Abstract
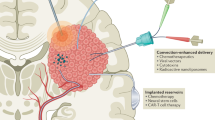
Malignant gliomas are very difficult neoplasms for clinicians to treat. The reason for this is multifaceted. Many treatments that are effective for systemic cancer are unable to cross the blood-brain barrier and/or have unacceptable systemic toxicities. Consequently, in recent years an effort has been placed on trying to develop innovative local treatments that bypass the blood-brain barrier and allow for direct treatment in the central nervous system (CNS)—interstitial treatment. In this paper, we present our extensive experience in using interstitial chemotherapy as a strategy to treat malignant brain tumors at a single institution (The Johns Hopkins Hospital). We provide a comprehensive summary of our preclinical work on interstitial chemotherapy at the Hunterian Neurosurgery Laboratory, reviewing data on rat, rabbit, and monkey studies. Additionally, we present our clinical experience with randomized placebo-controlled studies for the treatment of malignant gliomas. We compare survival statistics for those patients who received placebo versus Gliadel® as initial therapy (11.6 months vs. 13.9 months, respectively) and at the time of tumor recurrence (23 weeks vs. and 31 weeks, respectively). We also discuss the positive impact of local therapy in avoiding the toxicities associated with systemic treatments. Furthermore, we provide an overview of newer chemotherapeutic agents and other strategies used in interstitial treatment. Finally, we offer insight into some of the lessons we have learned from our unique perspective.

Similar content being viewed by others
References
Black PM (1991) Brain tumors. Part 1. N Engl J Med 324:1471–1476
Kornblith PL, Walker M (1988) Chemotherapy for malignant gliomas. J Neurosurg 68:1–17
Hochberg FH, Pruitt A (1980) Assumptions in the radiotherapy of glioblastoma. Neurology 30:907–911
Sampath P, Long DM, Brem H (2000) The Hunterian Neurosurgical Laboratory: the first 100 years of neurosurgical research. Neurosurgery 46:184–194
Guerin C, Laterra J, Drewes LR, Brem H, Goldstein GW (1992) Vascular expression of glucose transporter in experimental brain neoplasms. Am J Pathol 140:417–425
Guerin C, Wolff JE, Laterra J, Drewes LR, Brem H, Goldstein GW (1992) Vascular differentiation and glucose transporter expression in rat gliomas: effects of steroids. Ann Neurol 31:481–487
Sampath P, Hanes J, DiMeco F, Tyler BM, Brat D, Pardoll DM, Brem H (1999) Paracrine immunotherapy with interleukin-2 and local chemotherapy is synergistic in the treatment of experimental brain tumors. Cancer Res 59:2107–2114
Thompson RC, Pardoll DM, Jaffee EM, Ewend MG, Thomas MC, Tyler BM, Brem H (1996) Systemic and local paracrine cytokine therapies using transduced tumor cells are synergistic in treating intracranial tumors. J Immunother Emphasis Tumor Immunol 19:405–413
Weingart JD, Thompson RC, Tyler B, Colvin OM, Brem H (1995) Local delivery of the topoisomerase I inhibitor camptothecin sodium prolongs survival in the rat intracranial 9L gliosarcoma model. Int J Cancer 62:605–609
Weingart JD, Sipos EP, Brem H (1995) The role of minocycline in the treatment of intracranial 9L glioma. J Neurosurg 82:635–640
Sampath P, Brem H (1998) Implantable slow-release chemotherapeutic polymers for the treatment of malignant brain tumors. Cancer Control 5:130–137
Walter KA, Tamargo RJ, Olivi A, Burger PC, Brem H (1995) Intratumoral chemotherapy. Neurosurgery 37:1128–1145
Langer R, Folkman J (1996) Polymers for the sustained release of proteins and other macromolecules. Nature 263:797–800
Tamargo RJ, Myseros JS, Epstein JI, Yang MB, Chasin M, Brem H (1993) Interstitial chemotherapy of the 9L gliosarcoma: controlled release polymers for drug delivery in the brain. Cancer Res 53:329–333
Brem H (1990) Polymers to treat brain tumours. Biomaterials 11:699–701
Leong KW, Brott BC, Langer R (1985) Bioerodible polyanhydrides as drug-carrier matrices. I: characterization, degradation, and release characteristics. J Biomed Mater Res 19:941–955
Tamargo RJ, Epstein JI, Reinhard CS, Chasin M, Brem H (1989) Brain biocompatibility of a biodegradable, controlled-release polymer in rats. J Biomed Mater Res 23:253–266
Brem H, Kader A, Epstein JI, Tamargo RJ, Domb A, Langer R, Leong KW (1989) Biocompatibility of a biodegradable, controlled-release polymer in the rabbit brain. Sel Cancer Ther 5:55–65
