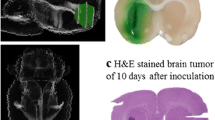
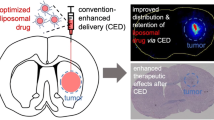
Abstract
Direct delivery of chemotherapy agents to the brain via degradable polymer delivery systems—such as Gliadel®—is a clinically proven method for treatment of glioblastoma multiforme, but there are important limitations with the current technology—including the requirement for surgery, profound local tissue toxicity, and limitations in diffusional penetration of agents—that limit its application and effectiveness. Here, we demonstrate another technique for direct, controlled delivery of chemotherapy to the brain that provides therapeutic benefit with fewer limitations. In our new approach, camptothecin (CPT)-loaded poly(lactic-co-glycolic acid) (PLGA) nanoparticles are infused via convection-enhanced delivery (CED) to a stereotactically defined location in the brain, allowing simultaneous control of location, spread, and duration of drug release. To test this approach, CPT-PLGA nanoparticles (~100 nm in diameter) were synthesized with 25% drug loading. When these nanoparticles were incubated in culture with 9L gliosarcoma cells, the IC50 of CPT-PLGA nanoparticles was 0.04 μM, compared to 0.3 μM for CPT alone. CPT-PLGA nanoparticles stereotactically delivered by CED improved survival in rats with intracranial 9L tumors: the median survival for rats treated with CPT-PLGA nanoparticles (22 days) was significantly longer than unloaded nanoparticles (15 days) and free CPT infusion (17 days). CPT-PLGA nanoparticle treatment also produced significantly more long-term survivors (30% of animals were free of disease at 60 days) than any other treatment. CPT was present in tissues harvested up to 53 days post-infusion, indicating prolonged residence at the local site of administration. These are the first results to demonstrate the effectiveness of combining polymer-controlled release nanoparticles with CED in treating fatal intracranial tumors.





Similar content being viewed by others
References
Attenello FJ, Mukherjee D et al (2008) Use of Gliadel (BCNU) wafer in the surgical treatment of malignant glioma: a 10-year institutional experience. Ann Surg Oncol 15(10):2887–2893
Blum JS, Saltzman WM (2008) High loading efficiency and tunable release of plasmid DNA encapsulated in submicron particles fabricated from PLGA conjugated with poly-L-lysine. J Control Release 129(1):66–72
Bobo RH, Laske DW et al (1994) Convection-enhanced delivery of macromolecules in the brain. Proc Natl Acad Sci USA 91(6):2076–2080
Brandsma D, van den Bent MJ (2007) Molecular targeted therapies and chemotherapy in malignant gliomas. Curr Opin Oncol 19(6):598–605
Brem H, Mahaley MS et al (1991) Interstitial chemotherapy with drug polymer implants for the treatment of recurrent gliomas. J Neurosurg 74:441–446
Chen MY, Hoffer A et al (2005) Surface properties, more than size, limiting convective distribution of virus-sized particles and viruses in the central nervous system. J Neurosurg 103(2):311–319
Deorah S, Lynch CF et al (2006) Trends in brain cancer incidence and survival in the United States: surveillance, epidemiology, and end results program, 1973 to 2001. Neurosurg Focus 20(4):E1
Ertl B, Platzer P et al (1999) Poly(D, L-lactic-co-glycolic acid) microspheres for sustained delivery and stabilization of camptothecin. J Control Release 61(3):305–317
Fahmy TM, Samstein RM et al (2005) Surface modification of biodegradable polyesters with fatty acid conjugates for improved drug targeting. Biomaterials 26(28):5727–5736
Friedman HS, Keir ST et al (2003) The emerging role of irinotecan (CPT-11) in the treatment of malignant glioma in brain tumors. Cancer 97(9 Suppl):2359–2362
Fung LK, Shin M et al (1996) Chemotherapeutic drugs released from polymers: distribution of 1, 3-bis(2-chloroethyl)-1-nitrosourea in the rat brain. Pharm Res 13(5):671–682
Fung LK, Ewend MG et al (1998) Pharmacokinetics of interstitial delivery of carmustine, 4-hydroperoxycyclophosphamide, and paclitaxel from a biodegradable polymer implant in the monkey brain. Cancer Res 58:672–684
Groothuis DR (2000) The blood-brain and blood-tumor barriers: a review of strategies for increasing drug delivery. Neuro-oncology 2(1):45–59
Hall WA, Sherr GT (2006) Convection-enhanced delivery: targeted toxin treatment of malignant glioma. Neurosurg Focus 20(4):E10
Kunwar S, Prados MD et al (2007) Direct intracerebral delivery of cintredekin besudotox (IL13-PE38QQR) in recurrent malignant glioma: a report by the Cintredekin Besudotox Intraparenchymal Study Group. J Clin Oncol 25(7):837–844
Laske DW, Morrison PF et al (1997) Chronic interstitial infusion of protein to primate brain: determination of drug distribution and clearance with single-photon emission computerized tomography imaging. J Neurosurg 87(4):586–594
Lawson HC, Sampath P et al (2007) Interstitial chemotherapy for malignant gliomas: the Johns Hopkins experience. J Neurooncol 83:61–70
Lidar Z, Mardor Y et al (2004) Convection-enhanced delivery of paclitaxel for the treatment of recurrent malignant glioma: a phase I/II clinical study. J Neurosurg 100(3):472–479
