Abstract
The design of biopolymers matrices for incorporating bioactive compounds represents a valuable technique for various biomedical and packaging applications. Propolis has developed as a natural byproduct from beekeeping for wound healing, food packaging, and food production applications. The current review focuses on the various composites prepared from propolis with polysaccharides like cellulose, chitosan, starch, and alginate, where the chemistry, synthesis, and application are seriously discussed. This study found that polysaccharide composite matrix with propolis may provide an appropriate platform for different applications such as wound healing and adequate biodegradable packaging. Using polysaccharide composite matrix with propolis is a promise policy for biodegradable active packaging upgrading and wound healing applications.
Similar content being viewed by others
Explore related subjects
Find the latest articles, discoveries, and news in related topics.Avoid common mistakes on your manuscript.
1 Introduction
The environmental problems, side effects, and shortage of synthetic materials encourage scientists to investigate new alternatives from natural resources. For example, biopolymers, consisting of different repeating units, were reported as promising biomaterials. These polymers are classified into polysaccharides (consist of sugar units), proteins (consist of amino acids), and nucleic acids (consist of nucleotides) [1]. Biopolymers are becoming more valuable as sustainable materials. They decrease dependency on non-renewable resources and reduce environmental impact [2]. Propolis affords an efficient and green strategy for adding unique properties and sustainability to material science.
Propolis is a natural substance known as bee glue. It is an elaborate mixture of gummous, resinous, and balsamic substances derived by honey bees from exudates and plant sprouts combined with bee saliva, pollen, and wax to create its final form [3]. Honey bees can collect propolis from plants like pine, poplar, conifer secretions, palm, mucilage, resins, gums, and leaf buds. Honey bees collect propolis to seal cracks and gaps presented in the hives. It was also employed to stabilize the honeycomb cells and defend against invaders and cold weather. Furthermore, honey bees use it as an antiseptic to protect their larvae, combs, and honey stores from microbial infection [4]. In addition, propolis is used to line honeycombs, assist the queen in laying eggs efficiently, and petrifact and preserve decay organisms (insects and beetles) that are not usually found in the hive [5].
Propolis has been applied recently as a food supplement and alternative medicine. Due to its wonderful properties, propolis finds vast applications in various fields such as active food packaging and wound healing applications. However, the active ingredients of propolis changed significantly with the geographical source. Moreover, the biological activity of propolis depends on plant sources, geographical zone, and seasons. Propolis contains complexes from the polyphenolic fractions, mostly flavonoids, followed by phenolic acid esters, aromatic acids, and lignans or terpenoid components [6]. In terms of their percentages, resins and vegetable balsams account for 50%, bee wax 30%, pollen 5%, essential and aromatic oils 10%, and some other organic compounds [4].
Propolis contains more than 300 compounds, such as polyphenols, sesquiterpene quinines, coumarins, essential oils, amino acids, waxes, and inorganic compounds [7]. Propolis is a lipophilic substance, i.e., in a colder environment, it showed firm, brittle characteristics while it showed a soft, flexible, and very sticky state in warm weather. The color consistency of propolis varies with the region and the plant source. The color of propolis changes from yellowish-green to dark brown according to the location, tropical forests, savannah, coastal, desert, and mountainous regions, where it is produced [7]. It was harvested from the beehives manually during the dry season (May–July). After collection, propolis was dried in the dark and reserved at a temperature of 4 °C until its processing [5].
Moreover, it has numerous biological properties, such as antibacterial, antifungal, antiviral, anti-inflammatory, anticancer, and antitumoral [8, 9]. Flavonoids, the main active compound in propolis, are responsible for propolis’ antimicrobial property, which is effective against various Gram-positive and Gram-negative bacteria [10]. Figure 1 shows some of the flavonoid structures isolated from propolis. Some authors review the carbohydrate composites for packaging [11] or wound healing applications [12], while others review propolis for that purpose [13]. This review emphasizes the various composites prepared from both propolis with various polysaccharides.
2 Propolis as appropriate material for medical treatment and packaging
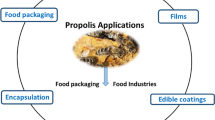
Propolis possesses miscellaneous pharmaceutical properties and has been utilized in folk medicines such as health foods and bio-cosmetics [14]. The health benefits of propolis usage are tremendous, and it is used in various ways. For example, mouthwashes and toothpaste with propolis extract effectively prevent microbial infection and reduce gum inflammation. Moreover, bioflavonoids, arginine, provitamin A, B complex, vitamin C, and some minerals have wound healing properties and enhance injury cure [15]. Finally, developing new compounds from propolis is crucial in controlling different pathogenic diseases [4]. Figure 2 demonstrates the application of polysaccharides/propolis composites for wound healing and food packaging.
2.1 Propolis as wound healing material
In recent decades, biomaterials have become increasingly popular in healthcare applications, especially wound dressing. These materials’ have biocompatibility, biodegradability, and renewable nature. Burn wounds can be treated more quickly with propolis because it contains many compounds that exhibit some biological properties. The property of propolis allows it to promote skin cell proliferation and activation and increase growth capacity in burn cases [16]. The biological activity of propolis mainly originated from the antioxidative property of polyphenols [17].
Moreover, propolis can quench free radicals in the skin and enhances the repair of burned tissue by the motivation of the wound bed template altering due to the aptitude of its flavonoid compounds to diminish lipid peroxidation and avoid cell necrosis [18,19,20]. Propolis reveals immunomodulatory and immunostimulatory effects in vitro on macrophages, while in vivo increases the ratio of CD4/CD8 T cells in rats. It improves the wound healing speed and epithelialization of diabetic wounds in rodents [21]. The healing properties of propolis originated from its immune-stimulating effect without side effects [22].
Few studies investigated the antibacterial effect of propolis alongside clinical wound isolates [23]. Among these trials, propolis prevents anaerobic bacteria isolated from wounds [24]. Moreover, the ethanolic extract displayed synergism with several antibiotics, for instance, those that hinder the synthesis of bacterial protein [25]. Some propolis extract components, such as flavonoids and benzoic acid, caffeic acid, and cinnamic acid, probably act on cell walls, causing structural and functional damage. Propolis’ ability to prevent biofilm formation or interrupt the already formed biofilm has been considered in dental or oral biofilms [26]. The positive effects of propolis on connective tissue fibroblasts, verifying its promising role in wound healing. Propolis enhances skin cell proliferation and growth capacity. It could stimulate favorable biochemical surroundings leading to re-epithelization [27].
