Abstract
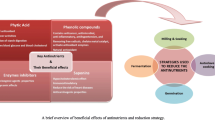
The changes in chemical composition, antioxidant activity and minerals content of horse gram seed after dehulling and germination of 12 advance lines were investigated. Dehulled samples had a higher protein content compared with the raw and germinated. Total soluble sugars (TSS) content increased significantly (p ≤ 0.05) after dehulling (29.31 %) and germination (98.73 %) whereas, the total lipids increased (10.98 %) significantly (p ≤ 0.05) after dehulling and decreased (36.41 %) significantly (p ≤ 0.05) after germination. Dehulling and germination significantly decreased the amount of phytic acid (PA), tannin (TN) and oxalic acid (OA). Trypsin inhibitor units decreased (26.79 %) significantly (p ≤ 0.05) after germination. The minerals (Ca, Fe and Cu) composition of the germinated horsegram flour samples was significantly higher than the raw and dehulled flour. The functional properties of flours were studied and found that the bulk density (11.85 %) and oil absorption capacity (18.92 %) significantly increased after germination. Raw samples followed by germinated samples showed the highest concentrations of phytochemicals responsible for the antioxidant activity and also the antioxidant capacities. principal component analysis revealed that in case of dehulled samples; TN, polyphenols, DPPH and ABTS radical inhibition, TSS, total antioxidant, OA, protein, FRAP value, Ca and Zn had positive correlation among themselves while in case of germinated samples, protein, oil absorption capacity, FRAP value, OA, total flavonoids, DPPH radical inhibition, Ca and Cu had positive correlation among themselves. Present study suggest that germination combined with dehulling process improved quality of horsegram by enhancing the nutritive value and reducing the antinutrients.



Similar content being viewed by others
References
Anderson RA, Conway HF, Pfeifer VF, Griffin EL (1969) Roll and extrusion – cooking of grain sorghum grits. Cereal Sci Today 14:372–380
AOAC (2005) Official methods of analysis, 18th edn. Association of Official Analytical Chemists, Washington
Arnao MB, Cano A, Acosta M (2001) The hydrophilic and lipophilic contribution to total antioxidant activity. Food Chem 73:239–244
Benzie I, Strain J (1996) The ferric reducing ability of plasma (FRAP) as a measure of antioxidant power: the FRAP assay. Anal Biochem 239:70–76
Bligh EG, Dyer WJ (1959) A rapid method of total lipid extraction and purification. Can J Biochem Phys 37(8):911–917
Borhade VP, Kadam SS, Salunke DK (1984) Solubilization and functional properties of mothbean (Vigna aconitifolia marechal) and horsegram (Macrotyloma uniflorum L. Verdc.). J Food Biochem 8:229–35
Brand-Williams W, Cuvelier ME, Berset C (1995) Use of free radical method to evaluate antioxidant activity. LWT - Food Sci Technol 28:25–30
Bravo L, Siddhuraju P, Saura-Calixto F (1999) Composition of underexploited Indian pulses. Comparison with common legumes. Food Chem 64:185–192
El-Adawy TA (2002) Nutritional composition and antinutritional factors of chickpeas (Cicer arietinum L.) undergoing different cooking methods and germination. Plant Food Hum Nutr 57:83–97
Frias J, Fernandez-Orozco R, Zielinski H, Piskula M, Kozlowska H, Vidal-Valverde C (2002) Effect of germination on the content of vitamin C and E of lentils. Polish J Food Nutr Sci 52:76–82
Ghavidel RA, Prakash J (2007) The impact of germination and dehulling on nutrients, antinutrients, in vitro iron and calcium bioavailability and in vitro starch and protein digestibility of some legume seeds. LWT Food Sci Technol 40:1292–1299
Guansheng M, Ying J, Jianhua P, Frans K (2005) Phytate, calcium, iron and zinc contents and their molar ratios in foods commonly consumed in China. J Agr Food Chem 53:10285–10290
Haug W, Lantzsch HJ (1983) Sensitive method for the rapid determination of phytate in cereals and cereal products. J Sci Food Agr 34:1423–1426
Kakade ML, Rackis JJ, Mcghee JE, Puski G (1974) Determination of trypsin inhibitor activity of soy products: a collaborative analysis of an improved procedure. Cereal Chem 51:376–382
Kawsar SMA, Huq E, Nahar N, Ozeki Y (2008) Identification and quantification of phenolic acids in Macrotyloma uniflorum by reversed phase HPLC. Am J Plant Physiol 3:165–172
Lopez A, Montano A, Garcia P, Garrido A (2006) Fatty acid profile of table olives and its multivariate characterization using unsupervised (PCA) and supervised (DA) chemometrics. J Agric Food Chem 54:6747–6753
Malleshi NG, Daodu MA, Chandrasekhar A (1989) Development of weaning food formulations based on malting and roller drying of sorghum and cowpea. Int J Food Sci Technol 24(5):519
