an overview. Contact Dermatitis. 2001:201–6.
Marks J, Elsner P, Deleo V. Allergic and irritant contact dermatitis. Contact and Occupational Dermatology. St Louis: Mosby; 2002. p. 3–12.
Dickel H, Kuss O, Schmidt A, Kretz J, Deipgen T. Importance of irritanct dermatitis in occupational skin disease. Am J Clin Dermatol. 2002;3:283–9.
Slodwnik D, Lee A, Nixon R. Irritant Contact Dermatitis: a review. Australas J Dermatol. 2008;49:1–11.
Menne T, Johansen J, Sommerlund M, Veien N. Hand eczema guidelines based on the Danish guidelines for the diagnosis and treatment of hand eczema. Contact Dermatitis. 2011;65(1):3–12.
• Lund T, Flachs E, Sorenson J, Ebbenhoj N, Bonde J, Agner T. A job-exposure matrix addressing hand exposure to wet work. Int Arch Occup Environ Health. 2019;92:959–66. Established framework to assess the extent of wet work exposure in different occupations.
Federal Institue for Occupational Safety and Health. Technical rules for hazardous substances: risks resulting from skin contact - identification, assessment, measures (TRGS 401). 2008.
Reinholz M, Kendziora B, Frey S, Oppel EM, Rueff F, Clanner-Engelshofen BM, et al. Increased prevalence of irritant hand eczema in health care workers in a dermatological clinic due to increased hygiene measures during the SARS-CoV-2 pandemic. Eur J Dermatol. 2021;31(3):392–5. https://doi.org/10.1684/ejd.2021.4046.
Borch L, Thorsteinsson K, Warner TC, Mikkelsen CS, Bjerring P, Lundbye-Christensen S, et al. COVID-19 reopening causes high risk of irritant contact dermatitis in children. Dan Med J. 2020;67(9).
Cristaudo A, Pigliacelli F, Pacifico A, Damiani G, Iacovelli P, Morrone A. Teledermatology and hygiene practices during the COVID-19 pandemic. Contact Dermatitis. 2020;83(6):536. https://doi.org/10.1111/cod.13683.
Fartasch M, Schnetz E, Diepgen T. Characterisation of detergent induced barrier alterations - effect of barrier creas on irritation. J Investig Dermatol Symp Proc. 1998;3:121–7.
Grubauer G, Elias P, Feingold K. Transepidermal water loss: the signal for recovery of barrier and function. J Lipid Res. 1989;30:323–33.
Aramaki J, Loffler C, Kawana S, Effendy I, Happle R, Loffler H. Irritant patch testing with sodium lauryl sulphate: interrelation between concentration and exposure times. Br J Dermatol. 2001;145:704–8.
Heinemann C, Paschold C, Fluhr J, Wigger-Alberti W, Schliemann-Willer S, Farwanah H, et al. Induction of a hardening phenomenon by repeated application of SLS. Analysis of lipid changes in the stratum corneum. Acta Dermato-Venereologica. 2005;85:290–5.
Hu C. Sweat-related dermatoses:old concept and new scenario. Dermatologica. 1991;182:73–6.
Fluhr J, Bornkessel A, Akengin A, Fuchs S, Norgauer J, Kleesz P, et al. Sequential application of cold and sodium lauryl sulphate decreases irritation and barrier disruption in vivo in humans. Br J Dermatol. 2005;152(4).
Zhai H, Meier-Davis S, Cayme B, Shudo J, Maibach H. Irritant contact dermatitis: effect of age. Cutan Ocul Toxicol. 2012;31(2):138–43.
Cua A, Wilhelm K, Maibach H. Cutaneous SLS irritation potential: age and regional variability. Br J Dermatol. 1990;123:607–13.
Meding B. Differences between the sexes with regard to work-related skin disease. Contact Dermatitis. 2000;43(2):65–71.
Robinson M. Population differences in acute skin irritation responses. Race,sex,age,sensitive skin,and repeat subject comparisons. Contact Dermatitits. 2002;46:86–93.
Berardesca E, Maibach H. Racial difference in sodium lauryl sulphate induced cutaneous irritation: black and white. Contact Dermatitis. 1988;18:65–70.
Anderson K, Maibach H. Black and white human skin differences. J Am Acad Dermatol. 1976;1:276–82.