2.2 Propolis for food packaging
Functional bio-based packaging materials have been developed to improve food quality and safety. Propolis is highly likely to be applied as an alternative biodegradable active food package with excellent natural antioxidants and antibacterial [28,29,30]. Promising studies on the integration of propolis to improve the packaging films have been displayed. The preparation of functional edible coatings is a reasonable choice to manage the growth of phytopathogens in foods throughout the postharvest shelf life. Recently, natural extracts, such as propolis, for food preservation have paid growing attention to preparing active edible coatings to control fungal pathogens [6].
Propolis has antimicrobial and antioxidant properties and is natural and safe to be used as a food preservative [31,32,33,34]. However, consumers’ satisfactoriness of foods preserved using propolis as a constituent is inadequate due to its bitterness which provides strong and unpleasant flavors [31]. Raw propolis is not appropriate as a food product due to the poor water solubility and high contaminants. Consequently, propolis should be purified via solvent extraction [35]. Encapsulation of propolis might be a hopeful method to produce standardized active release systems to preserve propolis polyphenols active until use [36]. Using propolis extract in food protection is inadequate due to its volatile aroma compounds, producing exclusive flavor and odor. Therefore, it is essential to manage the propolis concentration that can be inserted with no alterations in the sensory character of the food products. Edible coatings were developed and used to extend the shelf life. The incorporation of propolis affects the film’s mechanical properties, producing more deformable edible and flexible films, reducing film transparency [6].
3 Polysaccharides/propolis composites
This section will summarize various polysaccharides/propolis composites from the perspective of their potential for multiple applications. Hydrogen bonding observed between the polysaccharide and propolis helps elevate films’ physicochemical and biological properties.
3.1 Cellulose/propolis composite
Cellulose, the most abundant biopolymer from forest or agricultural biomasses, has emerged as a substitute for fossil fuel–based polymers [37]. Plants usually contain cellulose combined with lignin and other polysaccharides. Moreover, it can also be produced from bacteria, fungi, algae, and marine invertebrates [29, 30]. It is a polysaccharide with a linear polymer chain of D-glucopyranose rings connected via β-1,4-glycosidic links [38]. Cellulose fibers are characterized by high mechanical strength explained by their hierarchical structure and by hydrogen bridging at the fiber surface [39]. Due to its intrinsic features, cellulose is suitable for producing sustainable films and hydrogels, which can be applied in biomedical and adsorption applications [40, 41]. Bacterial cellulose is a talented safe fabric for wound healing. Carboxymethylation can modify cellulose, resulting in a linear anionic polymer with a carboxyl substitution, known as carboxymethyl cellulose (CMC) [42]. Cellulose’s acetate derivatives can be dissolved in acetone and some other solvents, whereas sodium carboxymethyl cellulose, ethyl hydroxyethyl cellulose, and methylcellulose are soluble in water. The term “nanocellulose materials” describes the variety of cellulose structures with fibers on the nanometer scale. The unique properties of nanocellulose, such as enhanced crystallinity, high surface area, rheological properties, alignment and orientation, biodegradability, biocompatibility, and low toxicity, are interesting for biomedical applications [43].
Promising efforts have been achieved to mix propolis with polysaccharides to prepare a perfect dressing for treating infected wounds and encouraging wound healing. Cellulose acetate (CA) and polycaprolactone (PCL) were investigated to prepare nanofibrous electrospinning for wound dressing, as shown in Fig. 3. The propolis-impregnated CA/PCL mats offered an elevated antioxidant activity, antibacterial activity, and a suitable surface for wound healing [44]. Furthermore, new sustainable cellulose-based film-loaded propolis was reported as a novel therapeutic technique to hasten diabetic wound healing. For example, propolis and/or vitamin C were encapsulated in the cellulose/PVA composite film. The results showed that the films facilitate the vitamin C release in a controlled way [45]. In another trial, electrospinning of cellulose acetate solution overloaded with propolis was evaluated. The results showed that the nanofibers are efficient inhibitors of the growth of S. aureus and E. coli. Thus, the attained composite can be used as a wound healing mat and antibacterial drug. Moreover, the in vitro release of propolis from CA nanofibers is continuous and controlled in neutral mediums [46].
An overview of the electrospinning process for fabrication of CA/PCL NFs (A) impregnated with propolis (B) [44]
In a recent trial, casting and freeze-thawing were used to prepare PVA-NaCMC hydrogels with propolis. A concentration of 15% of propolis in the samples inhibited the pathogenic bacteria by 80%. The merged gels of PVA and NaCMC and propolis release show the facility to merge critical features needed for promising wound healing [47]. Polylactic acid (PLA) and cellulose nanocrystal (CNC) films were prepared and mixed with Tanacetum balsamita essential oil and propolis extract via solvent casting method as a platform for biodegradable active packaging. This study proved the antimicrobial properties of attained films and their capability to increase vacuum-packed cooked sausages’ shelf life by 50 days of refrigerated storage [48]. Edible films were prepared using hydroxypropyl methyl cellulose and various concentrations of propolis extract. The propolis enhanced the water vapor permeability and produced less flexible, more rigid, and deformable films. Integration of propolis into hydroxypropyl methyl cellulose edible films enhanced the antifungal properties and consequently will be promising to organize the fungal decay in food products [17].
3.2 Chitosan/propolis composite
Chitin is the most plentiful natural polymer, following cellulose. The main difference compared to cellulose is the presence of an acetamide group as a substitute for a hydroxyl group at the C2 position of the glucose unit. Chitosan is a linear amino polysaccharide resulting from chitin through the deacetylation method [49, 50]. It is a copolymer composed of D-glucosamine and N-acetyl-D-glucosamine units with two hydroxyl groups and one amino group in each repeating glycosidic unit [51]. Chitosan comprises primary amino (NH2) and is soluble in dilute acid solutions [52, 53]. Chitosan has been widely employed in drug delivery systems, wound healing, and tissue engineering [54]. Antimicrobial properties and non-toxicity are critical features of chitosan [55]. Besides its biodegradability, chitosan is hemostatic, anticholestermic, anticarcinogenic, and fungistatic. Additionally, chitosan is a pH-sensitive polymer, reveals gel-forming properties, and has non-immunogenic and mucus adhesion properties. Chitosan’s pH sensitivity can be explained by protonation and deprotonation of the NH2 groups that enhance interactions with polyanions, for instance, sulfate, citrate, and tripolyphosphate, creating a robust cross-linked matrix [56].
