Abstract
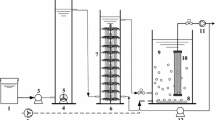
This work describes the performance of a sequencing batch reactor (SBR) and the involvement of a novel reconstituted bacterial consortium in olive mill wastewater (OMW) treatment. The organic loading rate applied to the SBR was serially increased in terms of initial COD from 10 to 75 g L−1 to allow gradual acclimatization of activated sludge to high concentrations of toxic compounds in OMW. After the acclimatization period, up to 60% of the total COD content were effectively biodegraded from OMW at 75 g L−1 COD within 30 day hydraulic retention time. The diversity and community composition of cultivable bacteria participating in the aerobic process of treating OMW were further assessed. A total of 91 bacterial strains were isolated from the reactor and analyzed by amplification of the 16S-23S rRNA internal transcribed spacer (ITS) region and by 16S rRNA gene sequencing. The most abundant phylum was Firmicutes (57.1%) followed by Proteobacteria (35.2%) and Actinobacteria (7.7%). The use of the Biolog® Phenotype Microarray system to evaluate the ability of isolated strains to utilize OMW phenolic compounds is reported in this work for the first time. Interestingly, results showed that all species tested were able to utilize phenolics as sole carbon and energy sources. The removals of COD and phenolics from undiluted OMW by the reconstituted bacterial consortium were almost similar to those obtained by the acclimatized activated sludge, which suggest that cultivable bacteria play the major role in OMW biodegradation. Phytotoxicity assays using tomato seeds showed a significant improvement of seed germination values for treated OMW. Our overall results suggest that the novel developed bacterial consortium could be considered as a good prospect for phenolics-rich wastewaters bioremediation applications.





Similar content being viewed by others
References
Abraham J, Silambarasan S (2016) Biodegradation of chlorpyrifos and its hydrolysis product 3,5,6-trichloro-2-pyridinol using a novel bacterium Ochrobactrum sp. JAS2: a proposal of its metabolic pathway. Pestic Biochem Phys 126:13–21
AFNOR (1983) Recueil de norme française: eau, méthodes d’essai, 2éme édition, Paris
Aggelis G, Iconomou D, Christou M, Bokas D, Kotzailias S, Christou G, Tsagou V, Papanikolaou S (2003) Phenolic removal in a model olive oil mill wastewater using Pleurotusostreatus in bioreactor cultures and biological evaluation of the process. Water Res 37:3897–3904
Aguilera M, Monteoliva-Sánchez M, Suárez A, Guerra V, Lizama C, Bennasar A, Ramos-Cormenzana A (2001) Paenibacillus jamilae sp. nov., an exopolysaccharide-producing bacterium able to grow in olive-mill wastewater. Int J Syst Evol Micr 51:1687–1692
Aissam H, Penninckx MJ, Benlemlih M (2007) Reduction of phenolics content and COD in olive oil mill wastewaters by indigenous yeasts and fungi. World J Microb Biot 23:1203–1208
Ammar E, Nasri M, Medhioub K (2005) Isolation of Enterobacteria able to degrade simple aromatic compounds from the wastewater from olive oil extraction. World J Microb Biot 21:253–259
APHA (1998) Standard methods for the examination of water and wastewater, 12 edn. American Public Health Association, Washington
Babiak P, Kyslíková E, Stepánek V, Vale Sová R, Palyzová A, Maresová H, Hájícek J, Kyslík P (2011) Whole-cell oxidation of omeprazole sulfide to enantiopure esomeprazole with Lysinibacillus sp. B71. Bioresour Technol 102:7621–7626
Basu A, Phale PS (2006) Inducible uptake and metabolism of glucose by the phosphorylative pathway in Pseudomonas putida CSV86. FEMS Microbiol Lett 259:311–316
Beccari M, Bonemazzi F, Majone M, Riccardi C (1996) Interaction between acidoge-nesis and metanogenesis in the anaerobic treatment of olive oil mill effluent. Water Res 30:183–189