Lee E, Kim S, Lee J, Cho SA, Shin K. Ethnic differences in objective and subjective skin irritation response: an international study. Skin Res Technol. 2014;20(3):265–9. https://doi.org/10.1111/srt.12111.
Meding B, Wrangsjo K, Jarvholm B. Fifteen-year follow-up of hand eczema: predictive factors. J Invest Dermatol. 2005;124(5):893–7. https://doi.org/10.1111/j.0022-202X.2005.23723.x.
Conti A, Di Nardo A, Seidenary S. No alteration of biophysical parameteres in the skin of subjects with respiratory atopy. Dermatology. 1996;192:317–20.
Gittler J, Kreuger J, Guttman-Yassky E. Atopic dermatits results in intrinsic barrier and immune abnormalities: implications for contact dermatitis. J Allergy Clin Immunol. 2012;131:300–13.
Kezic S, O’regan G, Lutter R, Jakasa I, Koster E, Saunders S, et al. Filaggrin loss-of-function mutations are associated with enhanced expression of IL-1 cytokines in the stratum corneum of patients with atopic dermatitis and in a murine model of filaggrin deficiency. J Allergy Clin Immunol. 2012;129:1031–9.
de Jongh C, Khrenova L, Verberk M, Calkoen F, van Dijk F, Voss H, et al. Loss-of-function polymorphisms in the filaggrin gene are associated with an increased susceptibility to chronic irritant contact dermatitis: a case-control study. Br J Dermatol. 2008;159:621–7.
Landeck L, Visser M, Skudlik C, Brans R, Kezic S, John S. Clinical course of occupational irritant contact dermatitis of the hands in relation to the filaggrin genotype status and atopy. Br J Dermatol. 2012;167:1302–9.
Yucesoy B, Talzhanov Y, Barmada MM, Johnson VJ, Kashon ML, Baron E, et al. Genetic basis of irritant susceptibility in health care workers. J Occup Environ Med. 2016;58(8):753–9. https://doi.org/10.1097/JOM.0000000000000784.
Yucesoy B, Talzhanov Y, Michael Barmada M, Johnson VJ, Kashon ML, Baron E, et al. Association of MHC region SNPs with irritant susceptibility in healthcare workers. J Immunotoxicol. 2016;13(5):738–44. https://doi.org/10.3109/1547691X.2016.1173135.
de Jongh CM, John SM, Bruynzeel DP, Calkoen F, van Dijk FJ, Khrenova L, et al. Cytokine gene polymorphisms and susceptibility to chronic irritant contact dermatitis. Contact Dermatitis. 2008;58(5):269–77. https://doi.org/10.1111/j.1600-0536.2008.01317.x.
Babic Z, Kezic S, Macan J. Individual susceptibility to contact sensitization: the role of TNFalpha 308G>A polymorphism and atopy. Eur J Dermatol. 2019;29(1):75–80. https://doi.org/10.1684/ejd.2018.3485.
Wang BJ, Shiao JS, Chen CJ, Lee YC, Guo YL. Tumour necrotizing factor-alpha promoter and GST-T1 genotype predict skin allergy to chromate in cement workers in Taiwan. Contact Dermatitis. 2007;57(5):309–15. https://doi.org/10.1111/j.1600-0536.2007.01242.x.
Landeck L, Visser M, Kezic S, John SM. Impact of tumour necrosis factor-alpha polymorphisms on irritant contact dermatitis. Contact Dermatitis. 2012;66(4):221–7. https://doi.org/10.1111/j.1600-0536.2011.02045.x.
Hohler T, Kruger A, Gerken G, Schneider PM, zum Buschenefelde MKH, Rittner C. A tumor necrosis factor-alpha (TNF-alpha) promoter polymorphism is associated with chronic hepatitis B infection. Clin Exp Immunol. 1998;111(3):579–82. https://doi.org/10.1046/j.1365-2249.1998.00534.x.
Huizinga TW, Westendorp RG, Bollen EL, Keijsers V, Brinkman BM, Langermans JA, et al. TNF-alpha promoter polymorphisms, production and susceptibility to multiple sclerosis in different groups of patients. J Neuroimmunol. 1997;72(2):149–53. https://doi.org/10.1016/s0165-5728(96)00182-8.
Levin C, Maibach H. Irritant contact dermatitis: is there an immunological component? Internation Immunopharmacology. 2002;2:183–9.