The formation of polyelectrolyte complexes is possible when polyanionic polymers interact with protonated NH2 groups. Further, protonation of the NH2 groups in chitosan enhanced mucoadhesion, thus causing a prolonged residence time for chitosan at the site of administration, as well as enhancing the permeation of the drug and controlling its release. Body enzymes also break down chitosan, allowing it to be excreted safely [57].
Many polymers can adhere to mucosal tissues by hydrogen bonding or noncovalent electrostatic attractions [58]. Thus, polymeric micelles are believed as nano particulates that integrate drugs by physical entrapment and are distinguished by superior absorption and stability in biological solutions.
Chitosan films containing different propolis extract percentages were prepared. The results showed oxygen, and water vapor permeability decreased with a rising propolis percentage. The prepared chitosan/propolis films can be used as active food packaging. Chitosan is a hydrophilic edible coating with potent antioxidant activities and antimicrobial against free radicals and foodborne pathogens [59].
Chitosan-coated polypropylene containing propolis extract was assessed against six foodborne pathogens. The results established that the prepared films have broad-spectrum antibacterial activity. These results prove that chitosan coating on synthetic polymer films is a convenient method of generating antimicrobial packaging materials, especially for moisture-containing foods. Propolis is a hopeful natural antimicrobial that can be added to biopolymer-based coatings and films [60]. Propolis can be extracted as a dispensable powder in aqueous and alcohol mediums with excellent antioxidant properties and storage stability. The antimicrobial efficiency of propolis is vital to organizing fruit quality losses.
For this reason, chitosan/propolis composite was applied as the coating substance. The results displayed a diminished deterioration rate and lowered infection with fungus and thus elongated the shelf life of the fruit by 48 h [61]. The propolis with high polyphenols prepared films showed increased tensile strength, elongation at break, and antioxidant activity due to the propolis addition. Chitosan and propolis can be blended further to decrease water vapor/oxygen permeability, increasing their potential as active materials for food packaging [33]. The blend of chitosan coating enriched with Zataria multiflora essential oil and propolis extract on the properties of poultry meat was studied. According, the use of chitosan enriched with propolis and oil had a strong antioxidant and antibacterial effect on some bacteria. It could improve chicken breast meat’s characteristics and properties, including color, smell, taste, texture, and total acceptability. This mixture has been revealed to increase the shelf life of chicken breast meat by about 16 days at 4 °C and may potentially be used as active packaging material [62]. The antioxidant and antimicrobial activities of chitosan coating propolis were reviewed [63]. In addition, the antioxidant and antimicrobial effects of mixing propolis and chitosan against lipid oxidation and food spoilage microorganisms have been explored [64]. The edible coating is a thin layer of edible material that offers protection and a barrier to the fruit, which improves its shelf life, preserves the fruit against pathogens, and delays spoilage. For example, edible propolis-chitosan nanoparticle films were investigated as a competent substitute for active packaging with antibacterial properties to preserve strawberries against foodborne bacteria. The addition of nanoparticles improved the mechanical properties of edible coatings, and the shelf life of food was enhanced by incorporating antimicrobial agents such as chitosan or propolis [65]. Therefore, propolis has been integrated into coatings or films to diminish spoilage and extend the fruit and vegetable shelf life [66]. For example, integrating propolis into chitosan coatings enhanced the safety of fresh-cut broccoli [67]. Chitosan films containing different proportions of propolis extract were developed. The film’s mechanical and barrier properties were enhanced by adding the propolis extract [33]. Improved aqueous solubility and controlled release of propolis were achieved by formulation into solid dispersion matrices and polymeric nanoparticles; subsequently, spray drying added it into chitosan microparticles [68].
3.3 Starch/propolis composite
Cereal grains and root vegetables are common sources of starch. Starch typically consists of two polysaccharides, amylose (a linear chain with few branches) and amylopectin (a highly branched chain). Each starch component has a different effect on the starch’s properties. For example, most amylose chains are shorter than those in amylopectin [69].
Starch materials with remarkable physical and chemical characteristics made it possible for various applications. Biodegradable, edible films based on starch and propolis have been developed for packaging application technology; propolis nanoparticles may act as a natural bio-plasticizer [70,71,72]. Moreover, the starch-modified chemistry and many reactive sites can hold biologically active compounds. Propolis extract blends with starch and hyaluronic acid were prepared using solvent casting. As shown in Fig. 4, cornstarch/hyaluronic film dressings displayed a rough surface. In addition, an increase in propolis extract concentration increased films’ surface roughness. The results showed that incorporating hyaluronic and propolis caused an insignificant effect on the films’ porosity, but considerable differences were seen in the roughness of the film’s surface. According to the cross-sectional SEM micrographs (Fig. 4), the incorporation of hyaluronic acid and propolis extract did not affect the porosity of the films. The film containing 0.5% propolis showed higher antibacterial activity and no cytotoxicity. It is suggested as a wound dressing material that could successfully accelerate the wound healing process [73]. Active blends from carrageenan and starch-containing natural antioxidants were prepared. New functional films with antioxidant properties are made with yerba mate and Cuban red propolis extracts. The design of polysaccharide blend for the release of the bioactive compound is a promising method to avoid the spoilage of food products [74].
SEM micrographs of the surface (top) and cross-section (down) of cornstarch (A), cornstarch/hyaluronic acid (B), cornstarch/hyaluronic acid with various ethanol extract propolis 0.25% (C), 0.5% (D) and 1% (E) film dressings [73]
3.4 Alginate propolis composite
Alginate is a natural anionic polysaccharide found in brown marine algae. Alginate is composed of α-l-guluronic and β-d-mannuronic acid residues coupled with 1,4-glycosidic links. It has been extensively used for biomedical applications because of its security, biocompatibility, and relatively low cost.
The use of crude propolis as food additives is limited; it is merely feasible to benefit from propolis by preparing its extract. However, propolis contains several bioactive compounds, including flavonoids, phenols, aldehydes, and ketones. Due to the alcohol solubility, intense flavor, and aroma, these materials are not suitable as food additives [75]. Encapsulation is a sensible approach to rising above these problems. Propolis extract was encapsulated using sodium-alginate. Beads were simulated in gastric and intestinal systems to measure their release properties. Microencapsulation is a way to trap compounds inside a material to immobilize, construct, and function. The core of a microparticle is composed of bioactive components, like propolis or herbal extracts, and the shell is made up of other ingredients.
