Abstract
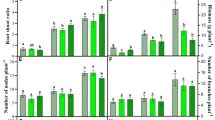
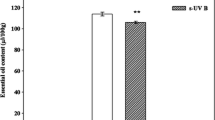
Psoralea corylifolia (bakuchi) is a traditional, medicinally important herbal plant of the family Fabaceae. In particular, seeds are vital in treating skin diseases, such as leprosy, psoriasis, and leukoderma. Global climate change and the threat of stratospheric ozone depletion are already marked, so unveiling the implications of UV-B radiation on medicinal plants is imperative. In this backdrop, the present study aimed to assess the effect of elevated UV-B (eUV-B; ambient + 7.2 kJ m−2 d−1) on the content and composition of essential oils and how other factors such as growth, anatomy and photosynthetic adaptations were involved in this. Plant growth and physiological parameters were lower under eUV-B treatment than in the control (except carotenoid and water use efficiency). Due to eUV-B exposure, the number of racemes, flowers and seeds reduced significantly compared to their numbers in the control. The essential oil content of seeds increased by 46.4% under the eUV-B treatment compared with that in the control. The GC–MS analysis of essential oil revealed that monoterpenes decreased, whereas meroterpene and sesquiterpenes increased under eUV-B treatment as compared to control. Caryophyllene, caryophyllene oxide and bakuchiol (which possesses anti-cancerous and anti-inflammatory activities) were identified as major metabolites and increased under eUV-B as compared to control. The study emphasizes that under eUV-B exposure, a reduction in growth and physiology of P. corylifolia was accompanied by an increase in essential oil content, antioxidant capacity and content of medicinally important compounds.






Similar content being viewed by others
Data availability
Not applicable.
Change history
24 July 2022
A missing space was added to the article title.
References
Abeli T, Brancaleoni L, Marchesini R, Orsenigo S, Rossi G, Gerdol R (2017) Fertiliser application positively affects plants performance but reduces seed viability in seashore mallow (Kosteletzkya pentacarpos): implication for biomass production and species conservation. Ann Appl Biol 170:263–272. https://doi.org/10.1111/aab.12339
Agrawal SB, Mishra S (2009) Effects of supplemental ultraviolet-B and cadmium on growth, antioxidants and yield of Pisum sativum L. Ecotoxicol Environ Saf 72:610–618. https://doi.org/10.1016/j.ecoenv.2007.10.007
Agrawal SB, Pandey A (2019) Pharmaceutical activities and effects of various abiotic stresses/elicitors on bioactive constituents of Psoralea corylifolia L. (Bakuchi). IJPE 5:186–191. https://doi.org/10.18811/ijpen.v5i03.6
Ali ST, Mahmooduzzafar MZ, Iqbal M (2008) Ontogenetic changes in foliar features and psoralen content of Psoralea corylifolia Linn. exposed to SO2 stress. J Environ Biol 29:661–668
Ansari N, Yadav DS, Agrawal M, Agrawal SB (2021) The impact of elevated ozone on growth, secondary metabolites, production of reactive oxygen species and antioxidant response in an anti-diabetic plant Costus pictus. Funct Plant Biol. https://doi.org/10.1071/FP20324
Ashitani T, Kusumoto N, Borg-Karlson AK, Fujita K, Takahashi K (2013) Antitermite activity of β-caryophyllene epoxide and episulfide. Zeitschrift Fur Naturforsch C 68:302–306. https://doi.org/10.1515/znc-2013-7-807
Barnes PW, Williamson CE, Lucas RM et al (2019) Ozone depletion, ultraviolet radiation, climate change and prospects for a sustainable future. Nat Sustain 2:569–579. https://doi.org/10.1038/s41893-019-0314-2
Bhat TM, Choudhary S, Ansari MYK et al (2015) Ameliorative response of some essential oil furanocoumarins and proteins from Psoralea corylifolia against gamma-irradiation induced oxidative stress. Ind Crops Prod 76:422–431. https://doi.org/10.1016/j.indcrop.2015.06.059
