Abstract
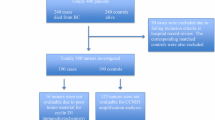
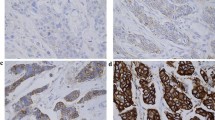
Cyclin E is a well-characterized cell cycle regulator and an amplified oncogene in breast cancer. Over-expression of cyclin E has generally been associated with poor survival. Recent studies have shown an interaction between HER-2 (ERBB2) and cyclin E, but the exact mechanism is unknown. Interestingly, cyclin E over-expression has been associated with trastuzumab resistance. We studied cyclin E over-expression, CCNE1 amplification, and relapse-free survival in HER-2-positive primary breast cancers treated with and without trastuzumab therapy. Formalin-fixed paraffin-embedded tissue samples from 202 HER-2-positive breast carcinomas were studied. Expression levels of cyclin E and proliferation marker Ki-67 were determined using immunohistochemistry. Chromogenic in situ hybridization (CISH) with a gene-specific bacterial artificial chromosome (BAC) probe was used to analyze presence of CCNE1 amplification. Majority of HER-2-positive breast carcinomas exhibited nuclear staining for cyclin E protein. Cyclin E was highly expressed (≥50 % cells) in 37 % of cases. Incidence of CCNE1 amplification (≥6 gene copies/cell or clusters) was 8 %. Cyclin E amplification and over-expression were strongly associated with each other, grade, hormone receptors, and Ki-67. Neither high cyclin E expression nor CCNE1 amplification was associated with relapse-free survival (RFS) irrespective of short-term (9-week regimen) adjuvant trastuzumab therapy. These results confirm cyclin E and HER-2 gene co-amplification in a fraction of HER-2-positive breast cancers. Cyclin E is frequently over-expressed but appears to have limited value as a prognostic or predictive factor in HER-2-positive breast cancer regardless of trastuzumab therapy.





Similar content being viewed by others
References
Autier P, Boniol M, La Vecchia C, et al. Disparities in breast cancer mortality trends between 30 European countries: retrospective trend analysis of WHO mortality database. BMJ. 2010;341:c3620.
Tevaarwerk AJ, Gray RJ, Schneider BP, et al. Survival in patients with metastatic recurrent breast cancer after adjuvant chemotherapy: little evidence of improvement over the past 30 years. Cancer. 2013;119(6):1140–8.
Slamon DJ, Clark GM, Wong SG, et al. Human breast cancer: correlation of relapse and survival with amplification of the HER-2/neu oncogene. Science. 1987;235:177–82.
Gilcrease MZ, Woodward WA, Nicolas MM, et al. Even low-level HER2 expression may be associated with worse outcome in node-positive breast cancer. Am J Surg Pathol. 2009;33:759–67.
Köninki K, Tanner M, Auvinen A, et al. HER-2 positive breast cancer: decreasing proportion but stable incidence in Finnish population from 1982 to 2005. Breast Cancer Res. 2009;11:R37.
Sircoulomb F, Bekhouche I, Finetti P, et al. Genome profiling of ERBB2-amplified breast cancers. BMC Cancer. 2010;10:539.
Palmieri C, Shah D, Krell J, et al. Management and outcome of HER2-positive early breast cancer treated with or without trastuzumab in the adjuvant trastuzumab era. Clin Breast Cancer. 2011;11:93–102.
Vu T, Claret FX. Trastuzumab: updated mechanisms of action and resistance in breast cancer. Review. Front Oncol. 2012;2:62.
Scott KA, Walker RA. Lack of cyclin E immunoreactivity in non-malignant breast and association with proliferation in breast cancer. Br J Cancer. 1997;76:1288–92.
He W, Huang T, Wang H. Expression of cyclin E and its relationship with the prognosis of patients with breast cancer. Chin-Ger J Clin Oncol. 2006;5:275–8.
Scaltriti M, Eichhorn PJ, Cortés J, et al. Cyclin E amplification/overexpression is a mechanism of trastuzumab resistance in HER2+ breast cancer patients. Proc Natl Acad Sci U S A. 2011;108:3761–6.
Natrajan R, Mackay A, Wilkerson PM, Lambros MB, Wetterskog D, Arnedos M, et al. Functional characterization of the 19q12 amplicon in grade III breast cancers. Breast Cancer Res. 2012;14:R53.
