Abstract
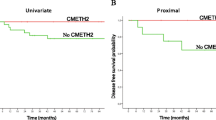
KISS1 is a metastasis suppressor lost in several solid malignancies. We evaluated the clinical relevance of KiSS-1 methylation and its protein expression in colorectal cancer. The epigenetic silencing of KiSS-1 by hypermethylation was tested in colon cancer cells (n = 5) before and after azacytidine treatment. KiSS-1 methylation was evaluated by methylation-specific PCR in colorectal cancer cells, and normal, benign, and tumor tissues (n = 352) were grouped in a training set (n = 62) and two independent validation cohorts (n = 100 and n = 190). KiSS-1 protein expression was analyzed by immunohistochemistry on tissue arrays. KiSS-1 hypermethylation correlated with transcript and protein expression loss, being increased in vitro by azacytidine. Methylation rates were 53.1, 70.0, and 80.0 % in the training and validation sets, respectively. In the training set, KiSS-1 methylation rendered a diagnostic accuracy of 72.7 % (p = 0.002). Combination of KiSS-1 methylation and serum CEA (p = 0.001) increased the prognostic utility of CEA alone (p = 0.022). In the first validation set, KiSS-1 methylation correlated with tumor grade (p = 0.011), predicted recurrence (p = 0.009), metastasis (p = 0.004), disease-free (p = 0.034), and overall survival (p = 0.015). In the second validation cohort, KiSS-1 methylation predicted disease-specific survival (p = 0.030). In the training set, cytoplasmic KiSS-1 expression was significantly higher in nonneoplastic biopsies as compared to colorectal tumors (p < 0.0005). In the validation set, loss of cytoplasmic expression correlated with tumor stage (p = 0.007), grade (p = 0.035), recurrence (p = 0.017), and disease-specific survival (p = 0.022). KiSS-1 was revealed epigenetically modified in colorectal cancer. The diagnostic and prognostic utility of KiSS-1 methylation and expression patterns suggests their assessment for the clinical management of colorectal cancer patients.




Similar content being viewed by others
References
Siegel R, Naishadham D, Jemal A. Cancer statistics, 2012. CA Cancer J Clin. 2012;62:10–29.
Cunningham D, Atkin W, Lenz H, et al. Colorectal cancer. Lancet. 2010;375:1030–47.
Grady WM, Carethers JM. Genomic and epigenetic instability in colorectal cancer pathogenesis. Gastroenterology. 2008;135:1079–99.
Carmona FJ, Esteller M. Epigenetics of human colon cancer. Mutat Res. 2010;693:53–60.
Rashid A, Issa JP. CpG island methylation in gastroenterologic neoplasia: a maturing field. Gastroenterology. 2004;127:1578–88.
Jordà M, Peinado MA. Methods for DNA methylation analysis and applications in colon cancer. Mutat Res. 2010;693:84–93.
Herman JG, Baylin SB. Gene silencing in cancer in association with promoter hypermethylation. N Engl J Med. 2003;349:2042–54.
Esteller M. CpG island hypermethylation and tumor suppressor genes: a booming present, a brighter future. Oncogene. 2002;21:5427–40.
Lee JH, Miele ME, Hicks DJ, et al. KiSS-1, a novel human malignant melanoma metastasis-suppressor gene. J Natl Cancer Inst. 1996;88:1731–7.
Lee JH, Welch DR. Identification of highly expressed genes in metastasis-suppressed chromosome 6/human malignant melanoma hybrid cells using subtractive hybridization and differential display. Int J Cancer. 1997;71:1035–44.
Lee JH, Welch DR. Suppression of metastasis in human breast carcinoma MDA-MB-435 cells after transfection with the metastasis suppressor gene, KiSS-1. Cancer Res. 1997;57:2384–7.
West A, Vojta PJ, Welch DR, et al. Chromosome localization and genomic structure of the KiSS1 metastasis suppressor gene (KISS1). Genomics. 1998;54:145–8.
Ohtaki T, Shintani Y, Honda S, et al. The metastasis suppressor gene KiSS-1 encodes peptide ligand of a G-protein-coupled receptor. Nature. 2001;411:613–6.
Muir AI, Chamberlain L, Elshourbagy NA, et al. AXOR12, a novel human G protein-coupled receptor, activated by the peptide KiSS-1. J Biol Chem. 2001;276:28969–75.