Yang MB, Tamargo RJ, Brem H (1989) Controlled delivery of 1,3-bis(2-chloroethyl)-1-nitrosourea from ethylene-vinyl acetate copolymer. Cancer Res 49:5103–5107
Grossman SA, Reinhard C, Colvin OM, Chasin M, Brundrett R, Tamargo RJ, Brem H (1992) The intracerebral distribution of BCNU delivered by surgically implanted biodegradable polymers. J Neurosurg 76:640–647
Brem H, Tamargo RJ, Olivi A, Pinn M, Weingart JD, Wharam M, Epstein JI (1994) Biodegradable polymers for controlled delivery of chemotherapy with and without radiation therapy in the monkey brain. J Neurosurg 80:283–290
Fung LK, Ewend MG, Sills A, Sipos EP, Thompson R, Watts M, Colvin OM, Brem H, Saltzman WM (1998) Pharmacokinetics of interstitial delivery of carmustine, 4-hydroperoxycyclophosphamide, and paclitaxel from a biodegradable polymer implant in the monkey brain. Cancer Res 58:672–684
Bohan E, Brem H (1997) Treatment of brain tumors by local delivery of chemotherapy via biodegradable polymers. Hospital Pharmacist Report (Supplement):12–16
Brem H, Ewend MG, Piantadosi S, Greenhoot J, Burger PC, Sisti M (1995) The safety of interstitial chemotherapy with BCNU-loaded polymer followed by radiation therapy in the treatment of newly diagnosed malignant gliomas: phase I trial. J Neurooncol 26:111–123
Brem H, Langer R (1996) Polymer-based drug delivery to the brain. Sci Med 3:2–11
Brem H, Mahaley MS Jr, Vick NA, Black KL, Schold SC Jr, Burger PC, Friedman AH, Ciric IS, Eller TW, Cozzens JW et al (1991) Interstitial chemotherapy with drug polymer implants for the treatment of recurrent gliomas. J Neurosurg 74:441–446
Brem H, Piantadosi S, Burger PC, Walker M, Selker R, Vick NA, Black K, Sisti M, Brem S, Mohr G et al (1995) Placebo-controlled trial of safety and efficacy of intraoperative controlled delivery by biodegradable polymers of chemotherapy for recurrent gliomas. The Polymer-brain Tumor Treatment Group. Lancet 345:1008–1012
Sipos EP, Tyler B, Piantadosi S, Burger PC, Brem H (1997) Optimizing interstitial delivery of BCNU from controlled release polymers for the treatment of brain tumors. Cancer Chemother Pharmacol 39:383–389
Olivi A, Grossman SA, Tatter S, Barker F, Judy K, Olsen J, Bruce J, Hilt D, Fisher J, Piantadosi S (2003) Dose escalation of carmustine in surgically implanted polymers in patients with recurrent malignant glioma: a new approaches to brain tumor therapy CNS consortium trial. J Clin Oncol 21:1845–1849
Valtonen S, Timonen U, Toivanen P, Kalimo H, Kivipelto L, Heiskanen O, Unsgaard G, Kuurne T (1997) Interstitial chemotherapy with carmustine-loaded polymers for high-grade gliomas: a randomized double-blind study. Neurosurgery 41:44–48
Westphal M, Hilt DC, Bortey E, Delavault P, Olivares R, Warnke PC, Whittle IR, Jaaskelainen J, Ram Z (2003) A phase 3 trial of local chemotherapy with biodegradable carmustine (BCNU) wafers (Gliadel wafers) in patients with primary malignant glioma. Neuro-oncol 5:79–88
Kleinberg LR, Weingart J, Burger P, Carson K, Grossman SA, Li K, Olivi A, Wharam MD, Brem H (2004) Clinical course and pathologic findings after Gliadel and radiotherapy for newly diagnosed malignant glioma: implications for patient management. Cancer Invest 22:1–9
Glantz MJ, Cole BF, Forsyth PA, Recht LD, Wen PY, Chamberlain MC, Grossman SA, Cairncross JG (2000) Practice parameter: anticonvulsant prophylaxis in patients with newly diagnosed brain tumors. Report of the Quality Standards Subcommittee of the American Academy of Neurology. Neurology 54:1886–1893
Renaudin J, Fewer D, Wilson CB, Boldrey EB, Calogero J, Enot KJ (1973) Dose dependency of decadron in patients with partially excised brain tumors. J Neurosurg 39:302–305
Ewend MG, Williams JA, Tabassi K, Tyler BM, Babel KM, Anderson RC, Pinn ML, Brat DJ, Brem H (1996) Local delivery of chemotherapy and concurrent external beam radiotherapy prolongs survival in metastatic brain tumor models. Cancer Res 56:5217–5223
Ewend MG, Sampath P, Williams JA, Tyler BM, Brem H (1998) Local delivery of chemotherapy prolongs survival in experimental brain metastases from breast carcinoma. Neurosurgery 43:1185–1193
Ewend MG, Brem S, Gilbert M, Goodkin R, Penar P (2004) Treating single brain metastasis with resection, placement of BCNU-polymer wafers, and radiation therapy (abstract). Presented at: 72nd Annual Meeting of the American Association of Neurological Surgeons, Toronto, Canada, April 24–26, 2001. Available at: http://www.aans.org/Library/Article.aspx?ArticleId=13719. Accessed October 15