Sánchez I, Dynlacht BD. New insights into cyclins, CDKs, and cell cycle control. Review. Semin Cell Dev Biol. 2005;16:311–21.
Ohtsubo M, Theodoras AM, Schumacher J, et al. Human cyclin E, a nuclear protein essential for the G1-to-S phase transition. Mol Cell Biol. 1995;15(5):2612–24.
Resnitzky D, Reed SI. Different roles for cyclins D1 and E in regulation of the G1-to-S transition. Mol Cell Biol. 1995;15(7):3463–9.
Donnellan R, Kleinschmidt I, Chetty R. Cyclin E immunoexpression in breast ductal carcinoma: pathologic correlations and prognostic implications. Hum Pathol. 2001;32:89–94.
Potemski P, Kusinska R, Watala C, et al. Cyclin E expression in breast cancer correlates with negative steroid receptor status, HER2 expression, tumor grade and proliferation. J Exp Clin Cancer Res. 2006;25:59–64.
Wang L, Shao ZM. Cyclin E expression and prognosis in breast cancer patients: a meta-analysis of published studies. Cancer Investig. 2006;24:581–7.
Gao S, Ma JJ, Lu C. Prognostic value of cyclin E expression in breast cancer: a meta-analysis. Tumor Biol. 2013;34:3423–30.
Timms JF, White SL, O’Hare MJ, et al. Effects of ErbB-2 overexpression on mitogenic signalling and cell cycle progression in human breast luminal epithelial cells. Oncogene. 2002;21:6573–86.
Mittendorf EA, Liu Y, Tucker SL, et al. A novel interaction between HER2/neu and cyclin E in breast cancer. Oncogene. 2010;29:3896–907.
Joensuu H, Kellokumpu-Lehtinen PL, Bono P, et al. Adjuvant docetaxel or vinorelbine with or without trastuzumab for breast cancer. N Engl J Med. 2006;354:809–20.
Potemski P, Pluciennik E, Bednarek AK, et al. Cyclin E expression in operable breast cancer quantified using real-time RT-PCR: a comparative study with immunostaining. Jpn J Clin Oncol. 2006;36:142–9.
Olvera M, Harris S, Amezcua CA, et al. Immunohistochemical expression of cell cycle proteins E2F-1, Cdk-2, Cyclin E, p27(kip1), and Ki-67 in normal placenta and gestational trophoblastic disease. Mod Pathol. 2001;14:1036–42.
Tuominen VJ, Ruotoistenmäki S, Viitanen A, et al. ImmunoRatio: a publicly available web application for quantitative image analysis of estrogen receptor (ER), progesterone receptor (PR), and Ki-67. Breast Cancer Res. 2010;12:R56.
Keyomarsi K, Pardee AB. Redundant cyclin overexpression and gene amplification in breast cancer cells. Proc Natl Acad Sci U S A. 1993;90:1112–6.
Tanner M, Kapanen AI, Junttila T, et al. Characterization of a novel cell line established from a patient with Herceptin-resistant breast cancer. Mol Cancer Ther. 2004;3(12):1585–92.
Boström P, Söderström M, Palokangas T, et al. Analysis of cyclins A, B1, D1 and E in breast cancer in relation to tumour grade and other prognostic factors. BMC Res Notes. 2009;2:140.
Keyomarsi K, Conte Jr D, Toyofuku W, et al. Deregulation of cyclin E in breast cancer. Oncogene. 1995;11(5):941–50.
Han S, Park K, Bae BN, et al. Prognostic implication of cyclin E expression and its relationship with cyclin D1 and p27Kip1 expression on tissue microarrays of node negative breast cancer. J Surg Oncol. 2003;83:241–7.
Kim HK, Park IA, Heo DS, et al. Cyclin E overexpression as an independent risk factor of visceral relapse in breast cancer. Eur J Surg Oncol. 2001;27:464–71.
Moelans CB, de Weger RA, Monsuur HN, et al. Molecular profiling of invasive breast cancer by multiplex ligation-dependent probe amplification-based copy number analysis of tumor suppressor and oncogenes. Mod Pathol. 2010;23(7):1029–39.
Russnes HG, Vollan HK, Lingjaerde OC, et al. Genomic architecture characterizes tumor progression paths and fate in breast cancer patients. Sci Transl Med. 2010;2:38ra47.