Kotani M, Detheux M, Vandenbogaerde A, et al. The metastasis suppressor gene KiSS-1 encodes kisspeptins, the natural ligands of the orphan G protein-coupled receptor GPR54. J Biol Chem. 2001;276:34631–6.
Sanchez-Carbayo M, Capodieci P, Cordon-Cardo C. Tumor suppressor role of KiSS-1 in bladder cancer: loss of KISS1 expression in associated with bladder cancer progression and clinical outcome. Am J Pathol. 2003;162:609–17.
Ikeguchi M, Yamaguchi K, Kaibara N. Clinical significance of the loss of KiSS1 and orphan G-protein-coupled receptor (hOT7T175) gene expression in esophageal squamous cell carcinoma. Clin Cancer Res. 2004;10:1379–83.
Hesling C, D'Incan M, Mansard S, et al. In vivo and in situ modulation of the expression of genes involved in metastasis and angiogenesis in a patient treated with topical imiquimod for melanoma skin metastasis. Br J Dermatol. 2004;150:761–7.
Dhar D, Naora H, Kubota H, et al. Downregulation of KiSS-1 expression is responsible for tumor invasion and worse prognosis in gastric carcinoma. Int J Cancer. 2004;111:868–72.
Masui T, Doi R, Mori T, et al. Metastin and its variant forms suppress migration of pancreatic cancer cells. Biochem Biophys Res Commun. 2004;315:85–92.
Martin TA, Watkins G, Jiang WG. KiSS-1 expression in human breast cancer. Clin Exp Metastasis. 2005;22:503–11.
Nicolle G, Comperat E, Nicolaïew N, et al. Metastin (KISS-1) and metastin-coupled receptor (GPR54) expression in transitional cell carcinoma of the bladder. Ann Oncol. 2007;18:605–6.
Zohrabian VM, Nandu H, Gulati N, et al. Gene expression profiling of metastatic brain cancer. Oncol Rep. 2007;18:321–8.
Hata K, Dhar DK, Watanabe Y, et al. Expression of metastin and a G-protein-coupled receptor (AXOR12) in epithelial ovarian cancer. Eur J Cancer. 2007;43:1452–9.
Makri A, Pissimissis N, Lembessis P, et al. The kisspeptin (KISS1)/GPR54 system in cancer biology. Cancer Treat Rev. 2008;34:682–92.
Yamashita S, Tsujino Y, Moriguchi K, et al. Chemical genomic screening for methylation-silenced genes in gastric cancer cell lines using 5-aza-2′-deoxycytidine treatment and oligonucleotide microarray. Cancer Sci. 2006;97:64–71.
Cebrian V, Fierro M, Orenes-Piñero E, et al. KISS1 methylation and expression as tumor stratification biomarkers and clinical outcome prognosticators for bladder cancer patients. Am J Pathol. 2011;179:540–6.
Puppa G, Sonzogni A, Colombari R, et al. TNM staging system of colorectal carcinoma: a critical appraisal of challenging issues. Arch Patol Lab Med. 2010;134:837–52.
Dawson-Saunders B, Trapp R. G. Basic and clinical biostatistics. 2nd ed, Norwalk: Appleton & Lange. 1994
Duffy MJ, van Dalen A, Haglund C, et al. Tumour markers in colorectal cancer: European Group on Tumour Markers (EGTM) guidelines for clinical use. Eur J Cancer. 2007;43:1348–60.
Acknowledgments
We thank all the members of the laboratory of M. Sánchez-Carbayo for the technical support and constructive suggestions in the preparation of this manuscript. We also would like to thank the Tumor Bank belonging to the Molecular Pathology Program at the Spanish National Cancer Center and all the members of our clinical collaborators at the different institutions involved in this study, for the support in facilitating the tumor specimens as well as the clinical follow-up of the colorectal cancer cases analyzed in this study. M. Sánchez-Carbayo received grant supports from Spanish Ministry of Science and Innovation grant SAF2009-13035 and Mutua Madrileña 2010.
Conflicts of interest
None
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Moya, P., Esteban, S., Fernandez-Suarez, A. et al. KiSS-1 methylation and protein expression patterns contribute to diagnostic and prognostic assessments in tissue specimens for colorectal cancer. Tumor Biol. 34, 471–479 (2013). https://doi.org/10.1007/s13277-012-0572-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13277-012-0572-3