Bittencourt MLF, Ribeiro PR, Franco RLP et al (2015) Metabolite profiling, antioxidant and antibacterial activities of Brazilian propolis: use of correlation and multivariate analyses to identify potential bioactive compounds. Food Res Int 76:449–457. https://doi.org/10.1016/j.foodres.2015.07.008
Bornman JF, Barnes PW, Robson TM et al (2019) Linkages between stratospheric ozone, UV radiation and climate change and their implications for terrestrial ecosystems. Photochem Photobiol Sci 18:681–716. https://doi.org/10.1039/c8pp90061b
Buckle J (2015) Basic plant taxonomy, basic essential oil chemistry, extraction, biosynthesis, and analysis. Clin Aromather. https://doi.org/10.1016/B978-0-7020-5440-2.00003-6
Cane DE (1999) Sesquiterpene biosynthesis: cyclization mechanisms. In Comprehensive natural products chemistry isoprenoids: including steroids and carotenoids, pp 155–200. https://doi.org/10.1016/b978-0-08-091283-7.00039-4
Castillejo N, Martínez-Zamora L, Artés-Hernández F (2021) Periodical UV-B radiation hormesis in biosynthesis of kale sprouts nutraceuticals. Plant Physiol Biochem 165:274–285. https://doi.org/10.1016/j.plaphy.2021.05.022
Chandra M, Prakash O, Kumar R et al (2017) β-Selinene-rich essential oils from the parts of Callicarpa macrophylla and their antioxidant and pharmacological activities. Medicines 4:52. https://doi.org/10.3390/medicines4030052
Chappell J, Coates RM (2010) Sesquiterpenes. In: Mander L, Liu HW (eds) Comprehensive natrual products II. Elsevier, Kidlington, pp 609–641. https://doi.org/10.1016/B978-008045382-8.00005-8
Chen W, Viljoen AM (2010) Geraniol—a review of a commercially important fragrance material. South Afr J Bot 76:643–651. https://doi.org/10.1016/j.sajb.2010.05.008
Chen M, Huang Y, Liu G et al (2016) Effects of enhanced UV-B radiation on morphology, physiology, biomass, leaf anatomy and ultrastructure in male and female mulberry (Morus alba) saplings. Environ Exp Bot 129:85–93. https://doi.org/10.1016/j.envexpbot.2016.03.006
Choudhary KK, Agrawal SB (2014) Ultraviolet-B induced changes in morphological, physiological and biochemical parameters of two cultivars of pea (Pisum sativum L.). Ecotoxicol Environ Saf 100:178–187. https://doi.org/10.1016/j.ecoenv.2013.10.032
Chrysargyris A, Xylia P, Botsaris G, Tzortzakis N (2017) Antioxidant and antibacterial activities, mineral and essential oil composition of spearmint (Mentha spicata L.) affected by the potassium levels. Ind Crops Prod 103:202–212. https://doi.org/10.1016/j.indcrop.2017.04.010
Coffey A, Prinsen E, Jansen MAK, Conway J (2017) The UVB photoreceptor UVR8 mediates accumulation of UV-absorbing pigments, but not changes in plant morphology, under outdoor conditions. Plant Cell Environ 40:2250–2260. https://doi.org/10.1111/pce.13025
Del Valle JC, Buide ML, Whittall JB et al (2020) UV radiation increases phenolic compound protection but decreases reproduction in Silene littorea. PLoS ONE 15:e0231611. https://doi.org/10.1371/journal.pone.0231611
Ebrahimi SN, Hadian J, Mirjalili MH, Sonboli A, Yousefzadi M (2008) Essential oil composition and antibacterial activity of Thymus caramanicus at different phenological stages. Food Chem 110:927–931. https://doi.org/10.1016/j.foodchem.2008.02.083
Elisabetsky E (2002) Traditional medicines and the new paradigm of psychotropic drug action. In: Iwu MM, Wootton JC (eds) Adv phytomed: ethnomedicine and drug discovery. Elsevier, pp 133–144. https://doi.org/10.1016/S1572-557X(02)80020-4
Fernandes ES, Passos GF, Medeiros R et al (2007) Anti-inflammatory effects of compounds alpha-humulene and (-)-trans-caryophyllene isolated from the essential oil of Cordia verbenacea. Eur J Pharmacol 569:228–236. https://doi.org/10.1016/j.ejphar.2007.04.059
