Abstract
Background
Adriamycin (ADR) is an effective treatment for breast cancer; nevertheless, it is often linked with acquired resistance in breast cancer cells, reducing ADR’s therapeutic efficacy and increasing the risk of recurrence and poor prognosis. It has been revealed that the zinc-finger transcription factor pleomorphic adenoma gene like-2 (PLAGL2) is required for epithelial to mesenchymal transition (EMT) in cancer cells. Recent data indicates that PLAGL2 is also involved in regulating chemotherapeutic drug resistance, albeit the exact mechanism by which this happens remains unknown.
Objective
This study examines the effect of PLAGL2 on adriamycin resistance and EMT in breast cancer cells.
Methods
The small interfering RNA (siRNA) targeting PLAGL2 was transfected to breast cancer cells to alter PLAGL2 expression. Cell counting kit-8 (CCK-8) and colony formation assay detected cell growth and proliferation rate. Moreover, wound-healing and transwell assays were conducted to evaluate migration and invasion. Western blot (WB) checked the apoptosis and EMT-associated proteins.
Results
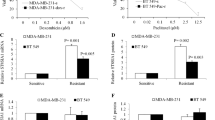
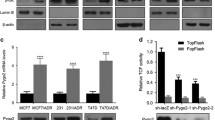
PLAGL2 expression is associated with breast cancer cells’ acquired resistance to ADR in this investigation. Additionally, deletion of PLAGL2 was associated with enhanced sensitivity to ADR, reduced proliferation, migration, and invasion capabilities, increased E-cadherin levels, and reduced Wnt6, β-catenin, and DVL1 levels in ADR-resistant breast cancer cells (MCF-7/ADR and MDA-MB-231/ADR cells). PLAGL2 could bind to the promoter region of Wnt6 and promote its expression. Additionally, the results of this research established that Wnt signaling is implicated in breast cancer cells’ resistance to ADR since BML-284, a Wnt signaling activator partly restored the sensitivity of MCF-7/ADR and MDA-MB-231/ADR cells to ADR.
Conclusion
PLAGL2 promotes adriamycin resistance and cell aggressiveness in breast cancer cells via activating the Wnt signaling pathway.






Similar content being viewed by others
Data availability
The data of this paper is available from the corresponding author on reasonable request.
References
Arnedos M, Vicier C, Loi S, Lefebvre C, Michiels S, Bonnefoi H, Andre F (2015) Precision medicine for metastatic breast cancer–limitations and solutions. Nat Rev Clin Oncol 12:693–704
Declercq J, Hensen K, Van De Ven W, Chavez M (2003) PLAG proteins: how they influence apoptosis and cell proliferation. Ann N Y Acad Sci 1010:264–265
Fischer KR, Durrans A, Lee S, Sheng J, Li F, Wong ST, Choi H, El Rayes T, Ryu S, Troeger J et al (2015) Epithelial-to-mesenchymal transition is not required for lung metastasis but contributes to chemoresistance. Nature 527:472–476
Hanker A, Sudhan D, Arteaga C (2020) Overcoming endocrine resistance in breast cancer. Cancer Cell 37:496–513
Hensen K, Van Valckenborgh I, Kas K, Van de Ven W, Voz M (2002) The tumorigenic diversity of the three PLAG family members is associated with different DNA binding capacities. Cancer Res 62:1510–1517
Li N, Li D, Du Y, Su C, Yang C, Lin C, Li X, Hu G (2019) Overexpressed PLAGL2 transcriptionally activates Wnt6 and promotes cancer development in colorectal cancer. Oncol Rep 41:875–884
Lim B, Lin Y, Navin N (2020) Advancing cancer research and medicine with single-cell genomics. Cancer Cell 37:456–470
Liu M, Yu X, Chen Z, Yang T, Yang D, Liu Q, Du K, Li B, Wang Z, Li S et al (2017) Aptamer selection and applications for breast cancer diagnostics and therapy. J Nanobiotechnol 15:81
