Abstract
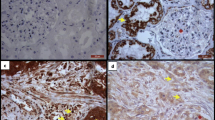
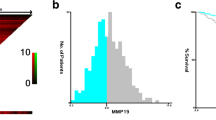
Matrix metalloproteinase-7 is capable of degrading many extracellular matrix proteins and cellular adhesions. In many malignancies, it is overexpressed, and it plays a role in cancer progression by enhancing tumor invasion and thereby metastatic potential. The purpose of this study was to evaluate the association between MMP-7 tissue expression and prognosis in colorectal cancer. From 623 patients who underwent surgery for colorectal cancer, surgical specimens were collected into tissue array blocks and stained by immunohistochemistry for MMP-7. Specimens from 545 patients were suitable for analysis. In specimens from 105 patients (19.3%), MMP-7 scored as high; in 103 (18.9%), as moderate; and in 134 (24.9%), as mild. In 203 cases (37.2%), immunoreactivity was negative. A significant correlation appeared between MMP-7 immunoexpression and tumor differentiation. High MMP-7 positivity associated with poor prognosis during a 5-year follow-up. During longer follow-up, the differences in survival between groups disappeared. MMP-7 is a potential target for tumor therapy, which should be evaluated in clinical trials.


Similar content being viewed by others
References
Globocan 2002 Database.
Zucker S, Vacirca J. Role of matrix metalloproteinases (MMPs) in colorectal cancer. Cancer Metastasis Rev. 2004;23:101–17.
Coussens LM, Werb Z. Matrix metalloproteinases and the development of cancer. Chem Biol. 1996;3:895–904.
MacDougall JR, Matrisian LM. Contributions of tumor and stromal matrix metalloproteinases to tumor progression, invasion and metastasis. Can Met Rev. 1995;14:351–62.
Powell WC, Matrisian LM. Complex roles of matrix metalloproteinases in tumor progression. Curr Top Microbiol Immunol. 1996;213:1–21.
Chakraborti S, Mandal M, Das S, Mandal A, Chakraborti T. Regulation of matrix metalloproteinases: an overview. Mol Cell Biochem. 2003;253:269–85.
Wang WS, Chen PM, Wang HS, Liang WY, Su Y. Matrix metalloproteinase-7 increases resistance to Fas-mediated apoptosis and is a poor prognostic factor of patients with colorectal carcinoma. Carcinogenesis. 2006;27:1113–20.
Von Bredow DC, Nagle RB, Bowden GT, Cress AE. Cleavage of beta 4 integrin by matrilysin. Exp Cell Res. 1997;236:341–5.
Ii M, Yamamoto H, Adachi Y, Maruyama Y, Shinomura Y. Role of matrix metalloproteinase-7 (matrilysin) in human cancer invasion, apoptosis, growth, and angiogenesis. Exp Biol Med. 2006;231:20–7.
Adachi Y, Yamamoto H, Itoh F, Hinoda Y, Okada Y, Imai K. Contribution of matrilysin (MMP-7) to the metastatic pathway of human colorectal cancers. Gut. 1999;45:252–8.
McDonnell S, Navre M, Coffey RJ Jr, Matrisian LM. Expression and localization of the matrix metalloproteinase pump-1 (MMP-7) in human gastric and colon carcinomas. Mol Carcinog. 1991;4:527–33.
Adachi Y, Itoh F, Yamamoto H, Matsuno K, Arimura Y, Kusano M, et al. Matrix metalloproteinase matrilysin (MMP-7) participates in the progression of human gastric and esophageal cancers. Int J Oncol. 1998;13:1931–5.
Crawford HC, Scoggins CR, Washington MK, Matrisian LM, Leach SD. Matrix metalloproteinase-7 is expressed by pancreatic cancer precursors and regulates acinar-to-ductal metaplasia in exocrine pancreas. J Clin Invest. 2002;109:1437–44.
Yamamoto H, Itoh F, Iku S, Adachi Y, Fukushima H, Sasaki S, et al. Expression of matrix metalloproteinases and tissue inhibitors of metalloproteinases in human pancreatic adenocarcinomas: clinicopathologic and prognostic significance of matrilysin expression. J Clin Oncol. 2001;19:1118–27.
Pjouh MS, Nagle RB, Breathnach R, Finch JS, Brawer MK, Bowden GT. Expression of metalloproteinase genes in human prostate cancer. J Cancer Res Clin Oncol. 1991;117:144–50.
Muller D, Breatchnach R, Engelmann A, Millon R, Bronner G, Flesch H, et al. Expression of collagenase-related metalloproteinase genes in human lung or head and neck tumours. Int J Cancer. 1991;48:550–6.
