Abstract
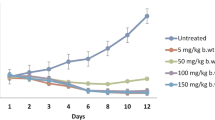
From our previous study extract of the fruit bodies of Ganoderma lucidum possessed promising curative potential against Plasmodium berghei in mice. Thus, we hypothesized that infection with chloroquine-sensitive Plasmodium berghei together with crude chloroform extract (CCE) of G. lucidum, at 250 mg/kg body weight would enhance various aspects of antiplasmodial activities in mice. Mice were treated with CCE or chloroquine (CQ) for seventy-two hours prior to infection with P. berghei and monitored closely until signs of death were observed in untreated mice and then sacrificed. Parasitaemia, hepatic function, and oxidative stress markers analyses were performed at the end of the experiment. CCE-treated mice displayed lower parasitemia compared with untreated mice. Plasma alanine aminotransferase (ALT), aspartate aminotransferase (AST) and gamma-glutamyltranspeptidase (γ-GT) activities; and the erythrocyte malondialdehyde (MDA) level were significantly (p<0.0001) lower in the extract and CQ-treated mice. Erythrocyte antioxidant enzymes (GPx, GST, SOD and G6PDH) activities were significantly (p<0.0001) higher in CCE-treated mice compared with CQ-treated and untreated control. Though chloroquine treatment reduced parasitemia and MDA considerably compared with extract, there was reduced production of the antioxidant defense enzymes. This study shows that CCE of G. lucidum reduces parasitemia and improves the attendant consequences of Plasmodium berghei-malarial infection in mice.




Similar content being viewed by others
References
Adams M, Christen M, Plitzko I, Zimmermann S, Brun R, Kaiser M, Hamburger M (2010) Antiplasmodial lanostanes from the Ganoderma lucidum mushroom. J Nat Prod 73(5):897–900
Adejumo T, Awosanya O (2005) Proximate and mineral composition of four edible mushroom species from south western Nigeria. Afr J Biotechnol 4(10):1084–1088
Binder R, Borrmann S, Adegnika A, Missinou M, Kremsner P, Kun J (2002) Polymorphisms in the parasite genes for pfcrt and pfmdr-1 as molecular markers for chloroquine resistance in Plasmodium falciparum in Lambarene, Gabon. Parasitol Res 88(5):475–476
Buege JA, Aust SD (1978) Microsomal lipid peroxidation. Methods Enzymol 52:302–310
Builders M, Wannang N, Aguiyi J (2011) Antiplasmodial activities of Parkia biglobosa leaves: In vivo and in vitro studies. Annals Bio Res 2(4):8–20
Camacho M, Croft S, Phillipson J (2000) Natural products as sources of antiprotozoal drugs. Curr Opin Anti-Infect Investig Drugs 2(1):47–62
Erel O, Kocyigit A, Avci S, Aktepe N, Bulut V (1997) Oxidative stress and antioxidative status of plasma and erythrocytes in patients with vivax malaria. Clin Biochem 30(8):631–639
Fridovich I (1989) Oxygen radicals from acetaldehyde. Free Radic Biol Med 7(5):557–558
Gornall AG, Bardawill CJ, David MM (1949) Determination of serum proteins by means of the biuret reaction. J Biol Chem 177(2):751–766
Guo F, Savelkoul H, Kwakkel R, Williams B, Verstegen M (2003) Immunoactive, medicinal properties of mushroom and herb polysaccharides and their potential use in chicken diets. Worlds Poult Sci J 59(04):427–440
Habig WH, Pabst MJ, Jakoby WB (1974) Glutathione s-transferases the first enzymatic step in mercapturic acid formation. J Biol Chem 249(22):7130–7139
Holzgrabe U, Bechthold A (1999) Paradigmenwechsel in der entwicklung antiinfektiver chemotherapeutika. Chemother J 8:69–78
Hu Y (1997) Progress in the study of immunopharmacology of chinese herbal medicine. Chin J Immunol 3:96–98
Lindequist U, Teuscher E, Narbe G (1990) Neue wirkstoffe aus basidiomyceten. Z Phytother 11:139–149
Lu H, Uesaka T, Katoh O, Kyo E, Watanabe H (2001) Prevention of the development of preneoplastic lesions, aberrant crypt foci, by a water-soluble extract from cultured medium of Ganoderma lucidum (rei-shi) mycelia in male f344 rats. Oncol Rep 8(6):1341–1345
Mbatchi S, Mbatchi B, Banzouzi J, Bansimba T, Ntandou GN, Ouamba J-M, Berry A, Benoit-Vical F (2006) In vitro antiplasmodial activity of 18 plants used in Congo Brazzaville traditional medicine. J Ethnopharmacol 104(1):168–174
