Abstract
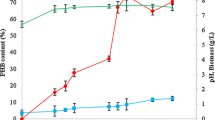
Experimental investigations were carried out for Cupriavidus necator (MTCC 1472)-based improved production of poly-3 hydroxy butyrate (PHB) through induced nitrogen limiting fed-batch cultivation strategies. Initially Plackett–Burman design and response surface methodology were implemented to optimize most influencing process parameters. With optimized process parameter values, continuous feeding strategies ware applied in a 5-l fermenter with table sugar concentration of 100 g/l, nitrogen concentration of 0.12 g/l for fed-batch fermentation with varying dilution rates of 0.02 and 0.046 1/h. To get enriched production of PHB, concentration of the sugar was further increased to 150 and 200 g/l in feeding. Maximum concentrations of PHB achieved were 22.35 and 23.07 g/l at those dilution rates when sugar concentration maintains at 200 g/l in feeding. At maximum concentration of PHB (23.07 g/l), productivity of 0.58 g/l h was achieved with maximum PHB accumulation efficiency up to 64% of the dry weight of biomass. High purity of PHB, close to medical grade was achieved after surfactant hypochlorite extraction method, and it was further confirmed by SEM, EDX, and XRD studies.








Similar content being viewed by others
References
Ahn WS, Park SJ, Lee SY (2001) Production of poly(3-hydroxybutyrate) from whey by cell recycle fed-batch culture of recombinant Escherichia coli. Biotechnol Lett 23:235–240. doi:10.1023/A:1005633418161
Arifin Y, Sabri S, Sugiarto H et al (2010) Deletion of cscR in Escherichia coli W improves growth and poly-3-hydroxybutyrate (PHB) production from sucrose in fed batch culture. J Biotechnol 156:275–278. doi:10.1016/j.jbiotec.2011.07.003
Bengtsson S, Werker A, Christensson M, Welander T (2008) Production of polyhydroxyalkanoates by activated sludge treating a paper mill wastewater. Bioresour Technol 99:509–516. doi:10.1016/j.biortech.2007.01.020
Bengtsson S, Pisco AR, Reis MAM, Lemos PC (2010) Production of polyhydroxyalkanoates from fermented sugar cane molasses by a mixed culture enriched in glycogen accumulating organisms. J Biotechnol 145:253–263. doi:10.1016/j.jbiotec.2009.11.016
Choi J, Lee SY (1999) Factors affecting the economics of polyhydroxyalkanoate production by bacterial fermentation. Appl Microbiol Biotechnol 51:13–21
Cui ZF, Muralidhara HS (eds) (2010) Membrane technology a practical guide to membrane technology and applications in food and bioprocessing. Elsevier, Amsterdam
Dong Z, Sun X (2000) A new method of recovering polyhydroxyalkanoate from Azotobacter chroococcum. Chin Sci Bull 45:252–256. doi:10.1007/BF02884685
Doran PM (1995) An impart of Elsevier
Du G, Chen J, Yu J, Lun S (2001) Continuous production of poly-3-hydroxybutyrate by Ralstonia eutropha in a two-stage culture system. J Biotechnol 88:59–65. doi:10.1016/S0168-1656(01)00266-8
Fahmy R, Kona R, Dandu R et al (2012) Quality by design I: application of failure mode effect analysis (FMEA) and Plackett–Burman design of experiments in the identification of “main factors” in the formulation and process design space for roller-compacted ciprofloxacin hydrochloride immediate-release tablets. AAPS PharmSciTech 13:1243–1254. doi:10.1208/s12249-012-9844-x
Gahlawat G, Sengupta B, Srivastava AK (2012) Enhanced production of poly(3-hydroxybutyrate) in a novel airlift reactor with in situ cell retention using Azohydromonas australica. J Ind Microbiol Biotechnol 39:1377–1384. doi:10.1007/s10295-012-1138-5
