Abstract
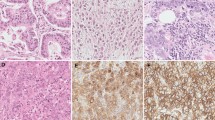
Malignant melanoma is an increasing disease in China, and its molecular mechanisms of development and progression are limited. The objective of this study was to investigate the expression of Ras interaction/interference 1 (RIN1) protein and its clinical significance in human melanoma. Immunohistochemistry was performed to detect the expression of RIN1 in 81 melanoma patients with a 5-year follow-up. The prognosis of the patients, classified by the clinicopathologic features and RIN1 expression, was assessed by multivariate analysis. RIN1 levels were then analyzed with overall survival (OS), progression-free survival (PFS), and recurrence-free survival (RFS) in the cohort. The biological function was determined by proliferation assay, flow cytometry analysis through knocking down of RIN1 in melanoma cells A375, as well as caspase-3 activation and PARP cleavage were detected by western blot or fluorometric assay. Data showed that RIN1 was overexpressed in melanoma samples. High-level RIN1 expression was observed in 49.4 % (40 of 81 cases), associated with thickness grade (P = 0.008) and lymph node metastasis (P < 0.001). Two distinguished subgroups were segregated by RIN1 levels within this set comparing prognostication of OS, PFS, and RFS. Importantly, RIN1 level was revealed as the significant independent prognostic factor for death and progression but a weak contribution for recurrence. Moreover, knock down of RIN1 expression in A375 cells, suppressed cell proliferation and induced apoptosis through caspase-3 activation and PARP cleavage. RIN1 expression could be a potential prognostic predictor for the melanoma patients and provide a potential target therapy for melanoma treatment.




Similar content being viewed by others
References
Jemal A, Siegel R, Ward E, Murray T, Xu J, Thun MJ. Cancer statistics. CA Cancer J Clin. 2007;57:43–66.
De Braud F, Khayat D, Kroon BB, Valdagni R, Bruzzi P, Cascinelli N. Malignant melanoma. Crit Rev Oncol Hematol. 2003;47:35–63.
Boyle GM. Therapy for metastatic melanoma: an overview and update. Expert Rev Anticancer Ther. 2011;11:725–37.
Colicelli J, Nicolette C, Birchmeier C, Rodgers L, Riggs M, Wigler M. Expression of three mammalian cDNAs that interfere with RAS function in Saccharomyces cerevisiae. Proc Natl Acad Sci USA. 1991;88:2913–17.
Han L, Wong D, Dhaka A, Afar D, White M, Xie W, Herschman H, Witte O, Colicelli J. Protein binding and signaling properties of RIN1 suggest a unique effector function. Proc Natl Acad Sci USA. 1997;94:4954–59.
Afar DE, Han L, McLaughlin J, Wong S, Dhaka A, Parmar K, Rosenberg N, Witte ON, Colicelli J. Regulation of the oncogenic activity of BCR-ABL by a tightly bound substrate protein RIN1. Immunity. 1997;6:773–82.
Hu H, Bliss JM, Wang Y, Colicelli J. RIN1 is an ABL tyrosine kinase activator and a regulator of epithelial-cell adhesion and migration. Curr Biol. 2005;15:815–23.
Cao X, Tanis KQ, Koleske AJ, Colicelli J. Enhancement of ABL kinase catalytic efficiency by a direct binding regulator is independent of other regulatory mechanisms. J Biol Chem. 2008;283:31401–7.
Barbieri MA, Kong C, Chen PI, Horazdovsky BF, Stahl PD. The SRC homology 2 domain of Rin1 mediates its binding to the epidermal growth factor receptor and regulates receptor endocytosis. J Biol Chem. 2003;278:32027–36.
Tall GG, Barbieri MA, Stahl PD, Horazdovsky BF. Ras-activated endocytosis is mediated by the Rab5 guanine nucleotide exchange activity of RIN1. Dev Cell. 2001;1:73–82.