Golden GA, Meldorf M, Group at PS (2004) Patients with metastatic brain cancer, undergoing resection and Gliadel® implantation experienced low local recurrence rates in the PROLONG® Registry (abstract). Presented at the Annual Meeting of the Society of Neuro-Oncology, Abstract TA-25, Toronto, Canada; November 18–21
Brem S, Staller A, Wotoczek-Obadia Mea, Robb J, Vrionis FD, Pearlman JL, Rojiani A, Snodgrass S (2004) Interstitial chemotherapy for local control of CNS metastases. Presented at the annual meeting of the Soc Neuro-Oncology, Abstract TA-06, Toronto, Canada; November 18–21
Pegg AE, Boosalis M, Samson L, Moschel RC, Byers TL, Swenn K, Dolan ME (1993) Mechanism of inactivation of human O6-alkylguanine-DNA alkyltransferase by O6-benzylguanine. Biochemistry 32:11998–12006
Felker GM, Friedman HS, Dolan ME, Moschel RC, Schold C (1993) Treatment of subcutaneous and intracranial brain tumor xenografts with O6-benzylguanine and 1,3-bis(2-chloroethyl)-1-nitrosourea. Cancer Chemother Pharmacol 32:471–476
Rhines LD, Sampath P, Dolan ME, Tyler BM, Brem H, Weingart J (2000) O6-benzylguanine potentiates the antitumor effect of locally delivered carmustine against an intracranial rat glioma. Cancer Res 60:6307–6310
Walter KA, Cahan MA, Gur A, Tyler B, Hilton J, Colvin OM, Burger PC, Domb A, Brem H (1994) Interstitial taxol delivered from a biodegradable polymer implant against experimental malignant glioma. Cancer Res 54:2207–2212
Cahan MA, Walter KA, Colvin OM, Brem H (1994) Cytotoxicity of taxol in vitro against human and rat malignant brain tumors. Cancer Chemother Pharmacol 33:441–444
Olivi A, Ewend MG, Utsuki T, Tyler B, Domb AJ, Brat DJ, Brem H (1996) Interstitial delivery of carboplatin via biodegradable polymers is effective against experimental glioma in the rat. Cancer Chemother Pharmacol 39:90–96
Storm PB, Moriarity JL, Tyler B, Burger PC, Brem H, Weingart J (2002) Polymer delivery of camptothecin against 9L gliosarcoma: release, distribution, and efficacy. J Neurooncol 56:209–217
Sampath P, Amundson E, Wall ME, Tyler BM, Wani MC, Alderson LM, Colvin M, Brem H, Weingart JD (2003) Camptothecin analogs in malignant gliomas: comparative analysis and characterization. J Neurosurg 98:570–577
Judy KD, Olivi A, Buahin KG, Domb A, Epstein JI, Colvin OM, Brem H (1995) Effectiveness of controlled release of a cyclophosphamide derivative with polymers against rat gliomas. J Neurosurg 82:481–486
Sipos EP, Witham TF, Ratan R, Burger PC, Baraban J, Li KW, Piantadosi S, Brem H (2001) l-buthionine sulfoximine potentiates the antitumor effect of 4-hydroperoxycyclophosphamide when administered locally in a rat glioma model. Neurosurgery 48:392–400
DiMeco F, Li KW, Tyler BM, Wolf AS, Brem H, Olivi A (2002) Local delivery of mitoxantrone for the treatment of malignant brain tumors in rats. J Neurosurg 97:1173–1178
Frazier JL, Wang PP, Case D, Tyler BM, Pradilla G, Weingart JD, Brem H (2003) Local delivery of minocycline and systemic BCNU have synergistic activity in the treatment of intracranial glioma. J Neurooncol 64:203–209
Rhines LD, Sampath P, DiMeco F, Lawson HC, Tyler BM, Hanes J, Olivi A, Brem H (2003) Local immunotherapy with interleukin-2 delivered from biodegradable polymer microspheres combined with interstitial chemotherapy: a novel treatment for experimental malignant glioma. Neurosurgery 52:872–879
Richards Grayson AC, Choi IS, Tyler BM, Wang PP, Brem H, Cima MJ, Langer R (2003) Multi-pulse drug delivery from a resorbable polymeric microchip device. Nat Mater 2:767–772
Acknowledgements
Disclosure: Dr. Henry Brem and the Johns Hopkins University are entitled to a share of royalty by MGI Pharma, Inc. on potential sales of some products that are in development and described in this work. The terms of this arrangement are being managed by the Johns Hopkins University in accordance with its conflict of interest policies. Dr. Prakash Sampath is a member of the Speakers Bureau for MGI Pharma, Inc., formerly Guildford Pharmaceuticals, Inc.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Lawson, H.C., Sampath, P., Bohan, E. et al. Interstitial chemotherapy for malignant gliomas: the Johns Hopkins experience. J Neurooncol 83, 61–70 (2007). https://doi.org/10.1007/s11060-006-9303-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11060-006-9303-1