Basset P, Bellocq JP, Wolf C, Stoll I, Hutin P, Limacher JM, et al. A novel metalloproteinase gene specifically expressed in stromal cells of breast carcinomas. Nature. 1990;348:699–704.
Yamamoto H, Itoh F, Adachi Y, Sakamoto H, Adachi M, Hinoda Y, et al. Relation of enhanced secretion of active matrix metalloproteinases with tumor spread in human hepatocellular carcinoma. Gastroenterology. 1997;112:1290–6.
Koskensalo S, Mrena J, Wiksten JP, Nordling S, Kokkola A, Hagström J, et al. MMP-7 overexpression is an independent prognostic marker in gastric cancer. Tumour Biol. 2010;31:149–55.
Davis NC, Evans EB, Cohen JR, Theile DE. Staging of colorectal cancer. The Australian clinico-pathological staging (ACPS) system compared with Dukes’ system. Dis Colon Rectum. 1984;27:707–13.
Yamashita K, Azumano I, Mai M, Okada Y. Expression and tissue localization of matrix metalloproteinase 7 (matrilysin) in human gastric carcinomas. Implications for vessel invasion and metastasis. Int J Cancer. 1998;79:187–94.
Railo M, Lundin J, Haglund C, von Smitten C, Nordling S. Ki-67, p53, ER receptors, ploidy and S phase as long-term prognostic factors in T1 node-negative breast cancer. Tumor Biol. 2007;28:45–51.
Hilsenbeck SG, Ravdin PM, deMoor CA, Chamness GC, Osborne CK, Clark GM. Time-dependence of hazard ratios for prognostic factors in primary breast cancer. Breast Cancer Res Treat. 1998;52:227–37.
Leivonen M, Nordling S, Lundin J, von Boguslawski K, Haglund C. STn and prognosis in breast cancer. Oncology. 2001;61:299–305.
Leivonen M, Nordling S, Lundin J, von Boguslawski K, Haglund C. p27 expression correlates with short-term, but not with long-term prognosis in breast cancer. Breast Cancer Res Treat. 2001;67:15–22.
Engellau J. Prognostic factors in soft tissue sarcoma. Tissue microarray for immunostaining, the importance of whole-tumor sections and time-dependence. Acta Orthop Scand Suppl. 2004;314:1–52.
Newell KJ, Witty JP, Rodgers WH, Matrisian LM. Expression and localization of matrix-degrading metalloproteinases during colorectal tumorigenesis. Mol Carcinog. 1994;10:199–206.
Kioi M, Yamamoto K, Higashi S, Koshikawa N, Fujita K, Miyazaki K. Matrilysin (MMP-7) induces homotypic adhesion of human colon cancer cells and enhances their metastatic potential in nude mouse model. Oncogene. 2003;22:8662–70.
Masaki T, Matsuoka H, Sugiyama M, Abe N, Goto A, Sakamoto A, et al. Matrilysin (MMP-7) as a significant determinant of malignant potential of early invasive colorectal carcinomas. Br J Cancer. 2001;84:1317–21.
Ishikawa T, Ichikawa Y, Mitsuhashi M, Momiyama N, Chishima T, Tanaka K, et al. Matrilysin is associated with progression of colorectal tumor. Cancer Lett. 1996;1(107):5–10.
Tokoro T, Okuno K, Hida J, Ishimaru E, Ueda K, Yoshifuji T. Depth of mesorectal invasion has prognostic significance in T3N0 low rectal cancer. Hepatogastroenterology. 2009;56:124–7.
Liebig C, Ayala G, Wilks JA, Berger DH, Albo D. Perineural invasion in cancer: a review of the literature. Cancer. 2009;115:3379–91.
Mimori K, Yamashita K, Ohta M, Yoshinaga K, Ishikawa K, Ishii H, et al. Coexpression of matrix metalloproteinase (MMP-7) and epidermal growth factor (EGF) receptor in colorectal cancer: an EGF receptor tyrosine kinase inhibitor is effective against MMP-7-expressing cancer cells. Clin Cancer Res. 2004;10:8243–9.
Acknowledgements
We thank Päivi Peltokangas, Tuire Koski, and Elina Aspiala for technical assistance.
Funding
This study was supported by Finska Läkaresällskapet, Medicinska Understödsföreningen Liv och Hälsa, and the Sigrid Jusélius Foundation.
Ethics approval
The study has been approved by local ethics committee and complies with the Declaration of Helsinki (Dnro HUS 226/E6/06).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Koskensalo, S., Louhimo, J., Nordling, S. et al. MMP-7 as a prognostic marker in colorectal cancer. Tumor Biol. 32, 259–264 (2011). https://doi.org/10.1007/s13277-010-0080-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13277-010-0080-2