Abstract
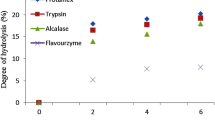
In this research enzymatic hydrolysis of rice bran protein concentrate (RBPC) and soybean Protein (SBP) as control were studied with 3 commercial enzymes (Alcalase®, Papain and acommercial 3-enzyme cocktail containing of 1.6 mg ml−1 Trypsin, 3.1 mg ml−1 Chymotrypsin, 1.3 mg ml−1Aminopeptidase (SIGMA P7500) and 7.95 mg ml−1pronase type XIV (SIGMA P5147) by the pH stat method. The hydrolysis was carried out at temperature of 28 C, 60 min and pH 8.00. Results were showed that RBPC, and SBP had higher Degree hydrolysis (DH %) with Alcalase® enzyme. Alcalase®had stronger capability for hydrolysis compared to the other tested enzymes. After 60 minute of hydrolysis time, the DH% of Alcalase® for RBPC and SBP was 12.69 and 12.50 %, respectively. In contrast, papain enzyme was showed lowest DH% in three substrates that 1.56 and 1.24 % were for SBP and RBPC, respectively.The hydrolysis of the protein fraction performed the three enzymes on the two substrates was followed in SDS-PAGE. RBPC and SBP showed almost complete digestion with Alcalase® enzyme after 60 minutes. 3-enzyme cocktail enzyme hydrolyzed better the RBPC than the SBP. Papain enzyme had less effect on the two substrates than other 2 enzymes. It was found that Alcalase® has highest capability for hydrolysis compared to other enzyme preparations. The high value protein hydrolysates prepared by Alcalase® can be used as value added ingredients in many food formulations. They are also suitable for a broad range of industrial food applications and also for cosmetic and personal care products.



Similar content being viewed by others
References
Adler-Nissen J (1979) Determination of the degree of hydrolysis of food protein hydrolysates by trinitrobenzenesulfonic acid. J Agric Food Chem 27:1256–1262
Adler-Nissen J (1986) Enzymic hydrolysis of food proteins. Elsevier Applied Science Publishers, Barking, Essex, UK, 427 pp
Aluko R, Monu E (2003) Functional and bioactive properties of quinoa seed protein hydrolysates. J Food Sci 68:1254–1258
Apar DK, Özbek B (2010) Corn Gluten hydrolysis by Alcalase: Kinetics of Hydrolysis. Chem Eng Commun 197:963–973
Barber S, De Barber CB (1991) Rice bran: chemistry and technology Rice: Utilization 2:313
Clemente A (2000) Enzymatic protein hydrolysates in human nutrition. Trends Food Sci Technol 11:254–262
Dawe D, Pandey S, Nelson A (2010) Emerging trends and spatial patterns of rice production Rice in the Global Economy: Strategic Research and Policy Issues for Food Security Los Baños, Philippines: International Rice Research Institute (IRRI) p 15–35
Demirhan E, Apar DK, Özbek B (2011) Sesame cake protein hydrolysis by alcalase: effects of process parameters on hydrolysis, solubilisation, and enzyme inactivation. Korean J Chem Eng 28:195–202
Ezquerra JM, García-Carreño FL, Civera R, Haard NF (1997) pH-stat method to predict protein digestibility in white shrimp (penaeus vannamei). Aquaculture 157:251–262
Garcia-Carreno FL, Hernandez-Cortes MP, Haard NF (1994) Enzymes with peptidase and proteinase activity from the digestive systems of a freshwater and a marine decapod. J Agric Food Chem 42:1456–1461
García-Carreño FL, Navarrete del Toro A, Ezquerra M (1997) Digestive shrimp proteases for evaluation of protein digestibility in vitro. I: effect of protease inhibitors in protein ingredients. J Mar Biotechnol 5:36–40.
Helm R, Burks A (1996) Hypoallergenicity of rice protein. Cereal Foods World 41:839–843
Jiamyangyuen S, Srijesdaruk V, Harper WJ (2005) Extraction of rice bran protein concentrate and its application in bread extraction. Songklanakarin J Sci Technol 27(1):55–64
Kim SY, Park PSW, Rhee KC (1990) Functional properties of proteolytic enzyme modified soy protein isolate. J Agric Food Chem 38:651–656
Korhonen H (2009) Milk-derived bioactive peptides: from science to applications. J Funct Foods 1:177–187
Korhonen H, Pihlanto A (2006) Bioactive peptides: production and functionality. Int Dairy J 16:945–960
Kumagai T et al. (2006) Production of rice protein by alkaline extraction improves its digestibility. J Nutr Sci Vitaminol 52:467–472
Laemmli UK (1970) Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 227:680–685
Lamsal B, Jung S, Johnson L (2007) Rheological properties of soy protein hydrolysates obtained from limited enzymatic hydrolysis. LWT-Food Science and Technology 40:1215–1223
Panyam D, Kilara A (1996) Enhancing the functionality of food proteins by enzymatic modification. Trends Food Sci Technol 7:120–125
Shih FF, Daigle KW (2000) Preparation and characterization of rice protein isolates. J Am Oil Chem Soc 77:885–889
Silpradit K, Tadakittasarn S, Rimkeeree H, Winitchai S, Haruthaithanasan V (2010) Optimization of rice bran protein hydrolysate production usingalcalase. Asian Journal Food Agriculture-Ind 3:221–231
Tang S, Hettiarachchy N, Horax R, Eswaranandam S (2003) Physicochemical properties and functionality of rice bran protein hydrolyzate prepared from heat-stabilized defatted rice bran with the aid of enzymes. J Food Sci 68:152–157
Tsumura K, Saito T, Tsuge K, Ashida H, Kugimiya W, Inouye K (2005) Functional properties of soy protein hydrolysates obtained by selective proteolysis. LWT-Food Science and Technology 38:255–261
Wang M, Hettiarachchy N, Qi M, Burks W, Siebenmorgen T (1999) Preparation and functional properties of rice bran protein isolate. J Agric Food Chem 47:411–416
Author information
Authors and Affiliations
Corresponding author
Additional information
Research highlights
• Rice bran protein concentrate can be used as a value-added product.
• The hydrolysis was used for the study of rice bran protein concentrate.
• For enzymatic studies, pH stat method and SDS-PAGE method were used.
• The Alcalase® enzyme for substrate hydrolysis was more efficient than the other two enzymes.
• SDS-PAGE could represent protein fraction of hydrolysates.
Rights and permissions
About this article
Cite this article
Ahmadifard, N., Murueta, J.H.C., Abedian-Kenari, A. et al. Comparison the effect of three commercial enzymes for enzymatic hydrolysis of two substrates (rice bran protein concentrate and soy-been protein) with SDS-PAGE. J Food Sci Technol 53, 1279–1284 (2016). https://doi.org/10.1007/s13197-015-2087-6
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13197-015-2087-6