Angelova-Fischer I. Irritants and skin barrier function. Curr Probl Dermatol. 2016;49:80–9.
Welss T, Basketter DA, Schroder KR. In vitro skin irritation: facts and future. State of the art review of mechanisms and models. Toxicol In Vitro. 2004;18(3):231–43. https://doi.org/10.1016/j.tiv.2003.09.009.
Bains S, Nash P, Fonacier L. Irritant contact dermatitis. Clin Rev Allergy Immunol. 2019;56:99–109.
Gittler JK, Krueger JG, Guttman-Yassky E. Atopic dermatitis results in intrinsic barrier and immune abnormalities: implications for contact dermatitis. J Allergy Clin Immunol. 2013;131(2):300–13. https://doi.org/10.1016/j.jaci.2012.06.048.
Spiekstra SW, Toebak MJ, Sampat-Sardjoepersad S, van Beek PJ, Boorsma DM, Stoof TJ, et al. Induction of cytokine (interleukin-1alpha and tumor necrosis factor-alpha) and chemokine (CCL20, CCL27, and CXCL8) alarm signals after allergen and irritant exposure. Exp Dermatol. 2005;14(2):109–16. https://doi.org/10.1111/j.0906-6705.2005.00226.x.
Feingold K, Schmuth M, Elias P. The regulation of permeability barrier homeostasis. J Investig Dermatol. 2007;127(7):1547–76.
Corsini E, Galli C. Epidermal cytokines in experimental contact dermatitis. Toxicology. 2000;142(3):203–11.
Lee H, Stieger M, Yawalkar N, Kakeda M. Cytokines and chemokines in irritant contact dermatitis. Mediators of Inflammation. 2013.
Eberhand Y, Ortiz S, Ruiz L, A, Kuznitzky R, Serra H. Up-regulation of the chemokine CCL21 in the skin of subjects exposed to irritants. BMC Immunology. 2004;26:7.
Soler D, Humphreys TL, Spinola SM, Campbell JJ. CCR4 versus CCR10 in human cutaneous TH lymphocyte trafficking. Blood. 2003;101(5):1677–82. https://doi.org/10.1182/blood-2002-07-2348.
Ale I, Maibach H. Irritant contact dermatitis. Rev Environ Health. 2014;29:195–206.
Nakai K, Yoneda K, Kubota Y. Oxidative stress in allergica and irritant dermatitis: from basic reseach to clinical management. Recent Pat Inflammation Allergy Drug Discovery. 2012;6(3):202–9.
Wagemaker TAL, Maia Campos P, Shimizu K, Kyotani D, Yoshida D. Antioxidant-based topical formulations influence on the inflammatory response of Japanese skin: a clinical study using non-invasive techniques. Eur J Pharm Biopharm. 2017;117:195–202. https://doi.org/10.1016/j.ejpb.2017.03.025.
Weltfriend S, Ramon N, Maibach H. Irritant dermatitis. Dermatotoxicology. Boca Raton: CRC Press; 2004. p. 181–228.
Antonov D, Schliemann S, Elsner P. Contact dermatitis due to Irritation. In: 3, editor. Kanerva's Occupational Dermatology. Switzerland: Springer Nature; 2020. p. 119–37.
Elsner P. Irritant dermatitis in the workplace. Dermatol Clin. 1994;12(3):461–7.
Malten K, den Arend J, Wiggers R. Delayed irritation: hexanediol diacrylate and butanediol diacrylate. Contact Dermatitis. 1979;5(3):178–84.
Frosch P, John S. Clinical aspects of irritant contact dermatitis. Contact dermatitis. Berlin: Springer; 2006. p. 255–94.
Wigger-Alberti W, Elsner P, Wahlberg J, Maibach H. Irritant dermatitis (irritation). Handbook of Occupational Dermatology. New York: Springer; 2000. p. 99–110.
Lee E, An S, Lee T, Kim H. Development of a novel method for quantitative evaluation of sensory skin irritation inhibitors. Skin Research and Technology. 2009;15(4):464–9.
Charbonnier VJ, Morrison B, Paye M, Maibach M. Subclinical, non erythematous irritation with an open assay model (washing): sodium lauryl sulfate (SLES). Food Chem Toxicol. 2001;39:279–86.
Malten K. Thoughts on irritant contact dermatitis. Contact Dermatitis. 1981;7(5):238–47.