Microcapsules must have the ability to release their core material in the right place and at the right time. Some articles describe the encapsulation of propolis extracts using biopolymers as an encapsulating agent, such as casein, gelatine, and chitosan. Encapsulation efficiency of the propolis extract was reported not to exceed 80% with these techniques. The ionic gelation method is simple and inexpensive, and does not require toxic solvents. As a result, this method could encapsulate propolis ethanol extract without using toxic solvents while also achieving high encapsulation effectiveness [76]. The microencapsulation of active ingredients such as propolis extract with sodium alginate was studied. The obtained capsules offer propolis consumption in a powder form without alcohol. Also, the capsules could find application as food constituents in the food industry [77]. The ionic gelation technique organized alginate beads with and without propolis extract using calcium chloride solution to prepare propolis-alginate and alginate beads [78]. Propolis’ bioactive components reached the upper part of the large intestine based on the release properties of the beads. The subsequent increase in bioavailability of these components may also be related to this situation. Beads may be consumed as dietary supplements or used as food additives in the food industry [79].
Alginate beads encapsulated with jabuticaba peel and propolis extracts were developed as a new antioxidant colorant. The encapsulation of anthocyanins and phenolic compounds obtained from jabuticaba peel fruit and propolis was studied to evaluate its significant potential advantages. Alginate was encapsulated via ionotropic gelation by soaking the alginate in the CaCl2 solution. This study demonstrates that encapsulation of anthocyanins (from jabuticaba) and phenolic compounds (from propolis extracts) can be considerably continued onto alginate hydrogel beads. The encapsulation offers a possible application as an oral delivery system and enables its usage as a natural healthy food/beverage colorant additive. In addition to being rich in essential oils and phenolic compounds, propolis has been reported to have high antibacterial, antioxidant, anti-inflammatory, and antitumor properties [80]. Besides being useful for food preservation, it also benefits human health [81]. Although propolis contains numerous bioactive compounds, its application as a food additive is restricted by its low oral bioavailability. An efficient approach to conserving the beneficial health properties of natural sources is integrating their extracts, rich in polyphenols, into polymeric matrices to enhance stability and bioavailability; moreover, this shield can cover its strong odors or harsh flavors. The encapsulation can develop the release methods and offer the extended and controlled release of food constituents [82].
To enhance propolis bio-accessibility, a one-step procedure was used to fabricate nanoparticle composite using propolis-loaded zein/caseinate/alginate without any organic solvents. As a result, propolis-loaded zein/caseinate/alginate nanoparticles have encapsulation efficiency and loading capacity of 86.5% and 59.6 μg mg−1, respectively. These results showed a promising green approach to encapsulating hydrophobic nutraceuticals for food and pharmaceutical applications [83].
In wound dressing, propolis containing wound dressing patches was prepared using the combination of propolis and sodium alginate using 3D printing technology. The highest mechanical and the desired morphological and biological properties were observed by mixing 4.5 wt % sodium alginate and 20 v/v % propolis. Moreover, at this concentration, the scaffold pore size was 172 µm, a suitable pore size for skin tissue engineering, i.e., the actual pore size of fibroblast cells binding for wound healing. The antimicrobial results showed that the 3D-printed scaffolds have promising for wound dressing applications [84].
3.5 Miscellaneous polysaccharides/propolis composite
The unique properties of propolis encourage the researcher to investigate the promising applications of polysaccharides/propolis material for biomedical applications. In addition to being glucose-based, dextran is composed of α-1,6-glucopyranosidic linkages. Dextran is produced by Leuconostoc mesenteroides bacteria. The dextransucrase enzyme is responsible for converting sucrose into dextran [85, 86].
A delivery system and wound dressing were carried out based on carrageenan (κ-CAR) and β-cyclodextrin (β-CD) to encapsulate propolis extract. As a cross-linking agent, the authors applied glyoxal to improve the insolubility of the propolis-loaded hydrogel to fit the cotton fabric handling. Water was retained the best in the neutral medium when 1.5% glyoxal, as a cross-linking agent, and 4% propolis were used.
Moreover, the in vitro release figure of propolis from κ–CAR/β-CD hydrogel was explored, and the results showed that the hydrogel swelling behavior significantly affects the propolis release. Also, the results showed that the antimicrobial actions of the attained hydrogel were improved by increasing the encapsulated propolis concentration. However, the preparation of hydrogel holding hydrophobic compounds such as propolis has various difficulties as the incompatibility of the hydrophobic drug with hydrophilic organic polymers. As a result, multiple trials were carried out to overcome these problems. Promising practices suggested that a semi-interpenetrating network of biodegradable blends assists the formation of synergistic gels [87].
The antibacterial and biocompatible polysaccharides-based scaffold with competent wound healing action can be suitable for wound dressing application. The polyurethane and hyaluronic acid nanofibrous wound dressing was prepared and enriched with propolis extracts (Fig. 5). The authors proved that 1 wt% propolis supplemented polyurethane and hyaluronic acid nanofibrous scaffold can be an applicant with extensive wound healing, biocompatibility, and antibacterial activities for additional biomedical applications [88]. Furthermore, propolis was encapsulated in Arabic gum and maltodextrin microcapsules. It was successfully encapsulated a-tocopherol and propolis in sunflower oil droplets stabilized by WPI and WPI-alginate [68].
Schematic illustration of the polyurethane-hyaluronic/ethanolic extract of propolis scaffold synthesis through electrospinning [88]
4 Conclusion
According to the current study, propolis has antimicrobial, anti-inflammatory, and immunomodulatory properties that help wounds heal and tissues regenerate. Moreover, enrichment of polysaccharides’ wound dressings with propolis will significantly improve their potential efficacy as wound dressing material. In addition, the electrospun mats produced from cellulose-based composites can have a prospective application due to their sustained release of active propolis from the nanofiber mat to be used as wound healing mats and antibacterial drugs.
Propolis can be incorporated into packaging films as a source of polyphenols with numerous biological activities. Propolis is considered a functional compound in medicine, cosmetics, foodstuffs, and food packaging owing to its antibacterial, antifungal, antioxidant, anti-inflammatory, and anesthetic properties. Effective collaboration between research institutes and industry is essential to overcome the difficulties of implementing this technology and encourage the consumers to green products. The recent advances discussed in this review can provide wound healing and packaging scientists with a better understanding of propolis’ probable and benefits, therefore, support in accelerating their commercial adoption.