Fidyt K, Fiedorowicz A, Strządała L, Szumny A (2016) β-caryophyllene and β-caryophyllene oxide—natural compounds of anticancer and analgesic properties. Cancer Med 5:3007–3017. https://doi.org/10.1002/cam4.816
Gan JH, Xiong ZT, Li JP, Chen DQ (2013) Differential response to copper stress in the reproductive resources and allocation of metallophyte Kummerowia stipulacea. Ecotoxicol Environ Saf 89:204–211. https://doi.org/10.1016/j.ecoenv.2012.11.033
Gautam M, Agrawal M (2017) Influence of metals on essential oil content and composition of lemongrass (Cymbopogon citratus (D.C.) Stapf.) grown under different levels of red mud in sewage sludge amended soil. Chemosphere 175:315–322. https://doi.org/10.1016/j.chemosphere.2017.02.065
Grammatikopoulos G, Karousou R, Kokkini S, Manetas Y (1998) Differential effects of enhanced UV-B radiation on reproductive effort in two chemotypes of Mentha spicata under field conditions. Funct Plant Biol 25:345–351
Hajlaoui H, Trabelsi N, Noumi E et al (2009) Biological activities of the essential oils and methanol extract of tow cultivated mint species (Mentha longifolia and Mentha pulegium) used in the Tunisian folkloric medicine. World J Microbiol Biotechnol 25:2227–2238. https://doi.org/10.1007/s11274-009-0130-3
Hartsel JA, Eades J, Hickory B, Makriyannis A (2016) Cannabis sativa and Hemp. In: Gupta RC (ed) Nutraceuticals. Academic Press, New York, pp 735–754. https://doi.org/10.1016/B978-0-12-802147-7.00053-X
Hideg É, Jansen MAK, Strid Å (2013) UV-B exposure, ROS, and stress: inseparable companions or loosely linked associates? Trends Plant Sci 18:107–115. https://doi.org/10.1016/j.tplants.2012.09.003
Inostroza-Blancheteau C, Reyes-Díaz M, Arellano A et al (2014) Effects of UV-B radiation on anatomical characteristics, phenolic compounds and gene expression of the phenylpropanoid pathway in highbush blueberry leaves. Plant Physiol Biochem 85:85–95. https://doi.org/10.1016/j.plaphy.2014.10.015
Jaiswal D, Agrawal SB (2021) Ultraviolet-B induced changes in physiology, phenylpropanoid pathway and essential oil composition in two Curcuma species (C. caesia Roxb. and C. longa L.). Ecotoxicol Environ Saf 208:111739. https://doi.org/10.1016/j.ecoenv.2020.111739
Jaiswal D, Pandey A, Mukherjee A et al (2020) Alterations in growth, antioxidative defense and medicinally important compounds of Curcuma caesia Roxb. under elevated ultraviolet-B radiation. Environ Exp Bot 177:104152. https://doi.org/10.1016/j.envexpbot.2020.104152
Jan S, Parween T, Siddiqi TO (2011) Gamma radiation effects on growth and yield attributes of Psoralea corylifolia L. with reference to enhanced production of psoralen. Plant Growth Regul 64:163–171. https://doi.org/10.1007/s10725-010-9552-z
Jan S, Parween T, Hamid R et al (2015) Elemental, biochemical and essential oil modulation in developing seedlings of Psoralea corylifolia L. exposed to different presowing gamma irradiation treatment. J Essent Oil Res 27:521–532. https://doi.org/10.1080/10412905.2015.1024890
Jenkins GI (2017) Photomorphogenic responses to ultraviolet-B light. Plant Cell Environ 40:2544–2557. https://doi.org/10.1111/pce.12934
Kakani VG, Reddy KR, Zhao D, Mohammed AR (2003) Effects of ultraviolet-B radiation on cotton (Gossypium hirsutum L.) morphology and anatomy. Ann Bot 91:817–826. https://doi.org/10.1093/aob/mcg086
Katare DP, Nabi G, Azooz MM, Aeri V, Ahmad P (2012) Biochemical modifications and enhancement of psoralen content in salt-stressed seedlings of Psoralea corylifolia Linn. J Funct Environ Bot 2:65–74. https://doi.org/10.5897/ajb2012.2925
