Abstract
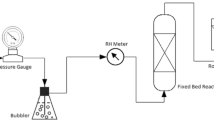
Oxidation of air-dried coal in mined-out areas is one leading factor contributing to coal spontaneous ignition during exploitation. Although some related work has been done, there is still some space for improvement. We screened out three coal samples with different ranks of coal and immersed them in distilled water for 200 days. Afterward, we identified some changes in pore structure and oxygen consumption characteristics of coal samples after water immersion. Additionally, we analyzed the composition of dissolved substance, both organic and inorganic. The results demonstrated that some parameters of three different residual coal samples increased, including average pore size, specific surface area and pore volume. In regard to the composition of dissolved substance, we identified Na+ and Cl−, which accounted for the largest proportion, along with some organic functional groups. We found that higher rank leads to a smaller total amount of dissolved substance. Additionally, we performed some experiments concerning coal oxidation rate and the amount of CO, CO2 and C2H4 generated during oxidation. The results showed that water immersion does play a significant role, along with coal rank. Considering all results and analysis, long-term water immersion is responsible for pore structure development in coal and the degree of reduction in spontaneous-ignition-proof substance, which helps to boost coal spontaneous ignition.
Similar content being viewed by others
References
Zhang Y., Wu J., Wang Y., Miao Z.Y., Si C.D., Shang X.L.: Effect of hydrothermal dewatering on the physico-chemical structure and surface properties of Shengli lignite. Fuel 164, 128–133 (2016)
Lyu Q., Ranjith P.G., Long X., Kang Y., Huang M.: A review of shale swelling by water adsorption. J. Nat. Gas Sci. Eng. 27, 1421–1431 (2015)
Zhang S., Chen Y.J., Liu D., Wang L., Mi J.X., Wang Y.G.: Hydrothermal treatment affected to pore structure and surface property of Shengli lignite. Coal Sci. Technol. 41, 117–121 (2013)
Zhang W., Yu T., Yang X., Jiang J., Zhang D.: Oxidizing gas generation rule of lignite under the condition of different water. Saf. Coal Mines 1, 75–76 (2009)
Li X., Song H., Wang Q., Meesri C., Wal L.T., Yu J.: Experimental study on drying and moisture re-adsorption kinetics of an Indonesian low rank coal. J. Environ. Sci. 21, 127–130 (2009)
Hodges D.T., Tucker J.R., Hartwick T.S.: Basic physical mechanisms determining performance of the CH3F laser. Infrared Phys. 16, 175–182 (1976)
Wang W., Wang G., Liu H.: Heat release regular pattern of different moisture content coal in low temperature. CSIRO Earth Sci. Resour. Eng. 94, 419–425 (2013)
King J., Krug D., Zepf D.: The role of oxygen complex in oxidation of carbonaceous compounds. Soc. Chem. Ind. Trans. Faraday Soc. 42, 297–299 (1964)
Jones R.E., Townend D.T.A.: The oxidation of coal. J. Soc. Chem. Ind. 68, 197–201 (1949)
Benfell K.E., Beamish B.B., Rodgers K.A.: Thermogravimetric analytical procedures for characterizing New Zealand and Eastern Australian coals. Thermochim. Acta 286, 67–74 (1996)
Banerjee S.C.: Spontaneous Combustion of Coal and Mine Fires. Balkema, Rotterdam (1985)
Wang H., Dlugogorski B.Z., Kennedy E.M.: Coal oxidation at low temperatures: oxygen consumption, oxidation products, reaction mechanism and kinetic modeling. Prog. Energy Combust. Sci. 29, 487–513 (2003)
Reich M.H., Snook I.K., Wagenfeld H.K.: A fractal interpretation of the effect of drying on the pore structure of Victorian brown coal. Fuel 71, 669–672 (1992)
Arisoy A., Akgün F.: Modelling of spontaneous combustion of coal with moisture content included. Fuel 73, 281–286 (1994)
Wang Q., Guo S., Sun J.: Oxygen and moisture effects on the thermal behavior of coal powder at elevated temperature. Fire Saf. Sci. 1, 1–5 (2009)
Pore size distribution and porosity of solid materials by mercury porosimetry and gas adsorption, Part 3: analysis of micropores by gas adsorption. ISO 15901-3 (2007)
The test method of oxidation kinetics for the propensity of coal to spontaneous combustion (AQ/T 1068-2008), China’s State Administration of Work Safety, 2008.11