References
Shamshina JL, Berton P, Rogers RD (2019) Advances in functional chitin materials: a review. ACS Sustain Chem Eng 7:6444–6457. https://doi.org/10.1021/acssuschemeng.8b06372
Salama A, Hesemann P (2020) Recent trends in elaboration, processing, and derivatization of cellulosic materials using ionic liquids. ACS Sustain Chem Eng 8:17893–17907. https://doi.org/10.1021/acssuschemeng.0c06913
Ghisalberti EL (1979) Propolis: a review. Bee World 60:59–84. https://doi.org/10.1080/0005772X.1979.11097738
Anjum SI, Ullah A, Khan KA, Attaullah M, Khan H, Ali H, Bashir MA, Tahir M, Ansari MJ, Ghramh HA, Adgaba N, Dash CK (2019) Composition and functional properties of propolis (bee glue): a review, Saudi. J Biol Sci 26:1695–1703. https://doi.org/10.1016/j.sjbs.2018.08.013
Alsayed MFS, Hashem A, Al-Hazzani AA, Abd_Allah EF (2020) Biological control of yeast contamination of industrial foods by propolis, Saudi. J Biol Sci 27:935–946. https://doi.org/10.1016/j.sjbs.2020.01.023
Moreno MA, Vallejo AM, Ballester AR, Zampini C, Isla MI, López-Rubio A, Fabra MJ (2020) Antifungal edible coatings containing Argentinian propolis extract and their application in raspberries. Food Hydrocoll 107. https://doi.org/10.1016/j.foodhyd.2020.105973
Piccinelli AL, Lotti C, Campone L, Cuesta-Rubio O, Campo Fernandez M, Rastrelli L (2011) Cuban and Brazilian red propolis: botanical origin and comparative analysis by high-performance liquid chromatography–photodiode array detection/electrospray ionization tandem mass spectrometry. J Agric Food Chem 59:6484–6491. https://doi.org/10.1021/jf201280z
Falcão SI, Vilas-Boas M, Estevinho LM, Barros C, Domingues MRM, Cardoso SM (2010) Phenolic characterization of Northeast Portuguese propolis: usual and unusual compounds. Anal Bioanal Chem 396:887–897. https://doi.org/10.1007/s00216-009-3232-8
El-Sheikh SMA, Abd El-Alim AE-AF, Ibrahim HA, Mobarez EA, El-Sayed WA, Galal AAA, Awad NFS (2021) Chitosan propolis nanocomposite alone or in combination with apramycin: an alternative therapy for multidrug-resistant Salmonella Typhimurium in rabbits: in vitro and in vivo study. J Med Microbiol 70:1412. https://doi.org/10.1099/jmm.0.001412
Cui J, Duan X, Ke L, Pan X, Liu J, Song X, Ma W, Zhang W, Liu Y, Fan Y (2022) Extraction, purification, structural character and biological properties of propolis flavonoids: a review. Fitoterapia 157:105106. https://doi.org/10.1016/j.fitote.2021.105106
Nechita P, Roman M (2020) (Iana-Roman), Review on polysaccharides used in coatings for food packaging papers. Coatings 10:566. https://doi.org/10.3390/coatings10060566
Alves P, Gonçalves F, Gil MH (2021) Polysaccharide-based composites for biomedical applications. 19–34. https://doi.org/10.1007/978-981-33-4753-3_2.
Pahlevanneshan Z, Deypour M, Kefayat A, Rafienia M, Sajkiewicz P, EsmaeelyNeisiany R, Enayati MS (2021) Polyurethane-nanolignin composite foam coated with propolis as a platform for wound dressing: synthesis and characterization. Polymers (Basel) 13:3191. https://doi.org/10.3390/polym13183191
Miłek M, Ciszkowicz E, Tomczyk M, Sidor E, Zaguła G, Lecka-Szlachta K, Pasternakiewicz A, Dżugan M (2022) The study of chemical profile and antioxidant properties of poplar-type Polish propolis considering local flora diversity in relation to antibacterial and anticancer activities in human breast cancer cells. Molecules 27:725. https://doi.org/10.3390/molecules27030725
Dumitru CD, Neacsu IA, Grumezescu AM, Andronescu E (2022) Bee-derived products: chemical composition and applications in skin tissue engineering. Pharmaceutics 14:750. https://doi.org/10.3390/pharmaceutics14040750
De Almeida EB, Cordeiro Cardoso J, Karla De Lima A, De Oliveira NL, De Pontes-Filho NT, Oliveira Lima S, Leal Souza IC, De Albuquerque-Júnior RLC (2013) The incorporation of Brazilian propolis into collagen-based dressing films improves dermal burn healing. J Ethnopharmacol 147:419–425. https://doi.org/10.1016/j.jep.2013.03.031
Pastor C, Sánchez-González L, Cháfer M, Chiralt A, González-Martínez C (2010) Physical and antifungal properties of hydroxypropylmethylcellulose based films containing propolis as affected by moisture content. Carbohydr Polym 82:1174–1183. https://doi.org/10.1016/j.carbpol.2010.06.051
Oryan A, Alemzadeh E, Moshiri A (2018) Biomedicine & Pharmacotherapy Potential role of propolis in wound healing : biological properties and therapeutic activities. Biomed Pharmacother 98:469–483. https://doi.org/10.1016/j.biopha.2017.12.069
Rojczyk E, Klama-bary A, Wojciech L (2020) Historical and modern research on propolis and its application in wound healing and other fields of medicine and contributions by Polish studies, 262. https://doi.org/10.1016/j.jep.2020.113159
Nader RA, Mackieh R, Wehbe R, El Obeid D, Sabatier JM, Fajloun Z (2021) Beehive products as antibacterial agents: a review. Antibiotics 10:717. https://doi.org/10.3390/antibiotics10060717
Matin G, Kargar N, Baha H (2016) Bio-monitoring of cadmium, lead, arsenic and mercury in industrial districts of Izmir, Turkey by using honey bees, propolis and pine tree leaves. Ecol Eng 90:331–335. https://doi.org/10.1016/j.ecoleng.2016.01.035