Chew A-L, Maibach H. Ten genotypes of irritant contact dermatitis. Irritant dermatitis. Berlin: Springer; 2006. p. 5–9.
Pedersen LK, Johansen JD, Held E, Agner T. Augmentation of skin response by exposure to a combination of allergens and irritants - a review. Contact Dermatitis. 2004;50(5):265–73. https://doi.org/10.1111/j.0105-1873.2004.00342.x.
McMullen E, Gawkrodger DJ. Physical friction is under-recognized as an irritant that can cause or contribute to contact dermatitis. Br J Dermatol. 2006;154(1):154–6. https://doi.org/10.1111/j.1365-2133.2005.06957.x.
Darr-Foit S, Tittelnbach J, Elsner P. Postraumatic irritant contact dermatitis - an underdiagnosed entity? Deutsch Dermatolgische Gesellschaft. 2014;12(4).
Diepgen TL, Andersen KE, Brandao FM, Bruze M, Bruynzeel DP, Frosch P, et al. Hand eczema classification: a cross-sectional, multicentre study of the aetiology and morphology of hand eczema. Br J Dermatol. 2009;160(2):353–8. https://doi.org/10.1111/j.1365-2133.2008.08907.x.
Lachapelle J. Enviromental airborne contact dermatoses. Reviews on Enviromental Health. 2014;29:2210231.
Santos R, Goossens A. An update on airborne contact dermatitis. Contact Dermatitis. 2007:353–60.
Woo P, Hay I, Ormerod A. An audit of the value of patch testing and its effect on quality of life. Contact Dermatitis. 2003;48:244–7.
Weedon D. Weedon's skin pathology. Churchill Livingstone; 2009.
Swindells K, Burnett N, Rius-Diaz F, Gonzalez E, Mihm MC, Gonzalez S. Reflectance confocal microscopy may differentiate acute allergic and irritant contact dermatitis in vivo. J Am Acad Dermatol. 2004;50(2):220–8. https://doi.org/10.1016/j.jaad.2003.08.005.
Astner S, Gonzalez S, Gonzalez E. Noninvasive evaluation of allergic and irritant contact dermatitis by in vivo reflectance confocal microscopy. Dermatitis. 2006;17(4):182–91. https://doi.org/10.2310/6620.2006.05052.
Maarouf M, Costello CM, Gonzalez S, Angulo I, Curiel-Lewandrowski CN, Shi VY. In vivo reflectance confocal microscopy: emerging role in noninvasive diagnosis and monitoring of eczematous dermatoses. Actas Dermosifiliogr. 2019;110(8):626–36. https://doi.org/10.1016/j.ad.2018.08.008.
Suarez-Perez J, Bosch R, Gonzalez S, Gonzalez E. Pathogenesis and diagnosis of contact dermatitis: application of reflectance confocal microscopy. World Journal of Dermatology. 2014;2.
The National Institute for Occupational Safety and Health (NIOSH). Hierachy of Controls. https://www.cdc.gov/niosh/topics/hierarchy/. Accessed April 2021.
Nichol K, Copes R, Spielmann S, Kersey K, Eriksson J, Holness DL. Workplace screening for hand dermatitis: a pilot study. Occup Med (Lond). 2016;66(1):46–9. https://doi.org/10.1093/occmed/kqv126.
Garrigoua A, Laurent C, Berthet A, Colosiod C, Jase N, Daubas-Letourneux V, JacksonFilhog JM, Jouzelh J-N, Samuel O, Baldia I, Lebailly P, Galey L, Goutille F, Judona N. Critical review of the role of PPE in the prevention of risks related to agricultural pesticide use. Safety Science. 2020;123.
Johnston GA, Exton LS, Mohd Mustapa MF, Slack JA, Coulson IH, English JS, et al. British Association of Dermatologists’ guidelines for the management of contact dermatitis 2017. Br J Dermatol. 2017;176(2):317–29. https://doi.org/10.1111/bjd.15239.
Andersson T, Bruze M, Gruvberger B, Bjorkner B. In vivo testing of the protection provided by non-latex gloves against a 2-hydroxyethyl methacrylate-containing acetone-based dentin-bonding product. Acta Derm Venereol. 2000;80(6):435–7. https://doi.org/10.1080/000155500300012891.