Shuster MI, Han L, Le Beau MM, Davis E, Sawicki M, Lese CM, Park NH, Colicelli J, Gollin SM. A consistent pattern of RIN1 rearrangements in oral squamous cell carcinoma cell lines supports a breakage-fusion-bridge cycle model for 11q13 amplification. Genes Chromosomes Cancer. 2000;28:153–63.
Zainabadi K, Benyamini P, Chakrabarti R, Veena MS, Chandrasekharappa SC, Gatti RA, Srivatsan ES. A 700-kb physical and transcription map of the cervical cancer tumor suppressor gene locus on chromosome 11q13. Genomics. 2005;85:704–14.
Tomshine JC, Severson SR, Wigle DA, Sun Z, Beleford DA, Shridhar V, Horazdovsky BF. Cell Proliferation and epidermal growth factor signaling in non-small cell lung adenocarcinoma cell lines are dependent on RIN1. J Biol Chem. 2009;284:26331–9.
Senda K, Goi T, Hirono Y, Katayama K, Yamaguchi A. Analysis of RIN1 gene expression in colorectal cancer. Oncol Rep. 2007;17:1171–5.
Wang Q, Gao Y, Tang Y, Ma L, Zhao M, Wang X. Prognostic significance of RIN1 gene expression in human non-small cell lung cancer. Acta Histochemica. doi:10.1016/j.acthis.2011.08.008
Colicelli J. Human RAS superfamily proteins and related GTPases. Sci STKE. 2004;250:RE13.
Han L, Colicelli J. A human protein selected for interference with RAS function interacts directly with RAS and competes with Raf1. Molecular and Cellular Biology. 1995;15:1318–23.
Wang Y, Waldron RT, Dhaka A, Patel A, Riley MM, Rozengurt E, Colicelli J. The RAS effector RIN1 directly competes with RAF and is regulated by 14-3-3 proteins. Mol Cell Biol. 2002;22:916–26.
Bliss JM, Venkatesh B, Colicelli J. The RIN family of Ras effectors. Methods in enzymology. 2006;6:335–44.
Bliss JM, Gray EE, Dhaka A, O'Dell TJ, Colicelli J. Fear learning and extinction are linked to neuronal plasticity through RIN1 signaling. J Neurosci Res. 2010;88:917–26.
Fujioka M, Goi T, Hirono Y. Cloning of a novel splicing variant of RIN1 and its expression in gastric and colon cancer. Oncol Res. 2009;17:593–9.
Chetcuti A, Aktas S, Mackie N. Expression profiling reveals MSX1 and EphB2 expression correlates with the invasion capacity of Wilms tumors. Pediatr Blood Cancer. 2011;57:950–7.
Samant RS, Debies MT, Shevde LA, Verderame MF, Welch DR. Identification and characterization of the murine ortholog(brms1)of breast-cancer metastasis supperssor1 (BRMS1). Int J Cancer. 2002;97:15–20.
Thai M, Ting PY, McLaughlin J, Cheng D, Muschen M, Witte ON, Colicelli J. ABL fusion oncogene transformation and inhibitor sensitivity are mediated by the cellular regulator RIN1. Leukemia. 2011;25:290–300.
Milstein M, Mooser CK, Hu H, Fejzo M, Slamon D, Goodglick L, Dry S, Colicelli J. RIN1 is a breast tumor suppressor gene. Cancer Res. 2007;67:11510–6.
Namura S, Zhu J, Fink K, Endres M, Srinivasan A. Activation and cleavage of caspase-3 in apoptosis induced by experimental cerebral ischemia. J Neurosci. 1998;18:3659–68.
Conflicts of interest
None.
Author information
Authors and Affiliations
Corresponding author
Additional information
Ping Fang and Xin Zhang have contributed equally to the manuscript.
Rights and permissions
About this article
Cite this article
Fang, P., Zhao, Z., Tian, H. et al. RIN1 exhibits oncogenic property to suppress apoptosis and its aberrant accumulation associates with poor prognosis in melanoma. Tumor Biol. 33, 1511–1518 (2012). https://doi.org/10.1007/s13277-012-0402-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13277-012-0402-7