Cao XP, Chen YF, Zhang JL, You MM, Wang K, Hu FL (2017) Mechanisms underlying the wound healing potential of propolis based on its in vitro antioxidant activity. Phytomedicine 34:76–84. https://doi.org/10.1016/j.phymed.2017.06.001
Mataraci E, Dosler S (2012) In vitro activities of antibiotics and antimicrobial cationic peptides alone and in combination against methicillin-resistant Staphylococcus aureus biofilms. Antimicrob Agents Chemother 56:6366–6371. https://doi.org/10.1128/AAC.01180-12
Freitas AS, Cunha A, Oliveira R, Almeida-Aguiar C (2022) Propolis antibacterial and antioxidant synergisms with gentamicin and honey. J Appl Microbiol 132:2733–2745. https://doi.org/10.1111/jam.15440
Stepanović S, Antić N, Dakić I, Švabić-Vlahović M (2003) In vitro antimicrobial activity of propolis and synergism between propolis and antimicrobial drugs. Microbiol Res 158:353–357. https://doi.org/10.1078/0944-5013-00215
Sojka M, Horniackova M, Bucekova M, Majtan V (2018) Antibiofilm efficacy of honeybee products against wound biofilm. https://doi.org/10.1007/15695
Sforcin JM (2016) Biological properties and therapeutic applications of propolis. Phyther Res 30:894–905
Mustafa P, Niazi MBK, Jahan Z, Samin G, Hussain A, Ahmed T, Naqvi SR (2020) PVA/starch/propolis/anthocyanins rosemary extract composite films as active and intelligent food packaging materials. J Food Saf 40. https://doi.org/10.1111/jfs.12725
Suriyatem R, Auras R, Rachtanapun C, Rachtanapun P (2018) Biodegradable rice starch/carboxymethyl chitosan films with added propolis extract for potential use as active food packaging. Polymers (Basel) 10:954. https://doi.org/10.3390/polym10090954
Andre II, Arief A, Apriantini A, Jayanegara C (2022) Budiman, Antimicrobial activity of propolis extract and their application as a natural preservative in livestock products: a meta-analysis. Food Sci Anim Resour 42:280–294. https://doi.org/10.5851/kosfa.2022.e4
Rufatto LC, Luchtenberg P, Garcia C, Thomassigny C, Bouttier S, Henriques JAP, Roesch-Ely M, Dumas F, Moura S (2018) Brazilian red propolis: chemical composition and antibacterial activity determined using bioguided fractionation. Microbiol Res 214:74–82. https://doi.org/10.1016/j.micres.2018.05.003
El-Deeb A (2017) Utilization of propolis extract as a natural preservative in raw milk. J Food Dairy Sci 8:315-321. https://doi.org/10.21608/jfds.2017.38890
Siripatrawan U, Vitchayakitti W (2016) Improving functional properties of chitosan films as active food packaging by incorporating with propolis. Food Hydrocoll 61:695–702. https://doi.org/10.1016/j.foodhyd.2016.06.001
Tosi EA, Ré E, Ortega ME, Cazzoli AF (2007) Food preservative based on propolis: bacteriostatic activity of propolis polyphenols and flavonoids upon Escherichia coli. Food Chem 104:1025–1029. https://doi.org/10.1016/j.foodchem.2007.01.011
Pobiega K, Kra K (2019) Trends in food science & technology application of propolis in antimicrobial and antioxidative protection of food quality – a review. 83:53–62. https://doi.org/10.1016/j.tifs.2018.11.007
Mascheroni E, Figoli A, Musatti A, Limbo S, Drioli E, Suevo R, Talarico S, Rollini M (2014) An alternative encapsulation approach for production of active chitosan – propolis beads. 1401–1407. https://doi.org/10.1111/ijfs.12442
Salama A (2019) Cellulose/calcium phosphate hybrids: new materials for biomedical and environmental applications. Int J Biol Macromol 127. https://doi.org/10.1016/j.ijbiomac.2019.01.130
Salama A, Neumann M, Günter C, Taubert A (2014) Ionic liquid-assisted formation of cellulose/calcium phosphate hybrid materials. Beilstein J Nanotechnol 5:1553–1568. https://doi.org/10.3762/bjnano.5.167
Salama A, El-Sakhawy M (2016) Regenerated cellulose/wool blend enhanced biomimetic hydroxyapatite mineralization. Int J Biol Macromol 92:920–925. https://doi.org/10.1016/j.ijbiomac.2016.07.077
Salama A (2018) Preparation of CMC-g-P(SPMA) super adsorbent hydrogels: exploring their capacity for MB removal from waste water. Int J Biol Macromol 106:940–946. https://doi.org/10.1016/j.ijbiomac.2017.08.097
Salama A (2020) Cellulose/silk fibroin assisted calcium phosphate growth: novel biocomposite for dye adsorption. Int J Biol Macromol 165:1970–1977. https://doi.org/10.1016/j.ijbiomac.2020.10.074
Salama A, El-Sakhawy M, Kamel S (2016) Carboxymethyl cellulose based hybrid material for sustained release of protein drugs. Int J Biol Macromol 93:1647–1652. https://doi.org/10.1016/j.ijbiomac.2016.04.029
Abouzeid RE, Khiari R, Salama A, Diab M, Beneventi D, Dufresne A (2020) In situ mineralization of nano-hydroxyapatite on bifunctional cellulose nano fi ber / polyvinyl alcohol / sodium alginate hydrogel using 3D printing. Int J Biol Macromol 160:538–547. https://doi.org/10.1016/j.ijbiomac.2020.05.181
Khoshnevisan K, Maleki H, Samadian H, Doostan M, Khorramizadeh MR (2019) Antibacterial and antioxidant assessment of cellulose acetate/polycaprolactone nanofibrous mats impregnated with propolis. Int J Biol Macromol 140:1260–1268. https://doi.org/10.1016/j.ijbiomac.2019.08.207
Voss GT, Gularte MS, Vogt AG, Giongo JL, Vaucher RA, Echenique JVZ, Soares MP, Luchese C, Wilhelm EA, Fajardo AR (2018) Polysaccharide-based film loaded with vitamin C and propolis: a promising device to accelerate diabetic wound healing. Int J Pharm 552:340–351. https://doi.org/10.1016/j.ijpharm.2018.10.009