Kersh AE, Helms S, de la Feld S. Glove-related allergic contact dermatitis. Dermatitis. 2018;29(1):13–21. https://doi.org/10.1097/DER.0000000000000335.
Ramadan MZ. The effects of industrial protective gloves and hand skin temperatures on hand grip strength and discomfort rating. Int J Environ Res Public Health. 2017;14(12). https://doi.org/10.3390/ijerph14121506.
Zare A, Choobineh A, Jahangiri M, Seif M, Dehghani F. Does size affect the rate of perforation? A cross-sectional study of medical gloves. Ann Work Expo Health. 2021. https://doi.org/10.1093/annweh/wxab007.
Bourke K, Coulson I, English J. Guidelines for the management of contact dermatitis: an update. Br J Dermatol. 2009;160:946–54.
Antonov D, Kleesz P, Elsner P, Schliemann S. Impact of glove occlusion on cumulative skin irritation with or without hand cleanser-comparison in an experimental repeated irritation model. Contact Dermatitis. 2013;68(5):293–9. https://doi.org/10.1111/cod.12028.
Oreskov KW, Sosted H, Johansen JD. Glove use among hairdressers: difficulties in the correct use of gloves among hairdressers and the effect of education. Contact Dermatitis. 2015;72(6):362–6. https://doi.org/10.1111/cod.12336.
Geens T, Aerts E, Borguet M, Haufroid V, Godderis L. Exposure of hairdressers to aromatic diamines: an interventional study confirming the protective effect of adequate glove use. Occup Environ Med. 2016;73(4):221–8. https://doi.org/10.1136/oemed-2014-102708.
Keegel TG, MacFarlane E, Nixon R, Lamontagne A. Provision of control measures for exposure of the hands to wet-working conditions in Australian workplaces. Int J Occup Environ Health. 2012;18(4):312–9. https://doi.org/10.1179/2049396712Y.0000000010.
Rowley K, Ajami D, Gervais D, Mooney L, Belote A, Kudla I, et al. Glove use and glove education in workers with hand dermatitis. Dermatitis. 2016;27(1):30–2. https://doi.org/10.1097/DER.0000000000000155.
Dickel H, Kuss O, Schmidt A, Diepgen T. Occupational skin diseases in Northern Bavaria between 1990 and 1999: a population-based study. Br J Dermatol. 2008;145(3):453–62.
Schwanitz HJ, Uter W. Interdigital dermatitis: sentinel skin damage in hairdressers. Br J Dermatol. 2000;142(5):1011–2. https://doi.org/10.1046/j.1365-2133.2000.03487.x.
Clemmensen KK, Randboll I, Ryborg MF, Ebbehoj NE, Agner T. Evidence-based training as primary prevention of hand eczema in a population of hospital cleaning workers. Contact Dermatitis. 2015;72(1):47–54. https://doi.org/10.1111/cod.12304.
Held E, Wolff C, Gyntelberg F, Agner T. Prevention of work-related skin problems in student auxiliary nurses: an intervention study. Contact Dermatitis. 2001;44(5):297–303. https://doi.org/10.1034/j.1600-0536.2001.440509.x.
Bregnhoj A, Menne T, Johansen JD, Sosted H. Prevention of hand eczema among Danish hairdressing apprentices: an intervention study. Occup Environ Med. 2012;69(5):310–6. https://doi.org/10.1136/oemed-2011-100294.
Abtahi-Naeini B. Frequent handwashing amidst the COVID-19 outbreak: prevention of hand irritant contact dermatitis and other considerations. Health Sci Rep. 2020;3(2): e163. https://doi.org/10.1002/hsr2.163.
Rundle CW, Presley CL, Militello M, Barber C, Powell DL, Jacob SE, et al. Hand hygiene during COVID-19: Recommendations from the American Contact Dermatitis Society. J Am Acad Dermatol. 2020;83(6):1730–7. https://doi.org/10.1016/j.jaad.2020.07.057.
Mostosi C, Simonart T. Effectiveness of barrier creams against irritant contact dermatitis. Dermatology. 2016;232(3):353–62. https://doi.org/10.1159/000444219.
•• Bauer A, Ronsch H, Elsner P, Dittmar D, Bennett C, Schuttelaar MLA, et al. Interventions for preventing occupational irritant hand dermatitis. Cochrane Database Syst Rev. 2018;4:CD004414. https://doi.org/10.1002/14651858.CD004414.pub3. Large systematic review of interventions in preventing occupational irritant hand dermatitis. It showed the protective role of moisturizers and educational strategies in preventing hand dermatitis.
