Abstract
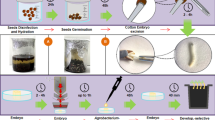
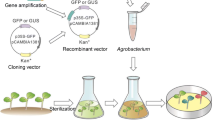
A cell line, named CCL-1 has been obtained in cotton which proliferates on phytohormone free MS medium containing high KNO3. This cell line can be maintained as a friable callus on solidified medium and also readily converted to cell suspensions and maintained in liquid medium. CCL-1 cell suspensions were transformed using a disarmed Agrobacterium strain with binary vectors containing selectable marker genes- nptII, hptII, bar and ALS dm to check the efficacy of these marker genes for selecting transformed cell lines. Transformation was monitored by reporter genes, either GUS or GFP. Transformed cell lines could be readily obtained with all the marker genes, hpt being the most efficient marker for unambiguous selection in 10 day time period. Transformed cell lines showed expression of the reporter genes. We developed a protocol for using these cell lines for insect feeding bioassays by using control CCL-1 and a transformed derivative line containing the cry1Ac gene. The most optimal feeding method is to mix lyophilized transformed cells with an artificial diet to achieve various concentrations of the toxin. The most useful selectable markers for insect feeding bioassay studies are bar and ALS dm as these do not interfere with insect growth. CCL-1 will be a convenient tool for studying transgene expression in cotton and will be particularly useful for testing a large number of genes for antifeedant/ insecticidal activity.




Similar content being viewed by others
Abbreviations
- GUS:
-
β-Glucuronidase
- hpt :
-
Hygromycin phosphotransferase
- nptII :
-
Neomycin phosphotransferase
- bar :
-
phosphinothricin acetyl transferase
- Als :
-
Acetolactate synthase
- MS medium:
-
Murashige and Skoog medium
References
Adang MJ, Staver MJ, Rocheleau TA, Leighton J, Barker RF, Thompson DV (1985) Characterized full length and truncated plasmid clones of the crystal protein of Bacillus thuringiensis subsp. Kurstaki HD-73 and their toxicity to Manduca sexta. Gene 36:289–300
Avilla C, Vargas-Osuna E, Gonzalez-Cabrera J, Ferre J, Gonzalez-Zamora JE (2005) Toxicity of several δ-endotoxins of Bacillus thuringiensis against Helicoverpa armigera (Lepidoptera: Noctuidae) from Spain. J Invertebr Pathol 90:51–54
Babu BG, Udayasuriyan V, Mariam MA, Sivakumar NC, Bharathi M, Balasubramanian G (2002) Comparative toxicity of Cry1Ac and Cry2Aa δ-endotoxins of Bacillus thuringiensis against Helicoverpa armigera (H.). Crop Prot 21:817–822
Bantscheff M, Schirle M, Sweetman G, Rick J, Kuster B (2007) Quantitative mass spectrometry in proteomics: a critical review. Anal Bioanal Chem 389:1017–1031
Bayley C, Trolinder N, Ray C, Morgan M, Quisenberry JE, Ow DW (1992) Engineering 2,4-D resistance into cotton. Theor Appl Genet 83:645–649
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254
Chakrabarti SK, Mandaokar A, Kumar PA, Sharma RP (1998) Efficacy of lepidopteran specific δ-endotoxins of Bacillus thuringiensis against Helicoverpa armigera. J Invertebr Pathol 72:336–337
Chaudhary B, Kumar S, Prasad KV, Oinam GS, Burma PK, Pental D (2003) Slow desiccation leads to high-frequency shoot recovery from transformed somatic embryos of cotton (Gossypium hirsutum L. cv. Coker 310 FR). Plant Cell Rep 21:955–960
David KM, Perrot-Rechenmann C (2001) Characterization of a tobacco BY2 cell line expressing the tetracycline repressor at a high level for strict regulation of transgene expression. Plant Physiol 125:1548–1553
Davidonis GH, Hamilton RH (1983) Plant regeneration from callus tissue of Gossypium hirsutum L. Plant Sci Lett 32:89–93
Finer JJ, McMullen MD (1990) Transformation of cotton (Gossypium hirsutum L.) via particle bombardment. Plant Cell Rep 8:586–589
Finney DJ (1971) Probit analysis, 3rd edn. Cambridge Univ. Press, Cambridge
Fraley RT, Rogers SG, Horsch RB, Sanders PR, Flick JS, Adams SP, Bittner ML, Brand LA, Fink CL, Fry JS, Galluppi GR, Goldberg SB, Hoffmann NL, Woo SC (1983) Expression of bacterial genes in plant cells. Proc Natl Acad Sci U S A 80:4803–4807
Gupta GP, Birah A, Rani S (2004) Development of artificial diet for mass rearing of American bollworm, Helicoverpa armigera. Indian J Agric Sci 74:548–551
Hale JE (2013) Advantageous uses of mass spectrometry for the quantification of proteins. Int J Proteomics. doi:10.1155/2013/219452
Hajdukiewicz P, Svab Z, Maliga P (1994) The small, versatile pPZP family of Agrobacterium binary vectors for plant transformation. Plant Mol Biol 25:989–994
James C (2013) Global status of commercialized biotech/GM crops: ISAAA briefs No. 46. ISAAA. ISAAA, Ithaca