Mackey JR, Martin M, Pienkowski T, Rolski J, Guastalla JP, Sami A, Glaspy J, Juhos E, Wardley A, Fornander T et al (2013) Adjuvant docetaxel, doxorubicin, and cyclophosphamide in node-positive breast cancer: 10-year follow-up of the phase 3 randomised BCIRG 001 trial. Lancet Oncol 14:72–80
Mallini P, Lennard T, Kirby J, Meeson A (2014) Epithelial-to-mesenchymal transition: what is the impact on breast cancer stem cells and drug resistance. Cancer Treat Rev 40:341–348
Merikhian P, Eisavand M, Farahmand L (2021) Triple-negative breast cancer: understanding Wnt signaling in drug resistance. Cancer Cell Int 21:419
Shome R, Ghosh S (2021) Tweaking EMT and MDR dynamics to constrain triple-negative breast cancer invasiveness by EGFR and Wnt/β-catenin signaling regulation. Cell Oncol (dordr) 44:405–422
Siegel RL, Miller KD, Jemal A (2020) Cancer statistics, 2020. CA Cancer J Clin 70:7–30
Sritharan S, Sivalingam N (2021) A comprehensive review on time-tested anticancer drug doxorubicin. Life Sci 278:119527
Stefanski C, Keffler K, McClintock S, Milac L, Prosperi J (2019) APC loss affects DNA damage repair causing doxorubicin resistance in breast cancer cells. Neoplasia (new York, NY) 21:1143–1150
Van Dyck F, Declercq J, Braem CV, Van de Ven WJ (2007) PLAG1, the prototype of the PLAG gene family: versatility in tumour development (review). Int J Oncol 30:765–774
Wu Y, Ginther C, Kim J, Mosher N, Chung S, Slamon D, Vadgama JV (2012) Expression of Wnt3 activates Wnt/β-catenin pathway and promotes EMT-like phenotype in trastuzumab-resistant HER2-overexpressing breast cancer cells. Mol Cancer Res 10:1597–1606
Wu L, Zhou Z, Han S, Chen J, Liu Z, Zhang X, Yuan W, Ji J, Shu X (2020) PLAGL2 promotes epithelial-mesenchymal transition and mediates colorectal cancer metastasis via β-catenin-dependent regulation of ZEB1. Br J Cancer 122:578–589
Wu L, Zhao N, Zhou Z, Chen J, Han S, Zhang X, Bao H, Yuan W, Shu X (2021) PLAGL2 promotes the proliferation and migration of gastric cancer cells via USP37-mediated deubiquitination of Snail1. Theranostics 11:700–714
Xu X, Zhang M, Xu F, Jiang S (2020) Wnt signaling in breast cancer: biological mechanisms, challenges and opportunities. Mol Cancer 19:165–165
Yang T, Huo J, Xu R, Su Q, Tang W, Zhang D, Zhu M, Zhan Y, Dai B, Zhang Y (2021) Selenium sulfide disrupts the PLAGL2/C-MET/STAT3-induced resistance against mitochondrial apoptosis in hepatocellular carcinoma. Clin Transl Med 11:e536
Zheng X, Carstens JL, Kim J, Scheible M, Kaye J, Sugimoto H, Wu CC, LeBleu VS, Kalluri R (2015) Epithelial-to-mesenchymal transition is dispensable for metastasis but induces chemoresistance in pancreatic cancer. Nature 527:525–530
Funding
This work has been financially supported by the Department of Pharmaceutical Administration, National Health Commission.
Author information
Authors and Affiliations
Contributions
YL and YL: conceived and designed the research; YL, RL, XH and WX: performed the experiments and analyzed the data. YL: wrote the manuscript. All authors read, commented, and accepted the final version.
Corresponding author
Ethics declarations
Conflict of interest
Yuxiao Li, Ruolin Liu, Xingzhao Han, Wei Xu and Yahui Liu declare that they have no conflict of interest.
Ethics approval
None.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Li, Y., Liu, R., Han, X. et al. PLAGL2 increases adriamycin resistance and EMT in breast cancer cells by activating the Wnt pathway. Genes Genom 45, 49–57 (2023). https://doi.org/10.1007/s13258-022-01330-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13258-022-01330-0