Benitez FJ, Beltran-Heredia J, Torregrosa J, Acero JL, Cercas V (1997) Aerobic degradation of olive mill wastewaters. Appl Microbiol Biot 47:185–188
Bezzaa FA, Beukes M, Nkhalambayausi Chirwaa EM (2015) Application of biosurfactant produced by Ochrobactrum intermedium CN3 for enhancing petroleum sludge bioremediation. Process Biochem 50:1911–1922
Cappelletti M, Presentato A, Milazzo G, Turner RJ, Fedi S, Frascari D, Zannoni D (2015) Growth of Rhodococcus sp. strain BCP1 on gaseous n-alkanes: new metabolic insights and transcriptional analysis of two soluble di-iron monooxygenase genes. Front Microbiol 6:393–407
Chatzisymeon E, Foteinis S, Mantzavinos D, Tsoutsos T (2013) Life cycle assessment of advanced oxidation processes for olive mill wastewater treatment. J Clean Prod 54:229–234
Chiavola A, Farabegoli G, Antonetti F (2014) Biological treatment of olive mill wastewater in a sequencing batch reactor. Biochem Eng J 85:71–78
Collier DN, Hager PW, Phibbs PVJr (1996) Catabolite repression control in the pseudomonads. Res Microbiol 147:551–561
Daâssi D, Belbahri L, Vallat A, Woodward S, Nasri M, Mechichi T (2014) Enhanced reduction of phenol content and toxicity in olive mill wastewaters by a newly isolated strain of Coriolopsis gallica. Environ Sci Pollut R 21:1746–1758
de Carvalho CC, Costa SS, Fernandes P, Couto I, Viveiros M (2014) Membrane transport systems and the biodegradation potential and pathogenicity of genus Rhodococcus. Front Physiol 5:133–145
El-Abbassi A, Kiai H, Raiti J, Hafidi A (2014) Application of ultrafiltration for olive processing wastewaters treatment. J Clean Prod 65:432–438
Ertuğrul S, Dönmez G, Takaç S (2007) Isolation of lipase producing Bacillus sp. from olive mill wastewater and improving its enzyme activity. J Hazard Mater 149:720–724
Fang W, Fang Z, Peng Z, Chang F, Hong Y, Zhang X, Peng H, Xiao Y (2012) Evidence for lignin oxidation by the giant panda fecal microbiome. PLoS One 7:1–10
Hasan SA, Jabeen S (2015) Degradation kinetics and pathway of phenol by Pseudomonas and Bacillus species. Biotechnol Biotech Equ 29:45–53
He Z, Niu C, Lu Z (2014) Individual or synchronous biodegradation of di-n-butyl phthalate and phenol by Rhodococcusruber strain DP-2. J Hazard Mater 273:104–109
Heiss GS, Gowan B, Dabbs ER (2006) Cloning of DNA from a Rhodococcus strain conferring the ability to decolorize sulfonated azo dyes. FEMS Microbiol Lett 99:221–226
Henriques AO, Moran CPJr (2000) Structure and assembly of the bacterial endospore coat. Methods 20:95–110
Hou J, Liu F, Wu N, Ju J, Yu B (2016) Efficient biodegradation of chlorophenols in aqueous phase by magnetically immobilized aniline-degrading Rhodococcus rhodochrous strain. J Nanobiotechnol 14:1–8
Institut National De La Normalisation Et De La Propriete Industrielle (1989) Environment protection—use of reclaimed water for agricultural purposes—physical, chemical and biological specifications (in French), Tunisian standards, INNORPI, NT 106.03
Jain PK, Gupta VK, Pathak H, Lowry M, Jaroli DP (2010) Characterization of 2T engine oil degrading indigenous bacteria, isolated from high altitude (Mussoorie). India World J Microb Biotechnol 26:1419–1426
Jaouani A, Guillen F, Penninckx MJ, Martinez AT, Martinez MJ (2005a) Role of Pycnoporus coccineus laccase in the degradation of aromatic compounds in olive oil mill wastewater. Enzyme Microb Techol 36:478–486
Jaouani A, Vanthournhout M, Penninckx MJ (2005b) Olive oil mill wastewater purification by combination of coagulation- flocculation and biological treatments. Environ Technol 26:633–641
Jarboui R, Sellami F, Azri C, Gharsallah N, Ammar E (2010) Olive mill wastewater evaporation management using PCA method: case study of natural degradation in stabilization ponds (Sfax, Tunisia). J Hazard Mater 176:992–1005