Schliemann S, Petri M, Elsner P. Preventing irritant contact dermatitis with protective creams: influence of the application dose. Contact Dermatitis. 2014;70(1):19–26. https://doi.org/10.1111/cod.12104.
Alvarez MS, Brown LH, Brancaccio RR. Are barrier creams actually effective? Curr Allergy Asthma Rep. 2001;1(4):337–41. https://doi.org/10.1007/s11882-001-0045-z.
Bonnekoh H, Vera C, Abad-Perez A, Radetzki S, Neuenschwander M, Specker E, et al. Topical inflammasome inhibition with disulfiram prevents irritant contact dermatitis. Clin Transl Allergy. 2021;11(5): e12045. https://doi.org/10.1002/clt2.12045.
Diepgen T, Anderson K, Brandao F, Bruze M, Bruynzeel D, Goncalo M, et al. Hand eczema classification:a cross-sectional multicentre study of the aetiology and morphology of hand eczema. Br J Dermatol. 2009;160(2):353–8.
Held E, Agner T. Effect of moisturizers on skin susceptibility to irritants. Acta Derm Venereol. 2001;81(2):104–7. https://doi.org/10.1080/00015550152384227.
Purnamawati S, Indrastuti N, Danarti R, Saefudin T. The role of moisturizers in addressing various kinds of dermatitis: a review. Clin Med Res. 2017;15(3–4):75–87. https://doi.org/10.3121/cmr.2017.1363.
Hines J, Wilkinson SM, John SM, Diepgen TL, English J, Rustemeyer T, et al. The three moments of skin cream application: an evidence-based proposal for use of skin creams in the prevention of irritant contact dermatitis in the workplace. J Eur Acad Dermatol Venereol. 2017;31(1):53–64. https://doi.org/10.1111/jdv.13851.
Saary J, Qureshi R, Palda V, DeKoven J, Pratt M, Skotnicki-Grant S, et al. A systematic review of contact dermatitis treatment and prevention. J Am Acad Dermatol. 2005;53(5):845. https://doi.org/10.1016/j.jaad.2005.04.075.
Zhai H, Hannon W, Hahn GS, Pelosi A, Harper RA, Maibach HI. Strontium nitrate suppresses chemically-induced sensory irritation in humans. Contact Dermatitis. 2000;42(2):98–100. https://doi.org/10.1034/j.1600-0536.2000.042002098.x.
Levin C, Maibach H. Do cool water or physiological saline compresses enhance resolution of experimentally-induced irritant contact dermatitis? Contact Dermatitis. 2001;45:146–50.
Yoshizawa Y, Tanojo H, Kim S, Maibach H. Sea water or its components alter experimental irritant contact dermatitis in man. Skin Res Technol. 2001;7:36–9.
Lin T, Man M, Santiago J, Park K, Roelandt T, Oda Y, et al. Topical antihistamines display potent anti-inflammatory activity linked in part to enhanced permeability barrier function. J Investig Dermatol. 2013;133:469–78.
Azizi N, Maibach HI. Are topical corticoids efficacious in acute irritant dermatitis: the evidence. Dermatitis. 2020;31(4):244–6. https://doi.org/10.1097/DER.0000000000000574.
Kao J, JW F, Man MQ F, AJ Hachem, JP, Crumrine D, Ahn S, BE B, et al. Short-term glucocorticoid treatment compromises both permeability barrier homeostasis and stratum corneum intergrity: inhibition of epidermal lipid synthesis accounts for functional abnormalities. J Investig Dermatol. 2003;120:456–64.
Elsner P, Agner T. Hand eczema: a “neglected” disease. J Eur Acad Dermatol Venereol. 2020;34(Suppl 1):3. https://doi.org/10.1111/jdv.16081.
Skudlik C, Weisshaar E, Scheidt R, Elsner P, Wulfhorst B, Schonfeld M, et al. First results from the multicentre study rehabilitation of occupational skin diseases–optimization and quality assurance of inpatient management (ROQ). Contact Dermatitis. 2012;66(3):140–7. https://doi.org/10.1111/j.1600-0536.2011.01991.x.