Khubeiz MJ, Mansour G (2016) In vitro antifungal, antimicrobial properties and chemical composition of Santolina chamaecyparissus essential oil in Syria. Int J Toxicol Pharmacol Res 8:372–378
Khushboo PS, Jadhav VM, Kadam VJ, Sathe NS (2010) Psoralea corylifolia Linn.-“Kushtanashini.” Pharmacogn Rev 4:69–76. https://doi.org/10.4103/0973-7847.65331
Koul B, Taak P, Kumar A, Sanyal I (2019) Genus Psoralea: a review of the traditional and modern uses, phytochemistry and pharmacology. J Ethnopharmacol 232:201–226. https://doi.org/10.1016/j.jep.2018.11.036
Kumari R, Agrawal SB (2010) Supplemental UV-B induced changes in leaf morphology, physiology and secondary metabolites of an indian aromatic plant Cymbopogon citratus (D.C.) staph under natural field conditions. Int J Environ Stud 67:655–675. https://doi.org/10.1080/00207233.2010.513828
Liu B, Liu XB, Li YS, Herbert SJ (2013) Effects of enhanced UV-B radiation on seed growth characteristics and yield components in soybean. Field Crops Res 154:158–163. https://doi.org/10.1016/j.fcr.2013.08.006
Machado F, Dias MC, de Pinho PG et al (2017) Photosynthetic performance and volatile organic compounds profile in Eucalyptus globulus after UVB radiation. Environ Exp Bot 140:141–149. https://doi.org/10.1016/j.envexpbot.2017.05.008
Mannucci A, Mariotti L, Castagna A et al (2020) Hormone profile changes occur in roots and leaves of Micro-Tom tomato plants when exposing the aerial part to low doses of UV-B radiation. Plant Physiol Biochem 148:291–301. https://doi.org/10.1016/j.plaphy.2020.01.030
Martínez-lüscher J, Morales F, Delrot S et al (2013) Short- and long-term physiological responses of grapevine leaves to UV-B radiation. Plant Sci 213:114–122. https://doi.org/10.1016/j.plantsci.2013.08.010
Mehta G, Nayak UR, Dev S (1966) Bakuchiol, a novel monoterpenoids. Tetrahedron Lett 7:4561–4567
Mulyaningsih S, Sporer F, Zimmermann S et al (2010) Synergistic properties of the terpenoids aromadendrene and 1,8-cineole from the essential oil of Eucalyptus globulus against antibiotic-susceptible and antibiotic-resistant pathogens. Phytomedicine 17:1061–1066. https://doi.org/10.1016/j.phymed.2010.06.018
Nazari M, Zarinkamar F (2020) Industrial Crops & Products Ultraviolet-B induced changes in Mentha aquatica (a medicinal plant ) at early and late vegetative growth stages: investigations at molecular and genetic levels. Ind Crops Prod 154:112618. https://doi.org/10.1016/j.indcrop.2020.112618
Pandey A, Agrawal SB (2020) Ultraviolet-B radiation: a potent regulator of flavonoids biosynthesis, accumulation and functions in plants. Curr Sci 119:176–185
Pandey A, Jaiswal D, Agrawal SB (2021) Ultraviolet-B mediated biochemical and metabolic responses of a medicinal plant AdhatodavasicaNees. at different growth stages. J Photochem Photobiol B Biol 216:112142. https://doi.org/10.1016/j.jphotobiol.2021.112142
Park HJ, Kim WY, Pardo JM, Yun DJ (2016) Molecular interactions between flowering time and abiotic stress pathways. In: Jeon KW, Galluzzi L (eds) International review of cell and molecular biology. Elsevier, pp 371–412. https://doi.org/10.1016/bs.ircmb.2016.07.001
Pérez-López A, Cirio AT, Rivas-Galindo VM et al (2011) Activity against Streptococcus pneumoniae of the essential oil and δ-cadinene isolated from Schinus molle fruit. J Essent Oil Res 23:25–28. https://doi.org/10.1080/10412905.2011.9700477
Petropoulou Y, Georgiou O, Psaras GK, Manetas Y (2001) Improved flower advertisement, pollinator rewards and seed yield by enhanced UV-B radiation in the Mediterranean annual Malcolmia maritima. New Phytol 152:85–90. https://doi.org/10.1046/j.0028-646X.2001.00241.x