El-naggar SSME (2018) Eco-friendly technology for preparation, characterization and promotion of honey bee propolis extract loaded cellulose acetate nanofibers in medical domains. Cellulose 25:5211–5220. https://doi.org/10.1007/s10570-018-1921-1
Oliveira RN, Moreira APD, da Silva Moreira Thiré RM, Quilty B, Passos TM, Simon P, Mancini MC, McGuinness GB (2017) Absorbent polyvinyl alcohol-sodium carboxymethyl cellulose hydrogels for propolis delivery in wound healing applications. Polym Eng Sci 57:1224–1233.https://doi.org/10.1002/pen.24500
Khodayari M, Akhondzadeh A, Khanjari A, Misaghi A, Kamkar A (2019) E ff ect of poly ( lactic acid ) fi lms incorporated with di ff erent concentrations of Tanacetum balsamita essential oil, propolis ethanolic extract and cellulose nanocrystals on shelf life extension of vacuum-packed cooked sausages, Food Packag. Shelf Life 19:200–209. https://doi.org/10.1016/j.fpsl.2018.11.009
Salama A, Hesemann P (2018) New N-guanidinium chitosan/silica ionic microhybrids as efficient adsorbent for dye removal from waste water. Int J Biol Macromol 111:762–768. https://doi.org/10.1016/j.ijbiomac.2018.01.049
Salama A, Hesemann P (2018) Synthesis of N-Guanidinium-chitosan/silica hybrid composites: Efficient adsorbents for anionic pollutants. J Polym Environ 26:1986–1997. https://doi.org/10.1007/s10924-017-1093-3
Salama A (2018) Chitosan based hydrogel assisted spongelike calcium phosphate mineralization for in-vitro BSA release. Int J Biol Macromol 108:471–476. https://doi.org/10.1016/j.ijbiomac.2017.12.035
Salama A, Hesemann P (2020) Synthesis and characterization of N -guanidinium chitosan / silica ionic hybrids as templates for calcium phosphate mineralization. Int J Biol Macromol 147:276–283. https://doi.org/10.1016/j.ijbiomac.2020.01.046
Salama A, Shoueir KR, Aljohani HA (2017) Preparation of sustainable nanocomposite as new adsorbent for dyes removal. Fibers Polym 18:1825–1830. https://doi.org/10.1007/s12221-017-7396-0
Salama A, El-Sakhawy M (2014) Preparation of polyelectrolyte/calcium phosphate hybrids for drug delivery application. Carbohydr Polym 113:500–506. https://doi.org/10.1016/j.carbpol.2014.07.022
Salama A, Hasanin M, Hesemann P (2020) Synthesis and antimicrobial properties of new chitosan derivatives containing guanidinium groups. Carbohydr Polym 241:116363. https://doi.org/10.1016/j.carbpol.2020.116363
Li H, Wang J, Luo Y, Bai B, Cao F (2022) pH-responsive eco-friendly chitosan–chlorella hydrogel beads for water retention and controlled release of humic acid. Water 14:1190. https://doi.org/10.3390/w14081190
Hamman JH (2010) Chitosan based polyelectrolyte complexes as potential carrier materials in drug delivery systems. Mar Drugs 8:1305–1322. https://doi.org/10.3390/md8041305
Upadhyaya L, Singh J, Agarwal V, Pandey AC, Verma SP, Das P, Tewari RP (2014) In situ grafted nanostructured ZnO/carboxymethyl cellulose nanocomposites for efficient delivery of curcumin to cancer. J Polym Res 21:550. https://doi.org/10.1007/s10965-014-0550-0
Miteluț AC, Popa EE, Drăghici MC, Popescu PA, Popa VI, Bujor O-C, Ion VA, Popa ME (2021) Latest developments in edible coatings on minimally processed fruits and vegetables: a review. Foods 10:2821. https://doi.org/10.3390/foods10112821
Torlak E (2013) Antibacterial effectiveness of chitosan – propolis coated polypropylene films against foodborne pathogens. 60:52–55. https://doi.org/10.1016/j.ijbiomac.2013.05.013
Barrera E, Gil J, Restrepo A, Mosquera K, Durango D (2015) A coating of chitosan and propolis extract for the postharvest treatment of papaya (Carica papaya L. cv. Hawaiiana). Rev Fac Nac Agron Medellín 68:7667–7678. https://doi.org/10.15446/rfnam.v68n2.50982
Mehdizadeh T, MojaddarLangroodi A (2019) Chitosan coatings incorporated with propolis extract and Zataria multiflora Boiss oil for active packaging of chicken breast meat. Int J Biol Macromol 141:401–409. https://doi.org/10.1016/j.ijbiomac.2019.08.267
Duman M, Özpolat E (2015) Effects of water extract of propolis on fresh shibuta (Barbus grypus) fillets during chilled storage. Food Chem 189:80–85. https://doi.org/10.1016/j.foodchem.2014.08.091
Piedrahíta Márquez DG, Fuenmayor CA, Suarez Mahecha H (2018) Effect of chitosan‐propolis edible coatings on stability of refrigerated cachama (Piaractus brachypomus) vacuum‐packed fish fillets. Packag Technol Sci. pts.2422. https://doi.org/10.1002/pts.2422
Rai M, Ingle AP, Gupta I, Pandit R, Paralikar P, Gade A, Chaud MV, dos Santos CA (2019) Smart nanopackaging for the enhancement of food shelf life. Environ Chem Lett 17:277–290. https://doi.org/10.1007/s10311-018-0794-8
Yang S, Zhou Y, Ye J, Fan G, Peng L, Pan S (2016) Effects of poplar buds as an alternative to propolis on postharvest diseases control of strawberry fruits. J Sci Food Agric 96:2136–2141. https://doi.org/10.1002/jsfa.7329
Alvarez MV, Ponce AG, Moreira M del R (2013) Antimicrobial efficiency of chitosan coating enriched with bioactive compounds to improve the safety of fresh cut broccoli. LWT - Food Sci Technol 50:78–87. https://doi.org/10.1016/j.lwt.2012.06.021.
