Abstract
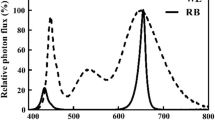
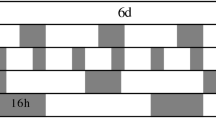
This study investigated the effects of constant light and alternating relatively high-intensity (500 µmol m−2 s−1) and low-intensity (150 µmol m−2 s−1) red–blue LEDs (4R:1B) on the biomass production and quality of hydroponic purple-leaf lettuce (Lactuca sativa L. cv. ‘Zishan’) in an environmentally controlled plant factory. Four treatments were set up to separate 1 h of high light into four different alternating frequencies in a 24-h light−dark cycle (16/8 h): one time (A1), three times (A3), six times (A6), and twelve times (A12). In addition, one constant light treatment with the same daily light integral (DLI, 9.8 mol m−2 per day) as other treatments was set as the control (CK, 170 µmol m−2 s−1). The results indicated that A6 significantly reduced shoot fresh weight and increased the root–shoot ratio of lettuce compared with CK, but there was no significant difference among other treatments. Alternating light treatments did not promote the accumulation of soluble sugar, soluble protein, and phenolic substances compared with CK. Meanwhile, A12 significantly promoted the accumulation of total ascorbate (TA) in lettuce leaves compared with other treatments but decreased ascorbate/TA ratio. Above all, under the same DLI condition, alternating high and low light did not have obvious positive effects on biomass production and the accumulation of nutrient substance in lettuce under constant light was better than that under alternating light. Therefore, compared with the fluctuating radiation with the same DLI, constant radiation is a better choice for lettuce production.





Similar content being viewed by others
References
Barrero JM, Downie AB, Xu Q, Gubler F (2014) A role for barley CRYPTOCHROME1 in light regulation of grain dormancy and germination. Plant Cell 26:1094–1104. https://doi.org/10.1105/tpc.113.121830
Blokhina O (2003) Antioxidants, oxidative damage and oxygen deprivation stress: a review. Ann Bot 91:179–194. https://doi.org/10.1093/aob/mcf118
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254. https://doi.org/10.1016/0003-2697(76)90527-3
Chalker-Scott L (1999) Environmental significance of anthocyanins in plant stress responses. Photochem Photobiol 70:1–9. https://doi.org/10.1111/j.1751-1097.1999.tb01944.x
Chen X, Yang Q (2018) Effects of intermittent light exposure with red and blue light emitting diodes on growth and carbohydrate accumulation of lettuce. Sci Hortic 234:220–226. https://doi.org/10.1016/j.scienta.2018.02.0557
Chen X, Wang L, Li T, Yang Q, Guo W (2019) Sugar accumulation and growth of lettuce exposed to different lighting modes of red and blue LED light. Sci Rep. https://doi.org/10.1038/s41598-019-43498-8
Coley PD, Bryant JP, Chapin FS (1985) Resource availability and plant antiherbivore defense. Science 230:895–899. https://doi.org/10.1126/science.230.4728.895
Conklin PL (2001) Recent advances in the role and biosynthesis of ascorbic acid in plants. Plant Cell Environ 24:383–394. https://doi.org/10.1046/j.1365-3040.2001.00686.x
Devlin PF, Kay SA (2000) Cryptochromes are required for phytochrome signaling to the circadian clock but not for rhythmicity. Plant Cell Online 12:2499. https://doi.org/10.2307/3871244
Dubois M, Gilles KA, Hamilton JK, Rebers PA, Smith F (1956) colorimetric method for determination of sugars and related substances. Anal Chem 28:350–356. https://doi.org/10.1021/ac60111a017
Farrar J, Pollock C, Gallagher J (2000) Sucrose and the integration of metabolism in vascular plants. Plant Sci 154:0–11. https://doi.org/10.1016/s0168-9452(99)00260-5
Franklin KA (2009) Light and temperature signal crosstalk in plant development. Curr Opin Plant Biol 12:63–68. https://doi.org/10.1016/j.pbi.2008.09.007
Giusti MM, Wrolstad RE (2001) Characterization and measurement of anthocyanins by UV–Visible spectroscopy. Curr Prot Food Anal Chem. https://doi.org/10.1002/0471142913.faf0102s00
Goins GD, Yorio NC, Sanwo MM, Brown CS (1997) Photomorphogenesis, photosynthesis, and seed yield of wheat plants grown under red light-emitting diodes (LEDs) with and without supplemental blue lighting. J Exp Bot. https://doi.org/10.1093/jxb/48.7.1407
Hoagland DR, Arnon DI (1950) The water-culture method for growing plants without soil. California Agric Exp Stat Circ 347:1–32
Jishi T, Kimura K, Matsuda R, Fujiwara K (2016) Effects of temporally shifted irradiation of blue and red LED light on cos lettuce growth and morphology. Sci Hortic 198:227–232. https://doi.org/10.1016/j.scienta.2015.12.005
Kami C, Lorrain S, Hornitschek P, Fankhauser C (2010) Light-regulated plant growth and development. Curr Top Dev Biol 91:29–66. https://doi.org/10.1016/s0070-2153(10)91002-8
Koukol J, Conn EE (1961) The metabolism of aromatic compounds in higher plants. IV. Purification and properties of the phenylalanine deaminase of Hordeum vulgare. J Biol Chem 236:2692–2698
Kozai T, Fujiwara K, Runkle ES (2016) Led lighting for urban agriculture. Springer, Berlin