Mishra S, Singh RB (2015) Biotechnological significance of mushroom of mushroom: an overview. Sci Int 3:1–6
National Research Council (1985) Guides for the care and use of laboratory animals. National Institutes of Health, Bethesda
Oei P (2003) Mushroom cultivation. Appropriate technology for mushroom growers. Backhuys Publishers, Leiden
Oluba OM, Olusola AO, Eidangbe GO, Babatola LJ, Onyeneke EC (2012a) Modulation of lipoprotein cholesterol levels in Plasmodium berghei malarial infection by crude aqueous extract of Ganoderma lucidum. Cholesterol 2012. https://doi.org/10.1155/2012/536396
Oluba OM, Olusola AO, Fagbohunka BS, Onyeneke EC (2012b) Antimalarial and hepatoprotective effects of crude ethanolic extract of lingzhi or reishi medicinal mushroom, Ganoderma lucidum (w. Curt.: Fr.) P. Karst.(higher basidiomycetes), in Plasmodium berghei-infected mice. Int J Med Mushrooms 14(5):459–466
Oluba O, Eidangbe G, Adebisi K, Odutuga A, Onyeneke E (2013) In vivo evaluation of antimalarial and cytotoxic properties of crude aqueous extract of the fruiting body of medicinal mushroom, Ganoderma lucidum (w. Curt.: Fr.) P. Karst in Plasmodium berghei-infected mice. J Immunol Tech Infect Dis 2:4. https://doi.org/10.4172/2329-9541.1000116
Omoregie ES, Pal A, Sisodia B (2011) In vitro antimalarial and cytotoxic activities of leaf extracts of Vernonia amygdalina (del.) Nig J Basic Appl Sci 9(1):121
Palmen NG, Evelo CT (1998) Oxidative effects in human erythrocytes caused by some oximes and hydroxylamine. Arch Toxicol 72(5):270–276
Philipson J, Wright C (1991) Antiprotozoal compounds from plants sources. Planta Medica 57:553–559
Qiu Y, Hu Y, Cui B, Zhang H, Kong X, Wang D, Wang Y (2007) Immunopotentiating effects of four chinese herbal polysaccharides administered at vaccination in chickens. Poultry Sci 86(12):2530–2535
Rodrigues JR, Gamboa ND (2009) Effect of dequalinium on the oxidative stress in Plasmodium berghei-infected erythrocytes. Parasitol Res 104(6):1491–1496
Rotruck J, Pope A, Ganther H, Swanson A, Hafeman DG, Hoekstra W (1973) Selenium: Biochemical role as a component of glutathione peroxidase. Science 179(4073):588–590
Shittu I, Emmanuel A, Nok AJ (2011) Antimalaria effect of the ethanolic stem bark extracts of Ficus platyphylla del. J Parasitol Res. https://doi.org/10.1155/2011/618209
Sirima SB, Konate A, Tiono AB, Convelbo N, Cousens S, Pagnoni F (2003) Early treatment of childhood fevers with pre-packaged antimalarial drugs in the home reduces severe malaria morbidity in Burkina Faso. Trop Med Int Health 8(2):133–139
Tao Y, Zhang L, Cheung PC (2006) Physicochemical properties and antitumor activities of water-soluble native and sulfated hyperbranched mushroom polysaccharides. Carbohyd Res 341(13):2261–2269
Wachtel-Galor S, Szeto Y-t, Tomlinson B, Benzie IF (2004) Ganoderma lucidum (‘lingzhi’); acute and short-term biomarker response to supplementation. Int J Food Sci Nutr 55(1):75–83
Wong KL, Chao HH, Chan P, Chang LP, Liu CF (2004) Antioxidant activity of Ganoderma lucidum in acute ethanol-induced heart toxicity. Phytother Res 18(12):1024–1026
Acknowledgments
The authors wish to acknowledge the Director, Institute for Advanced Medical Research and Training (IAMRAT), University College Hospital, Ibadan, for the gift of the malaria parasite.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethical Statement
This study was carried out in strict compliance with the Guide for the Care and Use of Laboratory Animals of the National Institutes of Health (NRC, 1985).
Conflict of Interest
Authors declare that there is no conflict of interest.
Rights and permissions
About this article
Cite this article
Oluba, O.M., Josiah, S.J., Adebisi, K.E. et al. Antiplasmodial and antioxidant activities of chloroform extract of Ganoderma lucidum fruit body in Plasmodium berghei-infected mice. Orient Pharm Exp Med 17, 389–395 (2017). https://doi.org/10.1007/s13596-017-0288-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13596-017-0288-4