García A, Segura D, Espín G et al (2014) High production of poly-β-hydroxybutyrate (PHB) by an Azotobacter vinelandii mutant altered in PHB regulation using a fed-batch fermentation process. Biochem Eng J 82:117–123. doi:10.1016/j.bej.2013.10.020
Grothe E, Moo-Young M, Chisti Y (1999) Fermentation optimization for the production of poly(β-hydroxybutyric acid) microbial thermoplastic. Enzyme Microb Technol 25:132–141. doi:10.1016/S0141-0229(99)00023-X
Hafuka A, Sakaida K, Satoh H et al (2011) Effect of feeding regimens on polyhydroxybutyrate production from food wastes by Cupriavidus necator. Bioresour Technol 102:3551–3553. doi:10.1016/j.biortech.2010.09.018
Ienczak JL, Schmidt M, Quines LK et al (2016) Poly(3-hydroxybutyrate) production in repeated fed-batch with cell recycle using a medium with low carbon source concentration. Appl Biochem Biotechnol 178:408–417. doi:10.1007/s12010-015-1883-9
Horwitz W (ed) (1980) Official methods of analysis of the Association of Official Analytical Chemists. Association of Official Analytical Chemists, Washington
Kaplan DL (1998) Introduction to biopolymers from renewable resources. In: Kaplan DL (ed) Biopolymers from renewable resources. Macromolecular systems — Materials approach. Springer, Berlin
Ke Y, Wu G, Wang Y (2014) PHBV/PAM scaffolds with local oriented structure through UV polymerization for tissue engineering. Biomed Res Int 2014:1–9. doi:10.1155/2014/157987
Khanna S, Srivastava AK (2005a) Recent advances in microbial polyhydroxyalkanoates. Process Biochem 40:607–619
Khanna S, Srivastava AK (2005b) Statistical media optimization studies for growth and PHB production by Ralstonia eutropha. Process Biochem 40:2173–2182. doi:10.1016/j.procbio.2004.08.011
Khanna S, Srivastava AK (2008a) Continuous production of poly-β-hydroxybutyrate by high-cell-density cultivation of Wautersia eutropha. J Chem Technol Biotechnol 83:799–805. doi:10.1002/jctb.1868
Khanna S, Srivastava AK (2008b) Productivity enhancement of poly-(β-hydroxybutyrate) by fed-batch cultivation of nutrients using variable (decreasing) nutrient rate by Wautersia eutropha. Chem Eng Commun 195:1424–1436. doi:10.1080/00986440801964087
Koller M, Muhr A (2014) Continuous production mode as a viable process-engineering tool for efficient poly(hydroxyalkanoate) (PHA) bio-production. Chem Biochem Eng Q 28:65–77
Koller M, Bona R, Hermann C et al (2005) Biotechnological production of poly(3-hydroxybutyrate) with Wautersia eutropha by application of green grass juice and silage juice as additional complex substrates. Biocatal Biotransform 23:329–337. doi:10.1080/10242420500292252
Kulpreecha S, Boonruangthavorn A, Meksiriporn B, Thongchul N (2009) Inexpensive fed-batch cultivation for high poly(3-hydroxybutyrate) production by a new isolate of Bacillus megaterium. J Biosci Bioeng 107:240–245. doi:10.1016/j.jbiosc.2008.10.006
Kunasundari B, Sudesh K (2011) Isolation and recovery of microbial polyhydroxyalkanoates. Express Polym Lett 5:620–634
Lee J, Lee SY, Park S, Middelberg AP (1999) Control of fed-batch fermentations. Biotechnol Adv 17:29–48
Lee SH, Oh DH, Ahn WS et al (2000) Production of poly(3-hydroxybutyrate-co-3-hydroxyhexanoate) by high-cell-density cultivation of Aeromonas hydrophila. Biotechnol Bioeng 67:240–244. doi:10.1002/(SICI)1097-0290(20000120)67:2<240:AID-BIT14>3.0.CO;2-F