Weisshaar E, Skudlik C, Scheidt R, Matterne U, Wulfhorst B, Schonfeld M, et al. Multicentre study ’rehabilitation of occupational skin diseases -optimization and quality assurance of inpatient management (ROQ)’-results from 12-month follow-up. Contact Dermatitis. 2013;68(3):169–74. https://doi.org/10.1111/j.1600-0536.2012.02170.x.
•• Brans R, Skudlik C, Weisshaar E, Scheidt R, Ofenloch R, Elsner P et al. Multicentre cohort study 'Rehabilitation of Occupational Skin Diseases - Optimization and Quality Assurance of Inpatient Management (ROQ)': results from a 3-year follow-up. Contact Dermatitis. 2016;75(4):205–12. https://doi.org/10.1111/cod.12614. Long term follow up multicentre cohort study assessing the impact of tertiary prevention in severe hand eczema demonstrated significant reduction in severity of hand eczema and use of topical corticosteroids and improvement in quality of life scores.
Skudlik C, Wulfhorst B, Gediga G, Bock M, Allmers H, John SM. Tertiary individual prevention of occupational skin diseases: a decade’s experience with recalcitrant occupational dermatitis. Int Arch Occup Environ Health. 2008;81(8):1059–64. https://doi.org/10.1007/s00420-008-0300-x.
Asgari MM, Tsai AL, Avalos L, Sokil M, Quesenberry CP Jr. Association between topical calcineurin inhibitor use and keratinocyte carcinoma risk among adults with atopic dermatitis. JAMA Dermatol. 2020;156(10):1066–73. https://doi.org/10.1001/jamadermatol.2020.2240.
Lam M, Zhu JW, Tadrous M, Drucker AM. Association between topical calcineurin inhibitor use and risk of cancer, including lymphoma, keratinocyte carcinoma, and melanoma: a systematic review and meta-analysis. JAMA Dermatol. 2021;157(5):549–58. https://doi.org/10.1001/jamadermatol.2021.0345.
Engel K, Reuter J, Seiler CSM, J, Jakob T, Schempp C. Anti inflammatory effect4 of pimecrolimus in the sodium laurly sulphate test. J Eur Acad Dermatol Venereol. 2007;22(4):447–50.
Grassberger.M, Steinhoff M, Schneider D, Luger T. Pimecrolimus - anti-inflammatory drug targeting the skin. Exp Dermatol. 2004;13(12):721–30.
Ferrucci S, Persichini P, Gola M, Scandagli I, Pigatto P, Legori A, et al. DECISA Project (DErmatology Clinics in Italy: Survey on Alitretinoin): a real-life retrospective cohort multicenter study on 438 subjects with chronic hand eczema. Dermatol Ther. 2021;34(3): e14911. https://doi.org/10.1111/dth.14911.
Zhu GA, Honari G, Ko JM, Chiou AS, Chen JK. Dupilumab for occupational irritant hand dermatitis in a nonatopic individual: a case report. JAAD Case Rep. 2020;6(4):296–8. https://doi.org/10.1016/j.jdcr.2020.02.010.
Loman L, Diercks GFH, Schuttelaar MLA. Three cases of non-atopic hyperkeratotic hand eczema treated with dupilumab. Contact Dermatitis. 2021;84(2):124–7. https://doi.org/10.1111/cod.13693.
English J. Current concepts of irritant contact dermatitis. Occupational Enviromental Medicine. 2004;61:722–6.
Simpson BM, Foster SK, Chapman CN, Simpson EL. The effectiveness of grenz ray therapy for chronic dermatoses of the hands and feet. Dermatitis. 2014;25(4):205–8. https://doi.org/10.1097/DER.0000000000000050.
Fenton L, Dawe RS. Six years’ experience of grenz ray therapy for the treatment of inflammatory skin conditions. Clin Exp Dermatol. 2016;41(8):864–70. https://doi.org/10.1111/ced.12960.
Cahill J, Keegel T, Nixon R. The prognosis of occupational contact dermatitis in. Contact Dermatitis. 2004;2004:219–56.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
Kajal Patel and Rosemary Nixon declare no conflicts of interest.
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article is part of the Topical Collection on Contact Dermatitis
Rights and permissions
About this article
Cite this article
Patel, K., Nixon, R. Irritant Contact Dermatitis — a Review. Curr Derm Rep 11, 41–51 (2022). https://doi.org/10.1007/s13671-021-00351-4
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13671-021-00351-4