Abstract
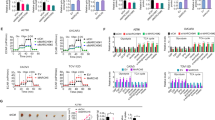
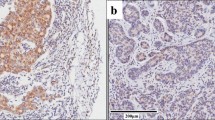
Several protein kinase C (PKC) isoforms have been shown to influence different cellular processes that may contribute to the malignancy of breast cancer cells. To obtain insight into mechanisms mediating the PKC effects, global gene expression was analyzed in MDA-MB-231 breast cancer cells in which PKCα, PKCδ or PKCε had been down-regulated with siRNA. Gene set enrichment analyses revealed that hypoxia-induced genes were enriched among genes that increased in PKCα-down-regulated cells. The STC1 mRNA, encoding stanniocalcin 1, was particularly up-regulated following depletion of PKCα and was also induced by hypoxia. Both hypoxia and PKCα down-regulation also led to increased STC1 protein levels. The results demonstrate that PKCα suppresses the expression of STC1 in breast cancer cells.





Similar content being viewed by others
References
Griner EM, Kazanietz MG. Protein kinase c and other diacylglycerol effectors in cancer. Nat Rev Cancer. 2007;7:281–94.
Konopatskaya O, Poole AW. Protein kinase cα: Disease regulator and therapeutic target. Trends Pharmacol Sci. 2010;31:8–14.
Newton AC. Protein kinase c: Poised to signal. Am J Physiol Endocrinol Metab. 2010;298:E395–402.
Ways DK, Kukoly CA. deVente J, Hooker JL, Bryant WO, Posekany KJ, Fletcher DJ, Cook PP, Parker PJ: Mcf-7 breast cancer cells transfected with protein kinase cα exhibit altered expression of other protein kinase c isoforms and display a more aggressive neoplastic phenotype. J Clin Invest. 1995;95:1906–15.
Soh JW, Lee YS, Weinstein IB. Effects of regulatory domains of specific isoforms of protein kinase c on growth control and apoptosis in mcf-7 breast cancer cells. J Exp Ther Oncol. 2003;3:115–26.
Tonetti DA, Chisamore MJ, Grdina W, Schurz H, Jordan VC. Stable transfection of protein kinase cα cdna in hormone-dependent breast cancer cell lines. Br J Cancer. 2000;83:782–91.
Lønne GK, Cornmark L, Zahirovic IO, Landberg G, Jirstrom K, Larsson C. Pkcα expression is a marker for breast cancer aggressiveness. Mol Cancer. 2010;9:76.
Lønne GK, Masoumi KC, Lennartsson J, Larsson C. Protein kinase cδ supports survival of mda-mb-231 breast cancer cells by suppressing the erk1/2 pathway. J Biol Chem. 2009;284:33456–65.
McCracken MA, Miraglia LJ, McKay RA, Strobl JS. Protein kinase cδ is a prosurvival factor in human breast tumor cell lines. Mol Cancer Ther. 2003;2:273–81.
Nabha SM, Glaros S, Hong M, Lykkesfeldt AE, Schiff R, Osborne K, et al. Upregulation of pkcδ contributes to antiestrogen resistance in mammary tumor cells. Oncogene. 2005;24:3166–76.
Pan Q, Bao LW, Kleer CG, Sabel MS, Griffith KA, Teknos TN, et al. Protein kinase cε is a predictive biomarker of aggressive breast cancer and a validated target for rna interference anticancer therapy. Cancer Res. 2005;65:8366–71.
Alonso-Escolano D, Medina C, Cieslik K, Radomski A, Jurasz P, Santos-Martinez MJ, et al. Protein kinase c delta mediates platelet-induced breast cancer cell invasion. J Pharmacol Exp Ther. 2006;318:373–80.
Grossoni VC, Falbo KB, Kazanietz MG. de Kier Joffe ED, Urtreger AJ: Protein kinase c delta enhances proliferation and survival of murine mammary cells. Mol Carcinog. 2007;46:381–90.