Johnson J, Shah B, Jain K, Parmar N, Hinsu A, Patel N, Chaitanya GJ, Madamwara D (2016) Draft genome sequence of Paenibacillus sp. strain DMB5, acclimatized and enriched for catabolizing anthropogenic compounds. Am Soc Microbiol 4:e00211–e00216
Kavvadias V, Doulaa MK, Komnitsas K, Liakopouloua N (2010) Disposal of olive oil mill wastes in evaporation ponds: effects on soil properties. J Hazard Mater 182:144–155
Khairy H, Wübbeler JH, Steinbüchel A (2015) Biodegradation of the organic disulfide 4, 4-dithiodibutyric acid by Rhodococcus spp. Appl Environ Microbiol 81:8294–8306
Komilis DP, Karatzas E, Halvadakis CP (2005) The effect of olive mill wastewater on seed germination after various pretreatment techniques. Australas J Environ Manag 74:339–348
Kostka JE, Prakash O, Overholt WA, Green SJ, Freyer G, Canion A, Delgardio J, Norton N, Hazen TC, Huettel M (2011) Hydrocarbon degrading bacteria and the bacterial community response in gulf of Mexico beach sands impacted by the deepwater horizon oil spill. Appl Environ Microbiol 77:7962–7974
Kotake T, Matsuzawa J, Suzuki-Minakuchi C, Okada K, Nojiri H, Iwata K (2016) Purification and partial characterization of the extradioldioxygenase, 2- carboxy-2, 3-dihydroxybiphenyl 1, 2-dioxygenase, in the fluorene degradation pathway from Rhodococcussp. strain DFA3. Biosci Biotechnol Biochem 80:1–7
Kumar S, Mishra VK, Kumar U, Kumar A, Varghese S (2013) Biodegradation of phenol by bacterial strains and their catalytic ability. Intl J Agric Environ Biotechnol 6:108–115
Kurade MB, Waghmode TR, Khandare RV, Jeon B-H, Govindwar SP (2016) Biodegradation and detoxification of textile dye Disperse Red 54 by Brevibacillus laterosporus and determination of its metabolic fate. J Biosci Bioeng 121:442–449
Lanciotti R, Gianotti A, Baldi D, Angrisani R, Suzzi G, Mastrocola D, Guerzoni ME (2005) Use of Yarrowia lipolytica strains for the treatment of olive mill wastewater. Bioresour Technol 96:317–322
McNamara CJ, Anastasiou CC, O’Flaherty V, Mitchell R (2008) Bioremediation of olive mill wastewater. Int Biodeter Biodegr 61:127–134
Mekki A, Arous F, Aloui F, Sayadi S (2017) Treatment and Valorization of Agro-wastes as Biofertilizers. Waste Biomass Valor 8:611–619
Michael I, Panagi A, Ioannou LA, Frontistis Z, Fatta-Kassinos D (2014) Utilizing solar energy for the purification of olive mill wastewater using a pilot-scale photocatalytic reactor after coagulation-flocculation. Water Res 60:28–40
Mondal KC, Banerjee D, Jana M, Pati BR (2001) Colorimetric assay method for determination of the tannin acyl hydrolase activity. Anal Biochem 295:168–171
Naclerio G, Antonio F, Petrella E, Nerone V, Cocco F, Celico F (2010) Potential role of Bacillus endospores in soil amended by olive mill wastewater. Water Sci Technol 61:2873–2879
Nawahwi MZ, Ibrahim Z, Yahya A (2013) Degradation of the Azo Dye Reactive Red 195 by Paenibacillus spp. R2. J Bioremed Biodeg 4:174–180
Neifar M, Jaouani A, Martínez MJ, Penninckx MJ (2012) Comparative study of olive oil mill wastewater treatment using free and immobilized Coriolopsis polyzona and Pycnoporus coccineus. J Microbiol 50:746–753
Ntougias S, Baldrian P, Ehaliotis C, Nerud F, Antoniou T, Merhautová V, Zervakis GI (2012) Biodegradation and detoxification of olive mill wastewater by selected strains of the mushroom genera Ganoderma. and Pleurotus. Chemosphere 88:620–626
Obied HK, Bedgood DR, Prenzler PD, Robards K (2007) Bioscreening of Australian olive mill waste extracts: biophenol content, antioxidant, antimicrobial and molluscicidalactivities. Food Chem Toxicol 45:1238–1248
Özgün ÖK, Özkök İP, Kutay C, Orhon D (2016) Characteristics and biodegradability of olive mill wastewaters. Environ Technol 37:1240–1248
Paraskeva P, Diamadopoulos E (2006) Technologies for olive mill wastewater (OMW) treatment: a review. J Chem Technol Biotechnol 81:1475–1485