Jefferson RA, Kavanagh TA, Bevan MW (1987) GUS fusions: β-glucuronidase as a sensitive and versatile gene fusion marker in higher plants. EMBO J 6:3901–3907
Jin S, Zhang X, Liang S, Nie Y, Guo X, Huang C (2005) Factors affecting transformation efficiency of embryogenic callus of Upland cotton (Gossypium hirsutum) with Agrobacterium tumefaciens. Plant Cell Tissue Organ Cult 81:229–237
Ke L, Liu R, Chu B, Yu X, Sun J, Jones B, Pan G, Cheng X, Wang H, Zhu S, Sun Y (2012) Cell suspension culture-mediated incorporation of the rice bel gene into transgenic cotton. PLoS One 7(7):e39974
Khan T, Reddy VS, Leelavathi S (2010) High-frequency regeneration via somatic embryogenesis of an elite recalcitrant cotton genotype (Gossypium hirsutum L.) and efficient Agrobacterium-mediated transformation. Plant Cell Tissue Organ Cult 101:323–330
Koncz C, Schell J (1986) The promoter of TL-DNA gene 5 controls the tissue-specific expression of chimeric gene carried by a novel type of Agrobacterium binary vector. Mol Gen Genet 204:383–396
Kumar M, Shukla AK, Singh H, Tuli R (2009) Development of insect resistant transgenic cotton lines expressing cry1EC gene from an insect bite and wound inducible promoter. J Biotechnol 140:143–148
Kumar S, Birah A, Chaudhary B, Burma PK, Gupta GP, Pental D (2005) Plant codon optimized cry genes of Bacillus thuringiensis can be expressed as soluble proteins in Escherichia coli BL21 codon plus strain as NusA-Cry proten fusion. J Invertebr Pathol 88:83–86
Kumar S, Sharma P, Pental D (1998) A genetic approach to in vitro regeneration of non-regenerating cotton (Gossypium hirsutum L.) cultivars. Plant Cell Rep 18:59–63
Lyon BR, Cousins YL, Llewellyn DJ, Dennis ES (1993) Cotton plants transformed with a bacterial degradation gene are protected from accidental spray drift damage by the herbicide 2,4-dichlorophenoxyacetic acid. Transgenic Res 2:162–169
Miao W, Wang X, Li M, Song C, Wang Y, Hu D, Wang J (2010) Genetic transformation of cotton with a hairpin-encoding gene hpa Xoo confers an enhanced defense response against different pathogens through a priming mechanism. BMC Plant Biol 10:67
Mishra R, Wang HY, Yadav NR, Wilkins TA (2003) Development of highly regenerable elite Acala cotton (Gossypium hirsutum cv. Maxxa)- a step towards genotype-independent regeneration. Plant Cell Tissue Organ Cult 73:21–35
Nagata T, Nemoto Y, Hasezawa S (1992) Tobacco BY-2 cell line as the “HeLa” cell in the cell biology of higher plants. Int Rev Cytol 132:1–30
Perlak FJ, Deaton RW, Armstrong TA, Fuchs RL, Sims SR, Greenplate JT, Fischhoff DA (1990) Insect resistant cotton plants. Nat Biotechnol 8:939–943
Rajasekaran K (2013) Biolistic transformation of cotton embryogenic cell suspension cultures. Methods Mol Biol 958:59–70
Steintz B, Gafni Y, Cohen Y, Diaz JP, Tabib Y, Levski S, Navos A (2002) Insecticidal activity of a Cry1A(C) transgene in callus derived from regenerated-recalcitrant cotton (Gossypium hirsutum L.). In Vitro Cell Dev Biol 38:247–251
Sunilkumar G, Rathore KS (2001) Transgenic cotton: factors influencing Agrobacterium-mediated transformation and regeneration. Mol Breed 8:37–52
Thomas JC, Adams DG, Keppenne VD, Wasmann CC, Brown JK, Kanost MR, Bohnert HJ (1995) Protease inhibitors of Manduca sexta expressed in transgenic cotton. Plant Cell Rep 14:758–762
XLSTAT (2013) Addinsoft™ Version 2013.3.03. Available online: http://www.xlstat.com
Zhang B (2013) Transgenic cotton: from biotransformation methods to agricultural application. Methods Mol Biol 958:3–15
Zhang T, Wu SJ (2012) Enhanced Agrobacterium-mediated transformation of embryogenic calli of upland cotton. Methods Mol Biol 847:245–253
Acknowledgments
DP acknowledges support of DST through a J. C. Bose fellowship. We thank Satish Kumar Yadava for his support with statistical analysis, Kumar Paritosh for help with reporter gene assays, Vinee Khanna for hpt::cry1Ac construct and Umakant for his help in insect feeding bioassays.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Supplementary Fig. 1
In vitro culture scheme used for the development of cell line CCL-1 (PPT 117 kb)
Supplementary Fig. 2
Growth of the CCL-1 cell line in liquid medium measured by increase in fresh weight of the suspension at different time intervals (PPT 102 kb)
Rights and permissions
About this article
Cite this article
Singh, A.K., Pental, D. Selection and genetic transformation of a fast-growing cell line in cotton (Gossypium hirsutum) for transgene expression studies. J. Plant Biochem. Biotechnol. 24, 225–232 (2015). https://doi.org/10.1007/s13562-014-0262-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13562-014-0262-x