Kiley SC, Clark KJ, Duddy SK, Welch DR, Jaken S. Increased protein kinase c delta in mammary tumor cells: Relationship to transformtion and metastatic progression. Oncogene. 1999;18:6748–57.
Kiley SC, Clark KJ, Goodnough M, Welch DR, Jaken S. Protein kinase c delta involvement in mammary tumor cell metastasis. Cancer Res. 1999;59:3230–8.
Pedram A, Razandi M, Wallace DC, Levin ER. Functional estrogen receptors in the mitochondria of breast cancer cells. Mol Biol Cell. 2006;17:2125–37.
Zeidan YH, Wu BX, Jenkins RW, Obeid LM, Hannun YA. A novel role for protein kinase cdelta-mediated phosphorylation of acid sphingomyelinase in uv light-induced mitochondrial injury. FASEB J. 2008;22:183–93.
Jellinek DA, Chang AC, Larsen MR, Wang X, Robinson PJ, Reddel RR. Stanniocalcin 1 and 2 are secreted as phosphoproteins from human fibrosarcoma cells. Biochem J. 2000;350:453–61.
Varghese R, Wong CK, Deol H, Wagner GF, DiMattia GE. Comparative analysis of mammalian stanniocalcin genes. Endocrinology. 1998;139:4714–25.
Zhang KZ, Westberg JA, Paetau A, von Boguslawsky K, Lindsberg P, Erlander M, et al. High expression of stanniocalcin in differentiated brain neurons. Am J Pathol. 1998;153:439–45.
McCudden CR, James KA, Hasilo C, Wagner GF. Characterization of mammalian stanniocalcin receptors. Mitochondrial targeting of ligand and receptor for regulation of cellular metabolism. J Biol Chem. 2002;277:45249–58.
Ellard JP, McCudden CR, Tanega C, James KA, Ratkovic S, Staples JF, et al. The respiratory effects of stanniocalcin-1 (stc-1) on intact mitochondria and cells: Stc-1 uncouples oxidative phosphorylation and its actions are modulated by nucleotide triphosphates. Mol Cell Endocrinol. 2007;264:90–101.
Wang Y, Huang L, Abdelrahim M, Cai Q, Truong A, Bick R, et al. Stanniocalcin-1 suppresses superoxide generation in macrophages through induction of mitochondrial ucp2. J Leukoc Biol. 2009;86:981–8.
Sheikh-Hamad D. Mammalian stanniocalcin-1 activates mitochondrial antioxidant pathways: New paradigms for regulation of macrophages and endothelium. Am J Physiol Renal Physiol. 2010;298:F248–254.
Zhang K, Lindsberg PJ, Tatlisumak T, Kaste M, Olsen HS, Andersson LC. Stanniocalcin: A molecular guard of neurons during cerebral ischemia. Proc Natl Acad Sci U S A. 2000;97:3637–42.
Lal A, Peters H. St Croix B, Haroon ZA, Dewhirst MW, Strausberg RL, Kaanders JH, van der Kogel AJ, Riggins GJ: Transcriptional response to hypoxia in human tumors. J Natl Cancer Inst. 2001;93:1337–43.
Yeung HY, Lai KP, Chan HY, Mak NK, Wagner GF, Wong CK. Hypoxia-inducible factor-1-mediated activation of stanniocalcin-1 in human cancer cells. Endocrinology. 2005;146:4951–60.
Welcsh PL, Lee MK, Gonzalez-Hernandez RM, Black DJ, Mahadevappa M, Swisher EM, et al. Brca1 transcriptionally regulates genes involved in breast tumorigenesis. Proc Natl Acad Sci U S A. 2002;99:7560–5.
Bouras T, Southey MC, Chang AC, Reddel RR, Willhite D, Glynne R, et al. Stanniocalcin 2 is an estrogen-responsive gene coexpressed with the estrogen receptor in human breast cancer. Cancer Res. 2002;62:1289–95.