Rai K, Agrawal SB (2020) Effect on essential oil components and wedelolactone content of a medicinal plant Eclipta alba due to modifications in the growth and morphology under different exposures of ultraviolet-B. Physiol Mol Biol Plants. https://doi.org/10.1007/s12298-020-00780-8
Ranjbarfordoei A, Samson R, Van Damme P (2011) Photosynthesis performance in sweet almond [Prunus dulcis (Mill) D. Webb] exposed to supplemental UV-B radiation. Photosynthetica 49:107–111. https://doi.org/10.1007/s11099-011-0017-z
Reddy KR, Singh SK, Koti S et al (2013) Quantifying the effects of corn growth and physiological responses to ultraviolet-B radiation for modeling. Agron J 105:1367–1377. https://doi.org/10.2134/agronj2013.0113
Romanatti PV, Rocha GA, Júnior VV et al (2019) Limitation to photosynthesis in leaves of eggplant under UVB according to anatomical changes and alterations on the antioxidant system. Sci Hortic 249:449–454. https://doi.org/10.1016/j.scienta.2019.01.060
Salehi B, Upadhyay S, Orhan IE, Jugran AK, Jayaweera SLD et al (2019) Therapeutic potential of α-and β-pinene: a miracle gift of nature. Biomolecules 9:738. https://doi.org/10.3390/biom9110738
Satdive RK, Suchita K, Shraddha S, Sudhir S, Fulzele DP (2014) The influence of cadmium as abiotic elicitor on the production of phytoestrogens in hairy root cultures of Psoralea corylifolia. Int J Pharmal Bio Sci 5:548–558
Schreiner M, Krumbein A, Mewis I, Ulrichs C, Huyskens-Keil S (2009) Short-term and moderate UV-B radiation effects on secondary plant metabolism in different organs of nasturtium (Tropaeolum majus L.). Innov Food Sci Emerg Technol 10:93–96. https://doi.org/10.1016/j.ifset.2008.10.001
Schreiner M, Mewis I, Huyskens-Keil S, Jansen MAK, Zrenner R, Winkler JB, O’brien N, Krumbein A (2012) UV-B-induced secondary plant metabolites-potential benefits for plant and human health. Crit Rev Plant Sci 31:229–240. https://doi.org/10.1080/07352689.2012.664979
Sellapan P, Rohani ER, Noor NM (2018) Sesquiterpene production in methyl Jasmonate-induced Persicaria minor cell suspension culture. Sains Malaysiana 47:3051–3059. https://doi.org/10.17576/jsm-2018-4712-15
Takshak S, Agrawal SB (2015) Defence strategies adopted by the medicinal plant Coleus forskohlii against supplemental ultraviolet-B radiation: augmentation of secondary metabolites and antioxidants. Plant Physiol Biochem 97:124–138. https://doi.org/10.1016/j.plaphy.2015.09.018
Takshak S, Agrawal SB (2018) Interactive effects of supplemental ultraviolet-B radiation and indole-3-acetic acid on Coleus forskohlii Briq.: alterations in morphological-, physiological-, and biochemical characteristics and essential oil content. Ecotoxicol Environ Saf 147:313–326. https://doi.org/10.1016/j.ecoenv.2017.08.059
Takshak S, Agrawal SB (2019) Defense potential of secondary metabolites in medicinal plants under UV-B stress. J Photochem Photobiol B Biol 193:51–88. https://doi.org/10.1016/j.jphotobiol.2019.02.002
Teramura AH (1983) Effects of ultraviolet-B radiation on the growth and yield of crop plants. Physiol Plant 58:415–427
Teramura AH, Lydon SJH (1990) Effects of UV-B radiation on soybean yield and seed quality: a 6-year field study. Physiol Plant 80:5–11
Tevini M, Teramura AH (1989) UV-B effects on terrestrial plants. Photochem Photobiol 50:479–487
Tripathi R, Agrawal SB (2012) Effects of ambient and elevated level of ozone on Brassica campestris L. with special reference to yield and oil quality parameters. Ecotoxicol Environ Saf 85:1–12. https://doi.org/10.1016/j.ecoenv.2012.08.012
Tripathi R, Agrawal SB (2013) Interactive effect of supplemental ultraviolet B and elevated ozone on seed yield and oil quality of two cultivars of linseed (Linumusitatissimum L.) carried out in open top chambers. J Sci Food Agric 93:1016–1025. https://doi.org/10.1002/jsfa.5838
