Abstract
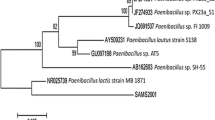
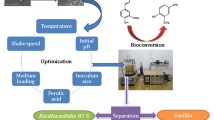
There is a growing interest in exploiting microbial bioconversions for the production of high value-added products such as vanillin and vanillic acid. We conducted this study to screen strains of bacteria which are able to degrade ferulic acid, and to evaluate the possibility of forming vanillic acid via conversion of ferulic acid under resting cell conditions. Twenty-seven different strains of bacteria were isolated from different samples which in turn were collected from a variety of sites in Iran and examined for bioconversion of ferulic acid into vanillic acid by thin-layer chromatography (TLC) and high-performance liquid chromatography (HPLC) analyses. Based on TLC and HPLC analyses, the strain SHL1 giving highest vanillic acid yield was selected and identified as Bacillus licheniformis (GenBank accession number HQ111516) based on morphological and physiochemical characteristics and its 16S rDNA gene sequence. In the presence of 1 g/l ferulic acid, vanillic acid reached a maximum value of 494 mg/l (molar yield of 60%) after 45 h of reaction time using resting cells of strain SHL1 without further optimization. The present study gives the first evidence for ferulic acid biotransformation by B. licheniformis.




Similar content being viewed by others
References
Abdelkafi S, Labat M, Gam ZBA, Lorquin J, Casalot L, Sayadi S (2008) Optimized conditions for the synthesis of vanillic acid under hypersaline conditions by Halomonas elongata DSM 2581 T resting cells. World J Microbiol Biotechnol 24:675–680
Abraham B, Berger RG, Schulz H (1994) Oakmoss impact volatiles generated by basidiomycete of the genus Polyporus. Flavour Fragr J 5:265
Achterholt S, Priefert H, Steinbüchel A (2000) Identification of Amycolatopsis sp. strain HR167 genes, involved in the bioconversion of ferulic acid to vanillin. Appl Microbiol Biotechnol 54:799–807
Allouche N, Damak M, Ellouz R, Sayadi S (2004) Use of whole cells of Pseudomonas aeruginosa for synthesis of the antioxidant hydroxytyrosol via conversion of tyrosol. Appl Environ Microbiol 70:2105–2109
Bock LH, Anderson JK (1955) Linear polyesters derived from vanillic acid. J Polym Sci 17:553–558
Brunati M, Marinelli F, Bertolini C, Gandolfi R, Daffonchio D, Molinari F (2004) Biotransformations of cinnamic and ferulic acid with actinomycetes. Enzyme Microb Technol 34:3–9
Crawford RL, Olson PP (1978) Microbial catabolism of vanillate: decarboxylation to guaiacol. Appl Environ Microbiol 36:539–543
Fadl AA, Nguyen AV, Khan MI (1995) Analysis of Salmonella enteritidis isolates by arbitrarily primed PCR. J Clin Microbiol 33:987–989
Falconnier B, Lapierre C, Lesage-Meessen L, Yonnet G, Brunerie P, Colonna Ceccaldi B, Corrieu G, Asther M (1994) Vanillin as a product of ferulic acid biotransformation by the white-rot fungus Pynnoporus cinnabarinus I-937: identification of metabolic pathways. J Biotechnol 37:123–132
Fitzgerald DJ, Stratfordb M, Narbada A (2003) Analysis of the inhibition of food spoilage yeasts by vanillin. Int J Food Microbiol 86:113–122
Gurujeyalakshmi G, Mahadevan A (1987) Dissimilation of ferulic acid by Bacillus subtilis. Curr Microbiol 16:69–73
Hagedorn S, Kaphammer B (1994) Microbial biocatalysts in the generation of flavor and fragrance chemicals. Annu Rev Microbiol 48:773–800
Huang Z, Dostal L, Rosazza JPN (1993) Microbial transformations of ferulic acid by Saccharomyces cerevisiae and Pseudomonas fluorescens. Appl Environ Microbiol 59:2244–2250
Huang Z, Dostal L, Rosazza JPN (1994) Mechanisms of ferulic acid conversions to vanillic acid and guaiacol by Rhodotorula rubra. J Biol Chem 268:23594–23598
Ishii T (1997) Structure and function of feruloylated polysaccharides. Plant Sci 127:111–127
Karmakar B, Vohra RM, Nandanwar H, Sharma P, Gupta KG, Sobti RC (2000) Rapid degradation of ferulic acid via 4-vinylguaiacol and vanillin by a newly isolated strain of Bacillus coagulans. J Biotechnol 80:195–202
Krings U, Berger RG (1998) Biotechnological production of flavors and fragrances. Appl Microbiol Biotechnol 49:1–8