Paredes C, Cegarra J, Bernal MP, Roig A (2005) Influence of olive mill wastewater in composting and impact of the compost on a Swiss chard crop and soil properties. Environ Int 31:305–312
Ranalli A (1992) Microbiological treatment of oil mill waste waters. Grasas Aceites 43:16–19
Rincòn B, Borja R, Gonzàlez JM, Portillo MC, Sàiz-Jiménez C (2008) Influence of organic loading rate and hydraulic retention time on the performance, stability and microbial communities of one-stage anaerobic digestion of two-phase olive mill solid residue. Biochem Eng J 40:253–261
Rusan MJ, Albalasmeh AA, Zuraiqi S, Bashabsheh M (2015) Evaluation of phytotoxicity effect of olive mill wastewater treated by different technologies on seed germination of barley (Hordeum vulgare L.). Environ Sci Pollut R 22:9127–9135
Saitou N, Nei M (1987) The neighbor-joining method: a new method for reconstructing phylogenetic trees. Mol Biol Evol 4:406–425
Saratale RG, Gandhi SS, Purankar MV, Kurade MB, Govindwar SP, Oh SE, Saratale GD (2013) Decolorization and detoxification of sulfonated azo dye C.I. Remazol Red and textile effluent by isolated Lysinibacillus sp. RGS. J Biosci Bioeng 115:658–667
Scalbert A (2012) Antimicrobial properties of tannins. Phytochemistry 30:3875–3883
Singh B, Kaur J, Singh K (2012) Transformation of malathion by Lysinibacillus sp. isolated from soil. Biotechnol Lett 34:863–867
Swain T, Hillis WE (1959) The quantitative analysis of phenolic constituents of Prunus domestica. J Sci Food Agric 10:63–68
Tamura K, Dudley J, Nei M (2007) MEGA4: molecular evolutionary genetics analysis (MEGA) software version 4.0. Mol Biol Evol 24:1596–1599
Tsiamis G, Tzagkaraki G, Chamalaki A, Xypteras N, Andersen G, Vayenas D, Bourtzis K (2012) Olive-Mill wastewater bacterial communities display a cultivar specific profile. Curr Microbiol 64:197–203
Tziotzios G, Michailakis S, Vayenas DV (2007) Aerobic biological treatment of olive mill wastewater by olive pulp bacteria. Int Biodeter Biodegr 60:209–214
Ventura M, Canchaya C, Tauch A, Chandra G, Fitzgerald GF, Chater KF, Van Sinderen D (2007) Genomics of actinobacteria: tracing the evolutionary history of an ancient phylum. Microbiol Mol Biol R 71:495–548
Yadav TC, Khardenavis AA, Kapley A (2014) Shifts in microbial community in response to dissolved oxygen levels in activated sludge. Bioresour Technol 165:257–264
Yang HY, Jia RB, Chen B, Li L (2014) Degradation of recalcitrant aliphatic and aromatic hydrocarbons by a dioxin-degrader Rhodococcus sp. strain p52. Environ Sci Pollut Res 21:11086–11093
Zerva A, Zervakis GI, Christakopoulos P, Topakas E (2016) Degradation of olive mill wastewater by the induced extracellular ligninolytic enzymes of two wood-rot fungi. J Environ Manag 203:791–798
Zouari N, Ellouz R (1996) Microbial consortia for the aerobic degradation of aromatic compounds in olive oil mill effluent. J Ind Microbiol 16:155–162
Zu L, Xiong J, Li G, Fang Y, An T (2014) Concurrent degradation of tetrabromobisphenol A by Ochrobactrum sp. T under aerobic condition and estrogenic transition during these processes. Ecotoxicol Environ Safe 104:220–225
Acknowledgements
This work was supported by the Ministry of Higher Education and Scientific Research of the Tunisian Republic and the MADFORWATER Horizon 2020 Research and Innovation Program [grant agreement No. 688320].
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest.
Rights and permissions
About this article
Cite this article
Arous, F., Hamdi, C., Kmiha, S. et al. Treatment of olive mill wastewater through employing sequencing batch reactor: performance and microbial diversity assessment. 3 Biotech 8, 481 (2018). https://doi.org/10.1007/s13205-018-1486-6
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s13205-018-1486-6