Brunauer S., Emmett P.H., Teller E.: Adsorption of gases in multimolecular layers. J. Am. Chem. Soc. 60, 309–319 (1938)
Barrett E.P., Joyner L.G., Halenda P.P.: The determination of pore volume and area distributions in porous substances. I. Computations from nitrogen isotherms. J. Am. Chem. Soc. 73, 373–380 (1951)
Dubinin M.M., Radushkevich L.V.: The equation of the characteristic curve of activated charcoal. Zhurnal Nevropatologii I Psikhiatrii Imeni S.s.korsakova 7, 843–848 (1946)
Vassilev S.V., Eskenazyb G.M., Vassilevaa C.G.: Contents, modes of occurrence and origin of chlorine and bromine in coal. Fuel 79, 903–921 (2000)
Durie R.A.: The Science of Victorian Brown Coal. Butterworth-Heinemann, Oxford (1991)
Dudzińska A.: The effect of pore volume of hard coals on their susceptibility to spontaneous combustion. J. Chem. 1, 1–7 (2014)
Tang Y.B.: The influences of manganese and phosphorus on the low-temperature oxidation of coal. Int. J. Coal Prep. Util. 35, 63–75 (2015)
Beamish B.B., Arisoy A.: Effect of mineral matter on coal self-heating rate. Fuel 87, 125–130 (2008)
Yang Y.L., Li Z.H., Tang Y.B., Ji H.J., Liu Z.: Soluble organic matter affecting low-temperature oxidation of coal with different ranks. Asian J. Chem. 26, 2487–2492 (2014)
Kumagai H., Chiba T., Nakamura K.: Change in physical and chemical characteristics of brown coal along with a progress of moisture release. Am. Chem. Soc. 44, 633–636 (1999)
Zheng Q., Liu Q., Wu Z., Zang Y., Shi S.: Ammonium–illite smectite interlayer clay minerals in coal-bearing strata in Jincheng district of Shanxi Province. J. China Coal Soc. 37, 231–236 (2012)
Clarkson C.R., Bustin R.M.: Variation in micropore capacity and size distribution with composition in bituminous coal of the Western Canadian Sedimentary Basin. Fuel 75, 1483–1498 (1996)
Wang F., Cheng Y., Lu S., Jin K., Zhao W.: Influence of coalification on the pore characteristics of middle–high rank coal. Energy Fuels 28, 5729–5736 (2014)
Clarkson C.R., Bustin R.M.: The effect of pore structure and gas pressure upon the transport properties of coal: a laboratory and modeling study. 1. Isotherms and pore volume distributions. Fuel 78, 1333–1344 (1999)
Clarkson C.R., Bustin R.M.: The effect of pore structure and gas pressure upon the transport properties of coal: a laboratory and modeling study. 2. Adsorption rate modeling. Fuel 78, 1345–1362 (1999)
Ruckenstein E., Vaidyanathan A.S., Youngquist G.R.: Sorption by solids with bidisperse pore structures. Chem. Eng. Sci. 26, 1305–1318 (1971)
Yuan W., Pan Z., Li X., Yang Y., Zhao C., Connell L.D., Li S., He J.: Experimental study and modelling of methane adsorption and diffusion in shale. Fuel 117, 509–519 (2014)
Yang Y., Li Z., Tang Y., Gu F., Liu Z.: Effects of low molecular weight compounds in coal on the characteristics of its spontaneous combustion. Can. J. Chem. Eng. 93, 648–657 (2015)
Ye, B.: Research on Inhibition Mechanism and Inhibition Characteristic to Prevent the Coal Spontaneous Combustion. Liaoning Technical University, Fuxin (2006)
Zhao Q.F.: Basic Research on Coal Spontaneous Combustion Inhibition Technology. Liaoning Technical University, Fuxin (2009)
Hao, C.; Zhao, Q.; Wang, J.; Chi, G.: Effect of hydrogen bonds to form complexes for N active structures which contained in coal and the Ca-based inhibitor. Adv. Mater. Res. 881–883, 118–121 (2014)
Wang X.: Structure and Characterization of Complexes Formed of Ca2+ and Coal Active Group. Liaoning Technical University, Fuxin (2009)
Li Z.: Mechanism of free radical reactions in spontaneous combustion of coal. J. China Univ. Min. Technol. 25, 111–114 (1996)
Yang Y., Li Z., Tang Y., Liu Z., Ji H.: Fine coal covering for preventing spontaneous combustion of coal pile. Nat. Hazards 74, 603–622 (2014)
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Yang, Y., Li, Z., Si, L. et al. Study Governing the Impact of Long-Term Water Immersion on Coal Spontaneous Ignition. Arab J Sci Eng 42, 1359–1369 (2017). https://doi.org/10.1007/s13369-016-2245-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13369-016-2245-9