Callagy G, Pharoah P, Chin SF, et al. Identification and validation of prognostic markers in breast cancer with the complementary use of array-CGH and tissue microarrays. J Pathol. 2005;205(3):388–96.
Lindahl T, Landberg G, Ahlgren J, et al. Overexpression of cyclin E protein is associated with specific mutation types in the p53 gene and poor survival in human breast cancer. Carcinogenesis. 2004;25:375–80.
Bukholm IR, Bukholm G, Nesland JM. Over-expression of cyclin A is highly associated with early relapse and reduced survival in patients with primary breast carcinomas. Int J Cancer. 2001;93:283–7.
Pillay K, McCleod H, Chetty R, et al. A study to investigate the role of p27 and cyclin E immunoexpression as a prognostic factor in early breast carcinoma. World J Surg Oncol. 2011;9:31.
Peters MG, Vidal Mdel C, Giménez L, et al. Prognostic value of cell cycle regulator molecules in surgically resected stage I and II breast cancer. Oncol Rep. 2004;12(5):1143–50.
Berglund P, Stighall M, Jirström K, et al. Cyclin E overexpression obstructs infiltrative behavior in breast cancer: a novel role reflected in the growth pattern of medullary breast cancers. Cancer Res. 2005;65(21):9727–34.
Kühling H, Alm P, Olsson H, et al. Expression of cyclins E, A, and B, and prognosis in lymph node-negative breast cancer. J Pathol. 2003;199(4):424–31.
Sieuwerts AM, Look MP, Meijer-van Gelder ME, et al. Which cyclin E prevails as prognostic marker for breast cancer? Results from a retrospective study involving 635 lymph node-negative breast cancer patients. Clin Cancer Res. 2006;12(11 Pt 1):3319–28.
Potemski P, Kusińska R, Pasz-Walczak G, et al. Prognostic relevance of cyclin E expression in operable breast cancer. Med Sci Monit. 2009;15(2):MT34–40.
Bagaria SP, Ray PS, Wang J, et al. Prognostic value of basal phenotype in HER2-overexpressing breast cancer. Ann Surg Oncol. 2012;19:935–40.
Vaz-Luis I, Ottesen RA, Hughes ME, et al. Impact of hormone receptor status on patterns of recurrence and clinical outcomes among patients with human epidermal growth factor-2-positive breast cancer in the National Comprehensive Cancer Network: a prospective cohort study. Breast Cancer Res. 2012;14(5):R129.
Keyomarsi K, Tucker SL, Buchholz TA, et al. Cyclin E and survival in patients with breast cancer. N Engl J Med. 2002;347:1566–75.
Harwell RM, Porter DC, Danes C, et al. Processing of cyclin E differs between normal and tumor breast cells. Cancer Res. 2000;60:481–9.
Wingate H, Puskas A, Duong M, et al. Low molecular weight cyclin E is specific in breast cancer and is associated with mechanisms of tumor progression. Cell Cycle. 2009;8:1062–8.
Porter DC, Zhang N, Danes C, et al. Tumor-specific proteolytic processing of cyclin E generates hyperactive lower-molecular-weight forms. Mol Cell Biol. 2001;21:6254–69.
Bagheri-Yarmand R, Biernacka A, Hunt KK, et al. Low molecular weight cyclin E overexpression shortens mitosis, leading to chromosome missegregation and centrosome amplification. Cancer Res. 2010;70:5074–84.
Tokai Y, Maeda S, Yamaguchi J, et al. Cyclin E low-molecular-weight isoform as a predictor of breast cancer in Japanese women. Int Surg. 2011;96:245–53.
Delk NA, Hunt KK, Keyomarsi K. Altered subcellular localization of tumor-specific cyclin E isoforms affects cyclin-dependent kinase 2 complex formation and proteasomal regulation. Cancer Res. 2009;69:2817–25.
Acknowledgments
Authors would like to thank Mrs. Sari Toivola and Mrs. Kristiina Salonoja for their excellent technical assistance.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
This study was approved by the local ethics committee (R07082).
Conflicts of interest
None
Rights and permissions
About this article
Cite this article
Luhtala, S., Staff, S., Tanner, M. et al. Cyclin E amplification, over-expression, and relapse-free survival in HER-2-positive primary breast cancer. Tumor Biol. 37, 9813–9823 (2016). https://doi.org/10.1007/s13277-016-4870-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13277-016-4870-z