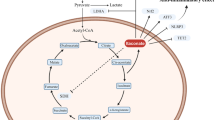
Abstract
Antipsychotic-induced weight gain is a well-established but poorly understood clinical phenomenon. New mechanistic insights into how antipsychotics modulate adipose physiology are sorely needed, in hopes of either devising a therapeutic intervention to ameliorate weight gain or contributing to improved design of future agents. In this study, we have hypothesized that the weight gain-associated tricyclic antipsychotics clozapine and chlorpromazine directly impact adipose tissue by potentiating adipogenic differentiation of preadipocytes. Utilizing a well-established in vitro model system (3T3-L1 preadipocyte cell line), we demonstrate that, when applied specifically during induction of adipogenic differentiation, both clozapine and chlorpromazine significantly potentiate in vitro adipogenesis, observed as morphological changes and increased intracellular lipid accumulation. These persistent effects, observed at endpoints well after the end of antipsychotic exposure, are accompanied by increased transcript- and protein-level expression of the mature adipocyte marker perilipin-1, as indicated by RT-qPCR and Western blotting, but not by further upregulation of pro-adipogenic transcription factors versus positive controls. Our findings point to a possible physiological mechanism of antipsychotic-induced hyperplasia, with potentiated expression of mature adipocyte markers enhancing the differentiation and maturation of preadipocytes.




Similar content being viewed by others
Abbreviations
- Clz:
-
Clozapine
- Cpz:
-
Chlorpromazine
- CEBP:
-
CCAAT-enhancer-binding protein
- ORO:
-
Oil Red O
- PPAR:
-
Peroxisome proliferator-activated receptor
- Plpn-1:
-
Perilipin-1
- PI:
-
Post-induction
- RT-qPCR:
-
Reverse-transcription quantitative (real-time) PCR
- UD:
-
Undifferentiated
References
J.M. Meyer, Pharmacotherapy of psychosis and mania, in: Goodman and Gilman’s The Pharmacological Basis of Therapeutics 13th ed., McGraw-Hill Education, 2018: pp. 279–302.
J.A. Lieberman, R.A. Rosenheck, S.M. Davis, J.K. Hsiao, Effectiveness of antipsychotic drugs in patients with chronic schizophrenia, New Eng J Med. (2005) 15.
Bak M, Fransen A, Janssen J, van Os J, Drukker M. Almost all antipsychotics result in weight gain: a meta-analysis. PLoS ONE. 2014;9:e94112. https://doi.org/10.1371/journal.pone.0094112.
Li Y, Zhao X, Feng X, Liu X, Deng C, Hu C-H. Berberine alleviates olanzapine-induced adipogenesis via the AMPKα–SREBP pathway in 3T3-L1 cells. Int J Mol Sci. 2016;17:1865. https://doi.org/10.3390/ijms17111865.
Yang Z, Yin J-Y, Gong Z-C, Huang Q, Chen H, Zhang W, Zhou H-H, Liu Z-Q. Evidence for an effect of clozapine on the regulation of fat-cell derived factors. Clin Chim Acta. 2009;408:98–104. https://doi.org/10.1016/j.cca.2009.07.021.
Tsubai T, Yoshimi A, Hamada Y, Nakao M, Arima H, Oiso Y, Noda Y. Effects of clozapine on adipokine secretions/productions and lipid droplets in 3T3-L1 adipocytes. J Pharmacol Sci. 2017;133:79–877. https://doi.org/10.1016/j.jphs.2017.01.004.
Chen C-C, Hsu L-W, Huang K-T, Goto S, Chen C-L, Nakano T. Overexpression of Insig-2 inhibits atypical antipsychotic-induced adipogenic differentiation and lipid biosynthesis in adipose-derived stem cells. Sci Rep. 2017;7:1–10. https://doi.org/10.1038/s41598-017-11323-9.
Sertié AL, Suzuki AM, Sertié RAL, Andreotti S, Lima FB, Passos-Bueno MR, Gattaz WF. Effects of antipsychotics with different weight gain liabilities on human in vitro models of adipose tissue differentiation and metabolism. Prog Neuropsychopharmacol Biol Psychiatry. 2011;35:1884–900. https://doi.org/10.1016/j.pnpbp.2011.07.017.
Green H, Kehinde O. An established preadipose cell line and its differentiation in culture II. Factors affecting the adipose conversion. Cell. 1975;5:19–27. https://doi.org/10.1016/0092-8674(75)90087-2.
