Abstract
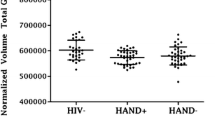
Previous studies have reported cognitive deficits among HIV-positive individuals infected with clade C virus. However, no study has examined whether individuals predominately infected with clade C virus exhibit brain atrophy relative to healthy controls. This study examined volumetric differences between 28 HIV+ individuals and 23 HIV− controls from South Africa. Volumetric measures were obtained from six regions of interest — caudate, thalamus, corpus callosum, total cortex, total gray matter, and total white matter. HIV+ participants had significantly lower volumes in the total white matter (p < 0.01), thalamus (p < 0.01) and total gray matter (inclusive of cortical and subcortical regions, p < 0.01). This study is the first to provide evidence of brain atrophy among HIV+ individuals in South Africa, where HIV clade C predominates. Additional research that integrates neuroimaging, comprehensive neuropsychological testing, genetic variance in clade-specific proteins, and the impact of treatment with Antiretrovirals (ARV) are necessary to understand the development of HIV-related neurocognitive disorders in South Africa.


Similar content being viewed by others
References
Amin J, Kaye M, Skidmore S, Pillay D, Cooper DA, Dore GJ (2004) HIV and hepatitis C coinfection within the CAESAR study. HIV Med 5:174–179
Ances BA, Ortega M, Vaida F, Heaps J, Paul R (2012) Independent effects of HIV, aging, and HAART on brain volumetric measures. JAIDS. Post acceptance 20 Jan 2012, doi:10.1097/QAI.0b013e318249db17
Clifford DB, Mitike MT, Mekonnen Y, Zhang J, Zenebe G, Melaku Z, Zewde A, Gessesse N, Wolday D, Messele T, Teshome M, Evans S (2007) Neurological evaluation of untreated human immunodeficiency virus infected adults in Ethiopia. J Neurovirol 13:67–72. doi:10.1080/13550280601169837
Constantino AA, Huang Y, Zhang H, Wood C, Zheng JC (2011) HIV-1 clade B and C isolates exhibit differential replication: relevance to macrophage-mediated neurotoxicity. Neurotox Res 20:277–288. doi:10.1007/s12640-011-9241-3
Elovaara I, Poutiainen E, Raininko R, Valanne L, Virta A, Valle SL, Lahdevirta J, Iivanainen M (1990) Mild brain atrophy in early HIV infection: the lack of association with cognitive deficits and HIV-specific intrathecal immune response. J Neurol Sci 99:121–136
Fischl B, Salat DH, Busa E, Albert M, Dieterich M, Haselgrove C, van der Kouwe A, Killiany R, Kennedy D, Klaveness S, Montillo A, Makris N, Rosen B, Dale AM (2002) Whole brain segmentation: automated labeling of neuroanatomical structures in the human brain. Neuron 33:341–355
Fischl B, Salat DH, van der Kouwe AJ, Makris N, Segonne F, Quinn BT, Dale AM (2004) Sequence-independent segmentation of magnetic resonance images. Neuroimage 23(Suppl 1):S69–S84. doi:10.1016/j.neuroimage.2004.07.016
Gandhi N, Saiyed Z, Thangavel S, Rodriguez J, Rao KV, Nair MP (2009) Differential effects of HIV type 1 clade B and clade C Tat protein on expression of proinflammatory and antiinflammatory cytokines by primary monocytes. AIDS Res Hum Retroviruses 25:691–699. doi:10.1089/aid.2008.0299
Gupta JD, Satishchandra P, Gopukumar K, Wilkie F, Waldrop-Valverde D, Ellis R, Ownby R, Subbakrishna DK, Desai A, Kamat A, Ravi V, Rao BS, Satish KS, Kumar M (2007) Neuropsychological deficits in human immunodeficiency virus type 1 clade C-seropositive adults from South India. J Neurovirol 13:195–202. doi:10.1080/13550280701258407
Hoare J, Fouche JP, Spottiswoode B, Sorsdahl K, Combrinck M, Stein DJ, Paul RH, Joska JA (2011) White-matter damage in clade C HIV-positive subjects: a diffusion tensor imaging study. J Neuropsychiatry Clin Neurosci 23:308–315. doi:10.1176/appi.neuropsych.23.3.308
Jacobs GB, Laten A, van Rensburg EJ, Bodem J, Weissbrich B, Rethwilm A, Preiser W, Englebrecht S (2008) Phylogenetic diversity and low level antiretroviral resistance mutations in HIV type 1 treatment-naive patients from Cape Town, South Africa. AIDS Res Hum Retroviruses 24:1009–1012. doi:10.1089/aid.2008.0028
Jacobs GB, Loxton AG, Laten A, Robson B, van Rensburg EJ, Engelbrecht S (2009) Emergence and diversity of different HIV-1 subtypes in South Africa, 2000–2001. J Med Virol 81:1852–1859. doi:10.1002/jmv.21609
Joska JA, Fincham DS, Stein DJ, Paul RH, Seedat S (2009) Clinical correlates of HIV-associated neurocognitive disorders in South Africa. AIDS Behav 14:371–378. doi:10.1007/s10461-009-9538-x
Joska JA, Gouse H, Paul RH, Stein DJ, Flisher AJ (2010a) Does highly active antiretroviral therapy improve neurocognitive function? A systematic review. J Neurovirol 16:101–114. doi:10.3109/13550281003682513