Miller GL (1959) Use of dinitrosalicylic acid reagent for determination of reducing sugar. Anal Chem 31:426–428. doi:10.1021/ac60147a030
Mozumder MSI, De Wever H, Volcke EIP, Garcia-Gonzalez L (2014) A robust fed-batch feeding strategy independent of the carbon source for optimal polyhydroxybutyrate production. Process Biochem 49:365–373. doi:10.1016/j.procbio.2013.12.004
Narayanan A, Ramana KV (2012) Polyhydroxybutyrate production in Bacillus mycoides DFC1 using response surface optimization for physico-chemical process parameters. 3Biotech 2:287–296. doi:10.1007/s13205-012-0054-8
Osman Y, Elrazak AA, Khater W (2016) Bioprocess optimization of microbial biopolymer production. J Biobased Mater Bioenergy 10:119–128. doi:10.1166/jbmb.2016.1582
Panayotidou E, Kroustalli A, Baklavaridis A, Deligianni D (2014) XRD diffraction patterns of pure PHB and the PHB/3 wt% C 18 MMT hybrid material—Figure 1 of 10. J Appl Polym Sci 132(11). https://www.researchgate.net/figure/268280877_fig1_Figure-2-XRD-diffraction-patterns-of-pure-PHB-and-the-PHB-3-wt-C-18-MMT-hybrid. Accessed 1 May 2017
Pohlmann A, Fricke WF, Reinecke F et al (2006) Genome sequence of the bioplastic-producing “Knallgas” bacterium Ralstonia eutropha H16. Nat Biotechnol 24:1257–1262. doi:10.1038/nbt1244
Ramsay JA, Berger E, Ramsay BA, Chavarie C (1990) Recovery of poly-3-hydroxyalkanoic acid granules by a surfactant-hypochlorite treatment. Biotechnol Tech 4:221–226. doi:10.1007/BF00158833
Rohini D, Phadnis S, Rawal SK (2006) Synthesis and characterization of poly-β-hydroxybutyrate from Bacillus thuringiensis R1. Indian J Biotechnol 5:276–283
Sabra W, Zeng AP, Lünsdorf H, Deckwer WD (2000) Effect of oxygen on formation and structure of Azotobacter vinelandii alginate and its role in protecting nitrogenase. Appl Environ Microbiol 66:4037–4044
Schmidt M, Ienczak JL, Quines LK et al (2016) Poly(3-hydroxybutyrate-co-3-hydroxyvalerate) production in a system with external cell recycle and limited nitrogen feeding during the production phase. Biochem Eng J 112:130–135. doi:10.1016/j.bej.2016.04.013
Valappil SP, Misra SK, Boccaccini AR et al (2007) Large-scale production and efficient recovery of PHB with desirable material properties, from the newly characterised Bacillus cereus SPV. J Biotechnol 132:251–258. doi:10.1016/j.jbiotec.2007.03.013
van Wegen RJ, Lee SY, Middelberg AP (2001) Metabolic and kinetic analysis of poly(3-hydroxybutyrate) production by recombinant Escherichia coli. Biotechnol Bioeng 74:70–80
Vijayendra SVN, Rastogi NK, Shamala TR et al (2007) Optimization of polyhydroxybutyrate production by Bacillus sp. CFR 256 with corn steep liquor as a nitrogen source. Indian J Microbiol 47:170–175. doi:10.1007/s12088-007-0033-7
Acknowledgements
The authors are thankful to the Government of India for financial support under the Technical Education Quality Improvement Programme (TEQIP) Phase‐II.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interests.
Rights and permissions
About this article
Cite this article
Dey, P., Rangarajan, V. Improved fed-batch production of high-purity PHB (poly-3 hydroxy butyrate) by Cupriavidus necator (MTCC 1472) from sucrose-based cheap substrates under response surface-optimized conditions. 3 Biotech 7, 310 (2017). https://doi.org/10.1007/s13205-017-0948-6
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s13205-017-0948-6