Lee SK, Kader AA (2000) Preharvest and postharvest factors influencing Vitamin C content of horticultural crops. Postharvest Biol Technol 20:207–220. https://doi.org/10.1016/s0925-5214(00)00133-2
Li Q, Kubota C (2009) Effects of supplemental light quality on growth and phytochemicals of baby leaf lettuce. Environ Exp Bot 67:59–64. https://doi.org/10.1016/j.envexpbot.2009.06.011
Lister CE, Lancaster JE, Walker JRL (1996) Developmental changes in enzymes of flavonoid biosynthesis in the skins of red and green apple cultivars. J Sci Food Agric 71:313–320. https://doi.org/10.1002/(SICI)1097-0010(199607)71:3<313::AID-JSFA586>3.0.CO;2-N
Liu X, Ardo S, Bunning M, Parry J, Zhou K, Stushnoff C, Kendall P (2007) Total phenolic content and DPPH radical scavenging activity of lettuce (Lactuca sativa L.) grown in Colorado. LWT-Food Sci Technol 40:552–557. https://doi.org/10.1016/j.lwt.2005.09.007
Mancinelli AL, Rabino I (1978) The “high irradiance responses” of plant photomorphogenesis. Bot Rev 44:129–180. https://doi.org/10.1007/bf02919078
Mancinelli AL, Rabino I (1985) Effectiveness of intermittent light treatments on anthocyanin synthesis in dark-grown and light-pretreated seedlings. Plant Physiol 78:203–206. https://doi.org/10.1104/pp.78.1.203
Massa GD, Kim HH, Wheeler RM, Mitchell CA (2008) Plant productivity in response to LED lighting. Hortscience. https://doi.org/10.21273/HORTSCI.43.7.1951
Mori T, Sakurai M, Sakuta M (2000) Changes in PAL, CHS, DAHP synthase (DS-Co and Ds-Mn) activity during anthocyanin synthesis in suspension culture of Fragaria ananassa. Plant Cell Tissue Organ Cult 62:135–139. https://doi.org/10.1023/a:1026560223795
Oh MM, Carey EE, Rajashekar CB (2009) Environmental stresses induce health-promoting phytochemicals in lettuce. Plant Physiol Biochem 47:578–583. https://doi.org/10.1016/j.plaphy.2009.02.008
Padayatty SJ, Katz A, Wang Y, Eck P, Kwon O, Lee JH, Levine M (2003) Vitamin C as an antioxidant: evaluation of its role in disease prevention. J Am Coll Nutr 22:18–35. https://doi.org/10.1080/07315724.2003.10719272
Pérez-López U, Sgherri C, Miranda-Apodaca J, Micaelli F, Lacuesta M, Mena-Petite A, Muñoz-Rueda A (2018) Concentration of phenolic compounds is increased in lettuce grown under high light intensity and elevated CO2. Plant Physiol Biochem 123:233–241. https://doi.org/10.1016/j.plaphy.2017.12.010
Phillips KM, Tarragó-Trani MT, Gebhardt SE, Exler J, Patterson KY, Haytowitz DB, Holden JM (2010) Stability of Vitamin C in frozen raw fruit and vegetable homogenates. J Food Compos Anal 23:253–259. https://doi.org/10.1016/j.jfca.2009.08.018
Shao M, Liu W, Zha L, Zou C, Zhang Y, Li B (2020) Differential effects of high light duration on growth, nutritional quality, and oxidative stress of hydroponic lettuce under red and blue LED irradiation. Sci Hortic. https://doi.org/10.1016/j.scienta.2020.109366
Sivakumar G, Heo JW, Kozai T, Paek KY (2006) Effect of continuous or intermittent radiation on sweet potato plantlets in vitro. J Hortic Sci Biotech 81:546–548. https://doi.org/10.1080/14620316.2006.11512101
Son KH, Jeon YM, Oh MM (2016) Application of supplementary white and pulsed light-emitting diodes to lettuce grown in a plant factory with artificial lighting. Hortic Environ Biote 57:560–572. https://doi.org/10.1007/s13580-016-0068-y
Song JY, Lee JS, An CS (1998) Expression of CHS, CHI, and DFR genes in response to light in small radish seedlings. J Plant Biol 41:277–282. https://doi.org/10.1007/bf03030328
Spínola V, Mendes B, Câmara JS, Castilho PC (2012) An improved and fast UHPLC-PDA methodology for determination of L-ascorbic and dehydroascorbic acids in fruits and vegetables. Evaluation of degradation rate during storage. Anal Bioanal Chem 403:1049–1058. https://doi.org/10.1007/s00216-011-5668-x
Yorio NC, Goins GD, Kagie HR, Wheeler RM, Sager JC (2001) Improving spinach, radish, and lettuce growth under red light-emitting diodes (LEDs) with blue light supplementation. Hortscience 36:380–383. https://doi.org/10.21273/HORTSCI.36.2.380
Zhang D, Quantick PC (1997) Effects of chitosan coating on enzymatic browning and decay during postharvest storage of litchi (Litchi chinensis Sonn.) fruit. Postharvest Biol Tec 12:195–202. https://doi.org/10.1016/s0925-5214(97)00057-4
Acknowledgements
This work was supported by the National Natural Science Foundation of China (NSFC) (No. 31672202).
Author information
Authors and Affiliations
Contributions
WL provided equipment and funds. WL and MS conceived the experiment methods. MS, LZ, YZ, CZ, and BL prepared the materials for the experiment, collected samples, and undertook experiments. MS analyzed the data, prepared and revised the manuscript.
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Communicated by Myung-Min Oh.
Rights and permissions
About this article
Cite this article
Shao, M., Liu, W., Zha, L. et al. Effects of constant versus fluctuating red–blue LED radiation on yield and quality of hydroponic purple-leaf lettuce. Hortic. Environ. Biotechnol. 61, 989–997 (2020). https://doi.org/10.1007/s13580-020-00285-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13580-020-00285-z