Tripathi R, Rai K, Singh S, Agrawal M, Agrawal SB (2019) Role of supplemental UV-B in changing the level of ozone toxicity in two cultivars of sun flower : growth, seed yield and oil quality. Ecotoxicology 28:277–293. https://doi.org/10.1007/s10646-019-02020-6
Turkez H, Togar B, Tatar A, Geyıkoglu F, Hacımuftuoglu A (2014) Cytotoxic and cytogenetic effects of α-copaene on rat neuron and N2a neuroblastoma cell lines. Biologia 69:936–942. https://doi.org/10.2478/s11756-014-0393-5
Turkez H, Togar B, Di Stefano A, Taspınar N, Sozio P (2015) Protective effects of cyclosativene on H2O2-induced injury in cultured rat primary cerebral cortex cells. Cytotechnology 67:299–309. https://doi.org/10.1007/s10616-013-9685-9
Voisin AS, Salon C, Jeudy C, Warembourg FR (2003) Seasonal patterns of 13C partitioning between shoots and nodulated roots of N2-or nitrate-fed Pisum sativum L. Ann Bot 91:539–546. https://doi.org/10.1093/aob/mcg055
Xin Z, Wu X, Ji T et al (2019) Bakuchiol: a newly discovered warrior against organ damage. Pharmacol Res 141:208–213. https://doi.org/10.1016/j.phrs.2019.01.001
Yao Y, Xuan Z, Li Y, He Y, Korpelainen H, Li C (2006) Effects of ultraviolet-B radiation on crop growth, development, yield and leaf pigment concentration of tartary buckwheat (Fagopyrum tataricum) under field conditions. Eur J Agron 25:215–222. https://doi.org/10.1016/j.eja.2006.05.004
Zhao D, Reddy KR, Kakani VG et al (2004) Leaf and canopy photosynthetic characteristics of cotton (Gossypium hirsutum) under elevated CO2 concentration and UV-B radiation. J Plant Physiol 161:581–590. https://doi.org/10.1078/0176-1617-01229
Zhao M, Liu B, Sun Y et al (2020) Presence and roles of myrtenol, myrtanol and myrtenal in Dendroctonus armandi (Coleoptera: Curculionidae: Scolytinae) and Pinus armandi (Pinales: Pinaceae: Pinoideae). Pest Manag Sci 76:188–197. https://doi.org/10.1002/ps.5492
Acknowledgements
The authors are grateful to the Head, Department of Botany, Banaras Hindu University, for providing all the necessary laboratory facilities. Coordinator, Centre of Advanced Study (CAS), Department of Botany, Department of Science and Technology (DST-FIST), Interdisciplinary School of Life Sciences (ISLS) are acknowledged for all research facilities. We are thankful to Indian Council of Agricultural Research-National Bureau of Plant Genetic Resources (ICAR-NBPGR), Indian Agricultural Research Institute (IARI), New Delhi, India for providing seeds of Psoralea corylifolia and Advanced Instrumentation Research Facility (AIRF), Jawaharlal Nehru University for GC-MS facility. University Grants Commission (UGC) is greatly acknowledged for providing the financial support in the form of Senior Research Fellowship to Avantika Pandey.
Funding
Avantika Pandey received financial support in the form of Senior Research Fellowship (SRF) from University Grants Commission (UGC), New Delhi, India.
Author information
Authors and Affiliations
Contributions
Conceptualization, Validation, Formal analysis, Investigation, Data curation, Writing-Original draft: AP; Visualization, Formal analysis: MA; Conceptualization, Validation, Supervision: SBA. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethics approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Additional information
Communicated by Jeong Hee Choi.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Pandey, A., Agrawal, M. & Agrawal, S.B. Ultraviolet-B induced modifications in growth, physiology, essential oil content and composition of a medicinal herbal plant Psoralea corylifolia. Hortic. Environ. Biotechnol. 63, 917–934 (2022). https://doi.org/10.1007/s13580-022-00454-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13580-022-00454-2