Chang X, Feng W, He L, Chen X, Liang L (2020) Fabrication and characterisation of whey protein isolate–propolis–alginate complex particles for stabilising α-tocopherol-contained emulsions. Int Dairy J 109:104756. https://doi.org/10.1016/j.idairyj.2020.104756
Zhang B, Xie F, Shamshina JL, Rogers RD, Mcnally T, Wang DK, Halley PJ, Truss RW, Zhao S, Chen L (2017) Facile preparation of starch-based electroconductive films with ionic liquid. https://doi.org/10.1021/acssuschemeng.7b00788
Pérez-Vergara LD, Cifuentes MT, Franco AP, Pérez-Cervera CE, Andrade-Pizarro RD (2020) Development and characterization of edible films based on native cassava starch, beeswax, and propolis. NFS J 21:39–49. https://doi.org/10.1016/j.nfs.2020.09.002
Cunha GF, Soares JC, de Sousa TL, Egea MB, de Alencar SM, Belisário CM, Plácido GR (2021) Cassava-starch-based films supplemented with propolis extract: physical, chemical, and microstructure characterization. Biointerface Res Appl Chem. 11:12149–12158. https://doi.org/10.33263/BRIAC114.1214912158
Villalobos K, Rojas H, González-Paz R, Granados DB, González-Masís J, Baudrit JV, Corrales-Ureña YR (2017) Production of starch films using propolis nanoparticles as novel bioplasticizer. J Renew Mater 5:189–198. https://doi.org/10.7569/JRM.2017.634109
Eskandarinia A, Kefayat A, Ra M, Agheb M, Navid S, Ebrahimpour K (2019) Cornstarch-based wound dressing incorporated with hyaluronic acid and propolis: In vitro and in vivo studies. Carbohydr Polym 216:25–35. https://doi.org/10.1016/j.carbpol.2019.03.091
Chang-Bravo L, López-Córdoba A, Martino M (2014) Biopolymeric matrices made of carrageenan and corn starch for the antioxidant extracts delivery of Cuban red propolis and yerba mate. React Funct Polym 85:11–19. https://doi.org/10.1016/j.reactfunctpolym.2014.09.025
Busch VM, Pereyra-Gonzalez A, Šegatin N, Santagapita PR, PoklarUlrih N, Buera MP (2017) Propolis encapsulation by spray drying: characterization and stability. LWT-Food Sci Technol 75:227–235. https://doi.org/10.1016/j.lwt.2016.08.055
Gouin S (2004) Microencapsulation: industrial appraisal of existing technologies and trends. Trends Food Sci Technol 15:330–347. https://doi.org/10.1016/j.tifs.2003.10.005
Peanparkdee M, Iwamoto S, Yamauchi R (2016) Microencapsulation: a review of applications in the food and pharmaceutical industries. Rev Agric Sci 4:56–65. https://doi.org/10.7831/ras.4.56
Anbinder PS, Deladino L, Navarro AS, Amalvy JI, Martino MN (2011) Yerba mate extract encapsulation with alginate and chitosan systems: interactions between active compound encapsulation polymers. J Encapsulation Adsorpt Sci 01:80–87. https://doi.org/10.4236/jeas.2011.14011
Keskin M, Keskin Ş, Kolayli S (2019) Preparation of alcohol free propolis-alginate microcapsules, characterization and release property. Lwt 108:89–96. https://doi.org/10.1016/j.lwt.2019.03.036
Elbaz NM, Khalil IA, Abd-Rabou AA, El-Sherbiny IM (2016) Chitosan-based nano-in-microparticle carriers for enhanced oral delivery and anticancer activity of propolis. Int J Biol Macromol 92:254–269. https://doi.org/10.1016/j.ijbiomac.2016.07.024
El-Sakhawy M (2022) Propolis harvesting and extraction. Egy J Chem accepted. https://doi.org/10.21608/EJCHEM.2022.122043.5469
Dalponte Dallabona I, de Lima GG, Cestaro BI, de S. Tasso I, Paiva TS, Laureanti EJG, de M. Jorge LM, da Silva BJG, Helm CV, Mathias AL, Jorge RMM (2020) Development of alginate beads with encapsulated jabuticaba peel and propolis extracts to achieve a new natural colorant antioxidant additive. Int J Biol Macromol 163:1421–1432.https://doi.org/10.1016/j.ijbiomac.2020.07.256
Zhang H, Fu Y, Xu Y, Niu F, Li Z, Ba C, Jin B, Chen G, Li X (2019) One-step assembly of zein/caseinate/alginate nanoparticles for encapsulation and improved bioaccessibility of propolis. Food Funct 10:635–645. https://doi.org/10.1039/c8fo01614c
Aranci K, Uzun M, Su S, Cesur S, Ulag S, Amin A, Guncu MM, Aksu B, Kolayli S, Ustundag CB, Silva JC, Ficai D, Ficai A, Gunduz O (2020) 3D Propolis-sodium alginate scaffolds: influence on structural parameters, release mechanisms, cell cytotoxicity and antibacterial activity. Molecules 25:5082. https://doi.org/10.3390/molecules25215082
Robyt JF, Yoon S-H, Mukerjea R (2008) Dextransucrase and the mechanism for dextran biosynthesis. Carbohydr Res 343:3039–3048. https://doi.org/10.1016/j.carres.2008.09.012
Besrour-Aouam N, Fhoula I, Hernández-Alcántara AM, Mohedano ML, Najjari A, Prieto A, Ruas-Madiedo P, López P, Ouzari H-I (2021) The role of dextran production in the metabolic context of Leuconostoc and Weissella Tunisian strains. Carbohydr Polym 253:117254. https://doi.org/10.1016/j.carbpol.2020.117254
Đorđević V, Balanč B, Belščak-Cvitanović A, Lević S, Trifković K, Kalušević A, Kostić I, Komes D, Bugarski B, Nedović V (2015) Trends in encapsulation technologies for delivery of food bioactive compounds. Food Eng Rev 7:452–490. https://doi.org/10.1007/s12393-014-9106-7
Eskandarinia A, Kefayat A, Gharakhloo M, Agheb M, Khodabakhshi D, Khorshidi M, Sheikhmoradi V, Rafienia M, Salehi H (2020) A propolis enriched polyurethane-hyaluronic acid nanofibrous wound dressing with remarkable antibacterial and wound healing activities. Int J Biol Macromol 149:467–476. https://doi.org/10.1016/j.ijbiomac.2020.01.255
Funding
Open access funding provided by The Science, Technology & Innovation Funding Authority (STDF) in cooperation with The Egyptian Knowledge Bank (EKB). This study was financially supported by the National Research Center, Egypt, projects number 12020101 and 12010402.
Author information
Authors and Affiliations
Contributions
All authors contributed to the idea for the article, the literature search, and data analysis and drafted and critically revised the work.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Salama, A., El-Sakhawy, M. Polysaccharides/propolis composite as promising materials with biomedical and packaging applications: a review. Biomass Conv. Bioref. 14, 4555–4565 (2024). https://doi.org/10.1007/s13399-022-02814-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13399-022-02814-5