Mishra KK, Pal RS, Arun KR, Chandrashekara C, Jain SK, Bhatt JC (2013) Antioxidant properties of different edible mushroom species and increased bioconversion efficiency of Pleurotus eryngii using locally available casing materials. Food Chem 138:1557–1563
Murugkar DA, Gulati P, Gupta C (2013) Effect of sprouting on physical properties and functional and nutritional components of multi-nutrient mixes. Int J Food Nutr Sci 2(2):2–15
Petchiammal C, Hopper W (2014) Antioxidant activity of proteins from fifteen varieties of legume seeds commonly consumed in India. Int J Pharm Pharm Sci 6(l 2):476–479
Prieto P, Pineda M, Aguilar M (1999) Spectrophotometric quantitation of antioxidant capacity through the formation of a Phosphomolybdenum complex: specific application to the determination of vitamin E. Anal Biochem 269:337–341
Ramadan EA (2012) Effect of processing and cooking methods on the chemical composition, sugars and phytic acid of soybeans. Food Public Health 2(1):11–15. doi:10.5923/j.fph.20120201.03
Rao BSN, Prabhavathi T (1982) Tannin content of foods commonly consumed in India and its influence on ionisable iron. J Sci Food Agr 33:89–96
Sangronis E, Machado CJ (2007) Influence of germination on the nutritional quality of Phaseolus vulgaris and Cajanus cajan. J Food Sci Agri Technol 40(1):116–120
Satwadhar PN, Kadam SS, Salunkhe DK (1981) Effects of germination and cooking on polyphenols and in vitro digestibility of horsegram and moth bean. Plant Foods Hum Nutr 31:71–76
Singleton VL, Rossi JA (1965) Colorimetry of total phenolics with phosphomolybdic-phosphotungstic acid reagents. Am J Enol Vitic 16:144–158
Sosulski FW (1962) The centrifuge method for determining flour absorption in hard red spring wheat. Cereal Chem 39:344–350
Sreerama YN, Sashikala VB, Pratape VM (2012a) Phenolic compounds in cowpea and horsegram flours in comparison to chickpea flour: evaluation of their antioxidant and enzyme inhibitory properties associated with hyperglycemia and hypertension. Food Chem 133:156–162
Sreerama YN, Sashikala VB, Pratape VM, Singh V (2012b) Nutrients and antinutrients in cowpea and horsegram flours in comparison to chickpea flour: evaluation of their flour functionality. Food Chem 131:462–468
Sudha N, Mushtari Begum J, Shambulingappa KG, Babu CK (1995) Nutrients and some anti-nutrients in horsegram (Macrotyloma uniflorum (Lam.) Verdc.). Food Nutr Bull 16(1):81–83
Sushma D, Yadav BK, Tarafdar JC (2008) Phytate phosphorus and mineral changes during soaking, boiling and germination of legumes and pearl millet. J Food Sci Technol 45(4):344–348
Tiwari AK, Manasa K, Kumar DA, Zehra A (2013) Raw horsegram seeds possess more in vitro antihyperglycaemic activities and antioxidant properties than their sprouts. Nutrafoods 12:47–54
Torres A, Frias J, Granito M, Vidal-Valverde C (2007) Germinated Cajanus cajan seeds as ingredients in pasta products: chemical, biological and sensory evaluation. Food Chem 101(1):202–211
Vidal-Valverde C, Frias J, Sierra I, Blazquez I, Lambien F, Kuo YH (2002) New functional legume food by germination: effect on the nutritive value of beans, lentils and peas. Eur Food Res Technol 215:472–476
Wang C, Kinsella JE (1976) Functional properties of novel proteins, AIfaIfa leaf protein. J Food Sci 41:286–292
Wang N, Lewis MJ, Brennan JG, Westby A (1997) Effect of processing methods on nutrients and antinutritional factors in cowpea. Food Chem 58:59–68
Acknowledgments
The authors are grateful to Indian Council of Agricultural Research (ICAR), for financial support to carry out this work at Vivekanand Parvatiya Krishi Anusandhan Sansthan (VPKAS), Almora (Uttarakhand) 263601.
Conflict of interest
The authors declare that there are no conflicts of interest.
Author information
Authors and Affiliations
Corresponding author
Additional information
Highlights
➢ Dehulling improves the protein, sugars and lipids content, significantly.
➢ Tannin, oxalic acid and phytic acid reduced significantly in dehulled samples over raw.
➢ Germination improved the nutrients content in horsegram, significantly.
➢ Anti-nutritive compounds were significantly decreased after germination.
➢ Antioxidant capacities decreased significantly after dehulling and germination.
Rights and permissions
About this article
Cite this article
Pal, R.S., Bhartiya, A., ArunKumar, R. et al. Impact of dehulling and germination on nutrients, antinutrients, and antioxidant properties in horsegram. J Food Sci Technol 53, 337–347 (2016). https://doi.org/10.1007/s13197-015-2037-3
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13197-015-2037-3