Green H, Meuth M. An established pre-adipose cell line and its differentiation in culture. Cell. 1974;3:127–33. https://doi.org/10.1016/0092-8674(74)90116-0.
Hemmrich K, Gummersbach C, Pallua N, Luckhaus C, Fehsel K. Clozapine enhances differentiation of adipocyte progenitor cells. Mol Psychiatry. 2006;11:980–1. https://doi.org/10.1038/sj.mp.4001892.
Yang L-H, Chen T-M, Yu S-T, Chen Y-H. Olanzapine induces SREBP-1-related adipogenesis in 3T3-L1 cells. Pharmacol Res. 2007;56:202–8. https://doi.org/10.1016/j.phrs.2007.05.007.
Nimura S, Yamaguchi T, Ueda K, Kadokura K, Aiuchi T, Kato R, Obama T, Itabe H. Olanzapine promotes the accumulation of lipid droplets and the expression of multiple perilipins in human adipocytes. Biochem Biophys Res Commun. 2015;467:906–12. https://doi.org/10.1016/j.bbrc.2015.10.045.
Cottingham C, Che P, Zhang W, Wang H, Wang RX, Percival S, Birky T, Zhou L, Jiao K, Wang Q. Diverse arrestin-recruiting and endocytic profiles of tricyclic antipsychotics acting as direct α 2A adrenergic receptor ligands. Neuropharmacology. 2017;116:38–49. https://doi.org/10.1016/j.neuropharm.2016.12.004.
Rosen ED, MacDougald OA. Adipocyte differentiation from the inside out. Nat Rev Mol Cell Biol. 2006;7:885–96. https://doi.org/10.1038/nrm2066.
Cristancho AG, Lazar MA. Forming functional fat: a growing understanding of adipocyte differentiation. Nat Rev Mol Cell Biol. 2011;12:722–34. https://doi.org/10.1038/nrm3198.
Schmittgen TD, Livak KJ. Analyzing real-time PCR data by the comparative CT method. Nat Protoc. 2008;3:1101–8. https://doi.org/10.1038/nprot.2008.73.
Cottingham C, Chen Y, Jiao K, Wang Q. The antidepressant desipramine is an arrestin-biased ligand at the α 2A -adrenergic receptor driving receptor down-regulation in vitro and in vivo. J Biol Chem. 2011;286:36063–755. https://doi.org/10.1074/jbc.M111.261578.
Cottingham C, Jones A, Wang Q. Desipramine selectively potentiates norepinephrine-elicited ERK1/2 activation through the α2A adrenergic receptor. Biochem Biophys Res Commun. 2012;420:161–5. https://doi.org/10.1016/j.bbrc.2012.02.135.
Tang QQ, Lane MD. Adipogenesis: from stem cell to adipocyte. Annu Rev Biochem. 2012;81:715–36. https://doi.org/10.1146/annurev-biochem-052110-115718.
Brasaemle DL. Thematic review series: Adipocyte Biology The perilipin family of structural lipid droplet proteins: stabilization of lipid droplets and control of lipolysis. J Lipid Res. 2007;48:2547–59. https://doi.org/10.1194/jlr.R700014-JLR200.
Lafontan M, Berlan M. Fat cell α2-adrenoceptors: the regulation of fat cell function and lipolysis*. Endocr Rev. 1995;16:716–38. https://doi.org/10.1210/edrv-16-6-716.
Shenoy SK, Lefkowitz RJ. Multifaceted roles of beta-arrestins in the regulation of seven-membrane-spanning receptor trafficking and signalling. Biochem J. 2003;375:503–15. https://doi.org/10.1042/BJ20031076.
Rajagopal S, Rajagopal K, Lefkowitz RJ. Teaching old receptors new tricks: biasing seven-transmembrane receptors. Nat Rev Drug Discov. 2010;9:373–86. https://doi.org/10.1038/nrd3024.
Acknowledgements
The authors thank Kurt Gibbs (Morehead State University) for advice regarding RT-qPCR assays, Amber Hall (formerly of Morehead State University) for contributions to RT-qPCR data collection, and Qin Wang (University of Alabama at Birmingham) for providing frozen 3T3-L1 stock.
Funding
This work was supported by the National Institutes of Health [grant number P20GM103436].
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest. Other ethics approvals are not applicable to this study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Cottingham, C.M., Patrick, T., Richards, M.A. et al. Tricyclic antipsychotics promote adipogenic gene expression to potentiate preadipocyte differentiation in vitro. Human Cell 33, 502–511 (2020). https://doi.org/10.1007/s13577-020-00372-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13577-020-00372-4