Joska JA, Westgarth-Taylor J, Myer L, Hoare J, Thomas KG, Combrinck M, Paul RH, Stein DJ, Flisher AJ (2010b) Characterization of HIV-associated neurocognitive disorders among individuals starting antiretroviral therapy in South Africa. AIDS Behav 15:1197–1203. doi:10.1007/s10461-010-9744-6
Kieburtz K, Ketonen L, Cox C, Grossman H, Holloway R, Booth H, Hickey C, Feigin A, Caine ED (1996) Cognitive performance and regional brain volume in human immunodeficiency virus type 1 infection. Arch Neurol 53:155–158
Lindl KA, Marks DR, Kolson DL, Jordan-Sciutto KL (2010) HIV-associated neurocognitive disorder: pathogenesis and therapeutic opportunities. J Neuroimmune Pharmacol 5:294–309. doi:10.1007/s11481-010-9205-z
Liner KJ 2nd, Ro MJ, Robertson KR (2010) HIV, antiretroviral therapies, and the brain. Curr HIV/AIDS Rep 7:85–91. doi:10.1007/s11904-010-0042-8
Patel SH, Kolson DL, Glosser G, Matozzo I, Ge Y, Babb JS, Mannon LJ, Grossman RI (2002) Correlation between percentage of brain parenchymal volume and neurocognitive performance in HIV-infected patients. AJNR Am J Neuroradiol 23:543–549
Paul R, Cohen R, Navia B, Tashima K (2002) Relationships between cognition and structural neuroimaging findings in adults with human immunodeficiency virus type-1. Neurosci Biobehav Rev 26:353–359
Ranga U, Shankarappa R, Siddappa NB, Ramakrishna L, Nagendran R, Mahalingam M, Mahadevan A, Jayasuryan N, Satishchandra P, Shankar SK, Prasad VR (2004) Tat protein of human immunodeficiency virus type 1 subtype C strains is a defective chemokine. J Virol 78:2586–2590
Rao VR, Sas AR, Eugenin EA, Siddappa NB, Bimonte-Nelson H, Berman JW, Ranga U, Tyor WR, Prasad VR (2008) HIV-1 clade-specific differences in the induction of neuropathogenesis. J Neurosci 28:10010–10016. doi:10.1523/JNEUROSCI.2955-08.2008
Riedel D, Ghate M, Nene M, Paranjape R, Mehendale S, Bollinger R, Sacktor N, McArthur J, Nath A (2006) Screening for human immunodeficiency virus (HIV) dementia in an HIV clade C-infected population in India. J Neurovirol 12:34–38. doi:10.1080/13550280500516500
Sacktor NC, Wong M, Nakasujja N, Skolasky RL, Selnes OA, Musisi S, Robertson K, McArthur JC, Ronald A, Katabira E (2005) The International HIV Dementia Scale: a new rapid screening test for HIV dementia. AIDS 19:1367–1374
Samikkannu T, Agudelo M, Gandhi N, Reddy PV, Saiyed ZM, Nwankwo D, Nair MP (2011) Human immunodeficiency virus type 1 clade B and C gp120 differentially induce neurotoxin arachidonic acid in human astrocytes: implications for neuroAIDS. J Neurovirol 17:230–238. doi:10.1007/s13365-011-0026-5
Satishchandra P, Nalini A, Gourie-Devi M, Khanna N, Santosh V, Ravi V, Desai A, Chandramuki A, Jayakumar PN, Shankar SK (2000) Profile of neurologic disorders associated with HIV/AIDS from Bangalore, South India (1989–96). Indian J Med Res 111:14–23
Shapshak P, Kangueane P, Fujimura RK, Commins D, Chiappelli F, Singer E, Levine AJ, Minagar A, Novembre FJ, Somboonwit C, Nath A, Sinnott JT (2011) Editorial neuroAIDS review. AIDS 25:123–141. doi:10.1097/QAD.0b013e328340fd42
Thompson PM, Dutton RA, Hayashi KM, Toga AW, Lopez OL, Aizenstein HJ, Becker JT (2005) Thinning of the cerebral cortex visualized in HIV/AIDS reflects CD4+ T lymphocyte decline. QProc Natl Acad Sci USA 102:15647–15652. doi:10.1073/pnas.0502548102
Thurnher MM, Donovan Post MJ (2008) Neuroimaging in the brain in HIV-1-infected patients. Neuroimaging Clin N Am 18:93–117. doi:10.1016/j.nic.2007.12.013, viii
Tucker KA, Robertson KR, Lin W, Smith JK, An H, Chen Y, Aylward SR, Hall CD (2004) Neuroimaging in human immunodeficiency virus infection. J Neuroimmunol 157:153–162. doi:10.1016/j.jneuroim.2004.08.036
Tustison NJ, Avants BB, Cook PA, Zheng Y, Egan A, Yushkevich PA, Gee JC (2010) N4ITK: improved N3 bias correction. IEEE Trans Med Imaging 29:1310–1320. doi:10.1109/TMI.2010.2046908
Yepthomi T, Paul R, Vallabhaneni S, Kumarasamy N, Tate DF, Solomon S, Flanigan T (2006) Neurocognitive consequences of HIV in southern India: a preliminary study of clade C virus. J Int Neuropsychol Soc: JINS 12:424–430
Zatz LM, Jernigan TL (1983) The ventricular–brain ratio on computed tomography scans: validity and proper use. Psychiatry Res 8:207–214
Acknowledgments
Funding supported by National Institute of Health MH065857
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Heaps, J.M., Joska, J., Hoare, J. et al. Neuroimaging markers of human immunodeficiency virus infection in South Africa. J. Neurovirol. 18, 151–156 (2012). https://doi.org/10.1007/s13365-012-0090-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13365-012-0090-5