Lieberman DM, Laske DW et al (1995) Convection-enhanced distribution of large molecules in gray matter during interstitial drug infusion. J Neurosurg 82(6):1021–1029
Liu J, Jiang Z et al (2009) Poly(ω-pentadecalactone-co-butylene-co-succinate) nanoparticles as biodegradable carriers for camptothecin delivery. Biomaterials 30:5707–5719
Loh JP, Ahmed AE (1990) Determination of camptothecin in biological fluids using reversed-phase high-performance liquid chromatography with fluorescence detection. J Chromatogr 530(2):367–376
Mamot C, Nguyen JB et al (2004) Extensive distribution of liposomes in rodent brains and brain tumors following convection-enhanced delivery. J Neurooncol 68(1):1–9
Neeves KB, Sawyer AJ et al (2007) Dilation and degradation of the brain extracellular matrix enhances penetration of infused polymer nanoparticles. Brain Res 1180:121–132
Noble CO, Krauze MT et al (2006) Novel nanoliposomal CPT-11 infused by convection-enhanced delivery in intracranial tumors: pharmacology and efficacy. Cancer Res 66(5):2801–2806
O'Leary J, Muggia FM (1998) Camptothecins: a review of their development and schedules of administration. Eur J Cancer 34(10):1500–1508
Park J, Fong PM et al (2009) PEGylated PLGA nanoparticles for the improved delivery of doxorubicin. Nanomedicine 5(4):410–418
Parney IF, Chang SM (2003) Current chemotherapy for glioblastoma. Cancer J 9(3):149–156
Popperl G, Goldbrunner R et al (2005) O-(2-[18 F]fluoroethyl)-L-tyrosine PET for monitoring the effects of convection-enhanced delivery of paclitaxel in patients with recurrent glioblastoma. Eur J Nucl Med Mol Imaging 32(9):1018–1025
Raghavan R, Brady ML et al (2006) Convection-enhanced delivery of therapeutics for brain disease, and its optimization. Neurosurg Focus 20(4):E12
Rasband WS (1997–2008) ImageJ. http://rsb.info.nih.gov/ij/. Bethesda, Maryland, U. S. National Institutes of Health
Saito R, Bringas JR et al (2004) Distribution of liposomes into brain and rat brain tumor models by convection-enhanced delivery monitored with magnetic resonance imaging. Cancer Res 64(7):2572–2579
Saito R, Krauze MT et al (2005) Gadolinium-loaded liposomes allow for real-time magnetic resonance imaging of convection-enhanced delivery in the primate brain. Exp Neurol 196(2):381–389
Saito R, Krauze MT et al (2006) Convection-enhanced delivery of Ls-TPT enables an effective, continuous, low-dose chemotherapy against malignant glioma xenograft model. Neuro-Oncology 8(3):205–214
Sampath P, Amundson E et al (2003) Camptothecin analogs in malignant gliomas: comparative analysis and characterization. J Neurosurg 98(3):570–577
Sampson JH, Raghavan R et al (2007) Clinical utility of a patient-specific algorithm for simulating intracerebral drug infusions. Neuro Oncology 9(3):343–353
Sawyer AJ, Piepmeier JM et al (2006) New methods for direct delivery of chemotherapy for treating brain tumors. Yale J Biol Med 79(3–4):141–152
Shimamura T, Husain SR et al (2006) The IL-4 and IL-13 pseudomonas exotoxins: new hope for brain tumor therapy. Neurosurg Focus 20(4):E11
Storm PB, Moriarity JL et al (2002) Polymer delivery of camptothecin against 9L gliosarcoma: release, distribution, and efficacy. J Neurooncol 56(3):209–217
Stupp R, Hegi ME et al (2007) Chemoradiotherapy in malignant glioma: standard of care and future directions. J Clin Oncol 25(26):4127–4136
Tanner PG, Holtmannspotter M et al (2007) Effects of drug efflux on convection-enhanced paclitaxel delivery to malignant gliomas: technical note. Neurosurgery 61(4):E880-2, discussion E882
Tewes F, Munnier E et al (2007) Comparative study of doxorubicin-loaded poly(lactide-co-glycolide) nanoparticles prepared by single and double emulsion methods. Eur J Pharm Biopharm 66(3):488–492
Weber EL, Goebel EA (2005) Cerebral edema associated with Gliadel wafers: two case studies. Neuro-Oncology 7:84–89
Weingart JD, Thompson RC et al (1995) Local delivery of the topoisomerase I inhibitor camptothecin sodium prolongs survival in the rat intracranial 9L gliosarcoma model. Int J Cancer 62(5):605–609
Westphal M, Hilt DC et al (2003) A phase 3 trial of local chemotherapy with biodegradable carmustine (BCNU) wafers (Gliadel wafers) in patients with primary malignant glioma. Neuro-Oncology 5:79–88
Win KY, Feng SS (2006) In vitro and in vivo studies on vitamin E TPGS-emulsified poly(D, L-lactic-co-glycolic acid) nanoparticles for paclitaxel formulation. Biomaterials 27(10):2285–2291
Acknowledgements
This work was supported by a grant from the National Institutes of Health (NS-045236). AJS was supported by a training grant from the National Institutes of Health (T90 DK-070068). JL was partially supported by the China Scholarship Council.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Sawyer, A.J., Saucier-Sawyer, J.K., Booth, C.J. et al. Convection-enhanced delivery of camptothecin-loaded polymer nanoparticles for treatment of intracranial tumors. Drug Deliv. and Transl. Res. 1, 34–42 (2011). https://doi.org/10.1007/s13346-010-0001-3
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13346-010-0001-3