Joensuu K, Heikkila P, Andersson LC. Tumor dormancy: Elevated expression of stanniocalcins in late relapsing breast cancer. Cancer Lett. 2008;265:76–83.
Mootha VK, Lindgren CM, Eriksson KF, Subramanian A, Sihag S, Lehar J, et al. Pgc-1α-responsive genes involved in oxidative phosphorylation are coordinately down-regulated in human diabetes. Nat Genet. 2003;34:267–73.
Subramanian A, Tamayo P, Mootha VK, Mukherjee S, Ebert BL, Gillette MA, et al. Gene set enrichment analysis: A knowledge-based approach for interpreting genome-wide expression profiles. Proc Natl Acad Sci U S A. 2005;102:15545–50.
Lofstedt T, Jogi A, Sigvardsson M, Gradin K, Poellinger L, Pahlman S, et al. Induction of id2 expression by hypoxia-inducible factor-1: A role in dedifferentiation of hypoxic neuroblastoma cells. J Biol Chem. 2004;279:39223–31.
De Preter K, Speleman F, Combaret V, Lunec J, Laureys G, Eussen BH, et al. Quantification of mycn, ddx1, and nag gene copy number in neuroblastoma using a real-time quantitative pcr assay. Mod Pathol. 2002;15:159–66.
Jogi A, Vallon-Christersson J, Holmquist L, Axelson H, Borg A, Pahlman S. Human neuroblastoma cells exposed to hypoxia: Induction of genes associated with growth, survival, and aggressive behavior. Exp Cell Res. 2004;295:469–87.
Fox SB, Generali DG, Harris AL. Breast tumour angiogenesis. Breast Cancer Res. 2007;9:216.
Erler JT, Bennewith KL, Nicolau M, Dornhofer N, Kong C, Le QT, et al. Lysyl oxidase is essential for hypoxia-induced metastasis. Nature. 2006;440:1222–6.
Parssinen J, Alarmo EL, Khan S, Karhu R, Vihinen M, Kallioniemi A. Identification of differentially expressed genes after pp m1d silencing in breast cancer. Cancer Lett. 2008;259:61–70.
Sheikh-Hamad D, Rouse D, Yang Y. Regulation of stanniocalcin in mdck cells by hypertonicity and extracellular calcium. Am J Physiol Renal Physiol. 2000;278:F417–424.
Block GJ, Ohkouchi S, Fung F, Frenkel J, Gregory C, Pochampally R, et al. Multipotent stromal cells are activated to reduce apoptosis in part by up-regulation and secretion of stanniocalcin-1. Stem Cells. 2009;27:670–81.
Liu G, Yang G, Chang B, Mercado-Uribe I, Huang M, Zheng J, et al. Stanniocalcin 1 and ovarian tumorigenesis. J Natl Cancer Inst. 2010;102:812–27.
Nguyen A, Chang AC, Reddel RR. Stanniocalcin-1 acts in a negative feedback loop in the prosurvival erk1/2 signaling pathway during oxidative stress. Oncogene. 2009;28:1982–92.
Law AY, Lai KP, Lui WC, Wan HT, Wong CK. Histone deacetylase inhibitor-induced cellular apoptosis involves stanniocalcin-1 activation. Exp Cell Res. 2008;314:2975–84.
Acknowledgements
Financial support was obtained from the Swedish Research Council, the Swedish Cancer Society, The Gunnar Nilsson, Crafoord, Ollie and Elof Ericsson and Kock Foundations, and Malmö University Hospital research funds.
Conflicts of interest
None
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
SupplementaryFig. 1
(JPEG 65 kb)
SupplementaryTable 1
(DOC 83 kb)
Rights and permissions
About this article
Cite this article
Cornmark, L., Lønne, G.K., Jögi, A. et al. Protein kinase Cα suppresses the expression of STC1 in MDA-MB-231 breast cancer cells. Tumor Biol. 32, 1023–1030 (2011). https://doi.org/10.1007/s13277-011-0205-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13277-011-0205-2