Labuda IM, Goers SK, Keon KA (1992) Bioconversion process for the production of vanillin. Patent application US 5128253
Lesage-Meessen L, Delattre M, Haon M, Thibault JF, Ceccaldi BC, Brunerie P, Asther M (1996) A two-step bioconversion process for vanillin production from ferulic acid combining Aspergillus niger and Pycnoporus cinnabarinus. J Biotechnol 50:107–113
Mathew S, Abraham TE, Sudheesh S (2007) Rapid conversion of ferulic acid to 4-vinyl guaiacol and vanillin metabolites by Debaryomyces hansenii. J Mol Catal B Enzym 44:48–52
Mulheim A, Lerch K (1999) Towards a high-yield bioconversion of ferulic acid to vanillin. Appl Microbiol Biotechnol 51:456–461
Oosterveld A, Beldman G, Schols HA, Voragen AGJ (2000) Characterization of arabinose and ferulic acid rich pectic polysaccharides and hemicelluloses from sugar beet pulp. Carbohydr Res 328:185–197
Peng X, Misawa N, Harayama S (2003) Isolation and characterization of thermophilic bacilli degrading cinnamic, 4-coumaric, and ferulic acids. Appl Environ Microbiol 69:1417–1427
Pometto AL, Crawford DL (1983) Whole-cell bioconversion of vanillin to vanillic acid by Streptomyces viridosporus. Appl Environ Microbiol 45:1582–1585
Priefert H, Babenhorst J, Steinbuchel A (2001) Biotechnological production of vanillin. Appl Microbiol Biotechnol 56:296–314
Ramachandra RS, Ravishankar GA (2000) Vanilla flavour: production by conventional and biotechnological routes. J Sci Food Agric 80:289–304
Rosazza JON, Huang Z, Dostal L, Rosseau B (1995) Biocatalytic transformation of ferulic acid: an abundant aromatic natural product. J Ind Microbiol 15:457–471
Sambrook J, Fritsch EF, Maniatis T (1989) Molecular Cloning: A Laboratory Manual. Cold Spring Harbor Laboratory, Cold Spring Harbor, NY
Schrader J, Etschmann MMW, Sell D, Hilmer JM, Rabenhorst J (2004) Applied biocatalysis for the synthesis of natural flavor compound-Current industrial processes and future prospects. Biotechnol Lett 26:463–472
Serra S, Fuganti C, Brenna E (2005) Biocatalytic preparation of natural flavours and fragrances. Trends Biotechnol 23:193–198
Shashwati G, Ashish S, Sukanta KS, Adinpunya M (2007) Microbial transformation of ferulic acid to vanillic acid by Streptomyces sannanensis MTCC 6637. J Ind Microbiol Biotechnol 34:131–138
Shimoni E, Ravid U, Shoham Y (2000) Isolation of a Bacillus sp. capable of transforming isoeugenol to vanillin. J Biotechnol 78:1–9
Simbert RM, Krieg NR (1994) Phenotypic characterization. In: Gerhardt P, Murray RGE, Wood WA, Krieg NR (eds) Methods for general and molecular bacteriology. Washington, DC, pp. 607–654.
Studholme DJ, Jackson RA, Leak DJ (1999) Phylogenetic analysis of transformable strains of thermophilic Bacillus species. FEMS Microbiol Lett 172:85–90
Tamura K, Dudley J, Nei M, Kumar S (2007) Molecular Evolutionary Genetics Analysis (MEGA) software version 4.0. Mol Biol Evol 24(8):1596–1599
Toms A, Wood JM (1970) The degradation of trans-feruic acid by Pseudomonas acidovorans. Biochemistry 9:337–343
Torre P, De Faveri D, Perego P, Ruzzi M, Barghini P, Gandolfi R, Converti A (2004) Bioconversion of ferulate into vanillin by Escherichia coli strain JM109/pBB1 in an immobilized-cell reactor. Ann Microbiol 54(4):517–527
Tsujiyama SI, Ueno M (2008) Formation of 4-vinyl guaiacol as an intermediate in bioconversion of ferulic acid by Schizophyllum commune. Biosci Biotechnol Biochem 72(1):212–215
Walton NJ (2003) Molecules of interest: vanillin. Phytochemistry 63:505–515
Xu P, Hua D, Ma C (2007) Microbial transformation of propenylbenzenes for natural flavor production. Trends Biotechnol 25:571–576
Acknowledgement
This study was supported by a grant from the Postgraduate Administration Office of the University of Isfahan to M.Ashengroph for obtaining a PhD degree
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ashengroph, M., Nahvi, I., Zarkesh-Esfahani, H. et al. Novel strain of Bacillus licheniformis SHL1 with potential converting ferulic acid into vanillic acid. Ann Microbiol 62, 553–558 (2012). https://doi.org/10.1007/s13213-011-0291-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13213-011-0291-9